 |
 |
 |
| |
|
Pharmacokinetic and Pharmacodynamic Evaluation of the Antiretroviral Compound MK-2048 Released From an Intravaginal Ring in Rhesus Macaques
|
| |
| |
James Cummins
Division of AIDS, NIAID, NIH, United States
Reported by Jules Levin
HIVR4P 2019 Oct 21-25 Madrid
WEBCAST:
http://webcasts.hivr4p.org/console/player/40419?mediaType=slideVideo&&crd_fl=1&ssmsrq=1540648681480&ctms=5000&csmsrq=1138

program abstract
Pharmacokinetic and Pharmacodynamic Evaluation of the Antiretroviral Compound MK-2048 Released From an Intravaginal Ring in Rhesus Macaques
Ranajit Pal1, H.K. Chung1, Anabel Lowry2, Leslie Marshall2, Johnathan Misamore3, Sharon Orndorff1, Natasha Parker2, Nicola Richardson-Harman4, Julie Strizki5, Glenn Swartz1, Ye Tian5, Jim Turpin2, Fulvia Veronese2, Deborah Weiss3, James Cummins2 1Advanced BioScience Laboratories Inc., United States, 2NIAID, NIH, United States, 3BioQual Inc., United States, 4Alpha StatConsult LLC, United States, 5Merck & Co., Inc., United States
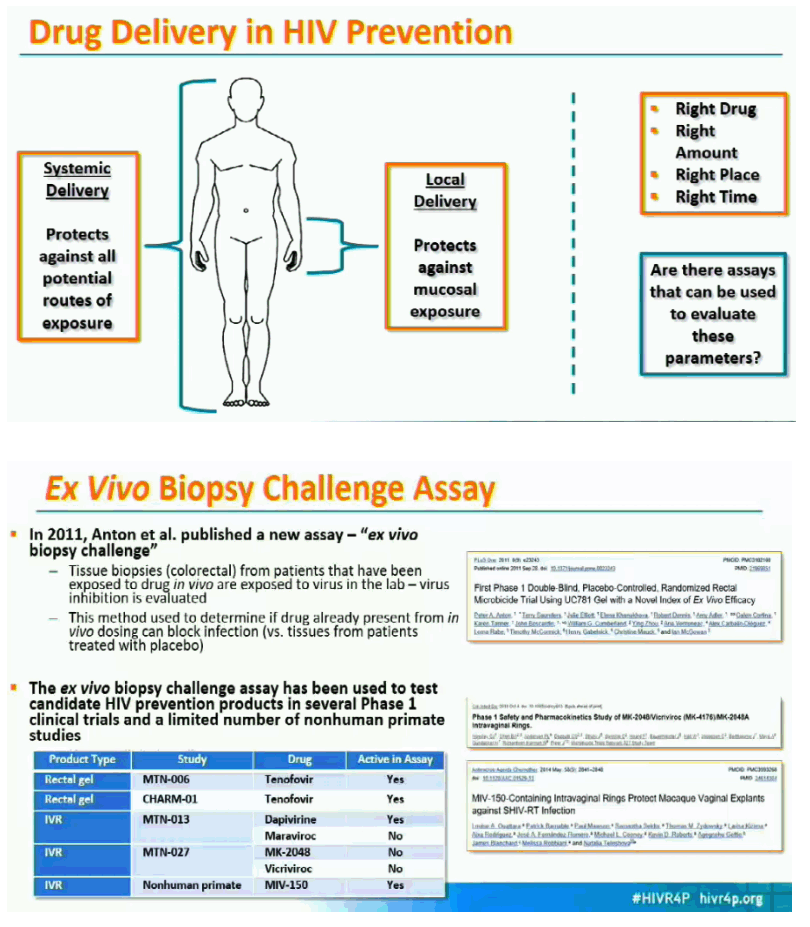
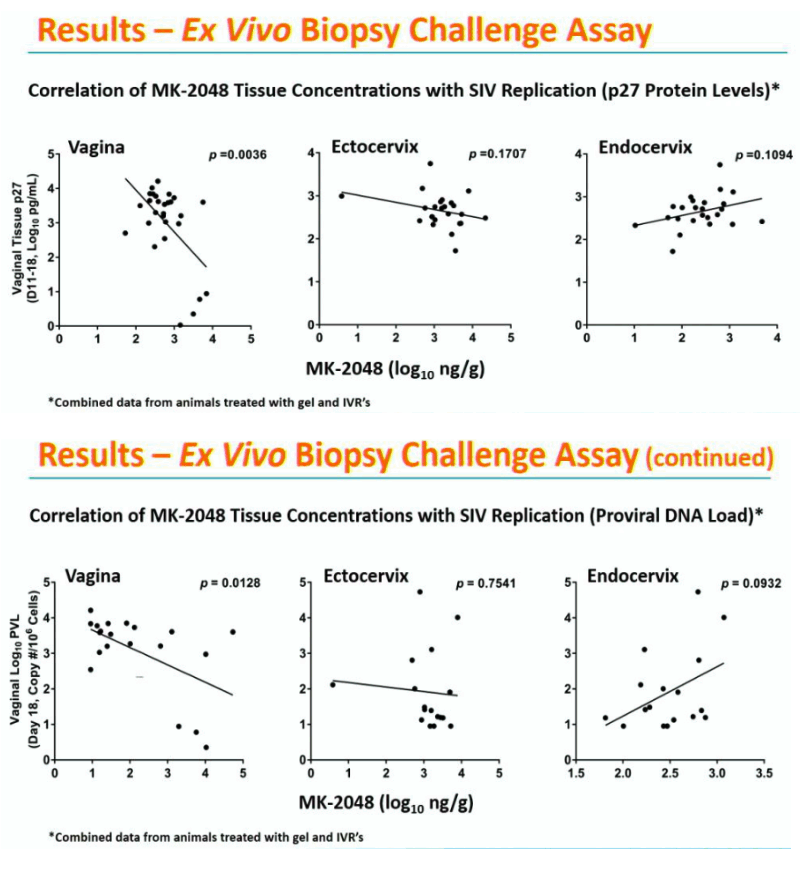
Background: Rhesus macaques (RM) may be a useful model for evaluating the pharmacokinetic-pharmacodynamic (PK-PD) properties of drug-containing intravaginal rings (IVRs) before they are tested in humans. RM were treated with IVRs containing the HIV-1 integrase inhibitor MK-2048 (n=30) or placebo (n=4), and evaluated for in vivo drug release from the IVRs, PK profiles in plasma, secretions, and tissues, and ex vivo vaginal biopsy challenge with SIV.
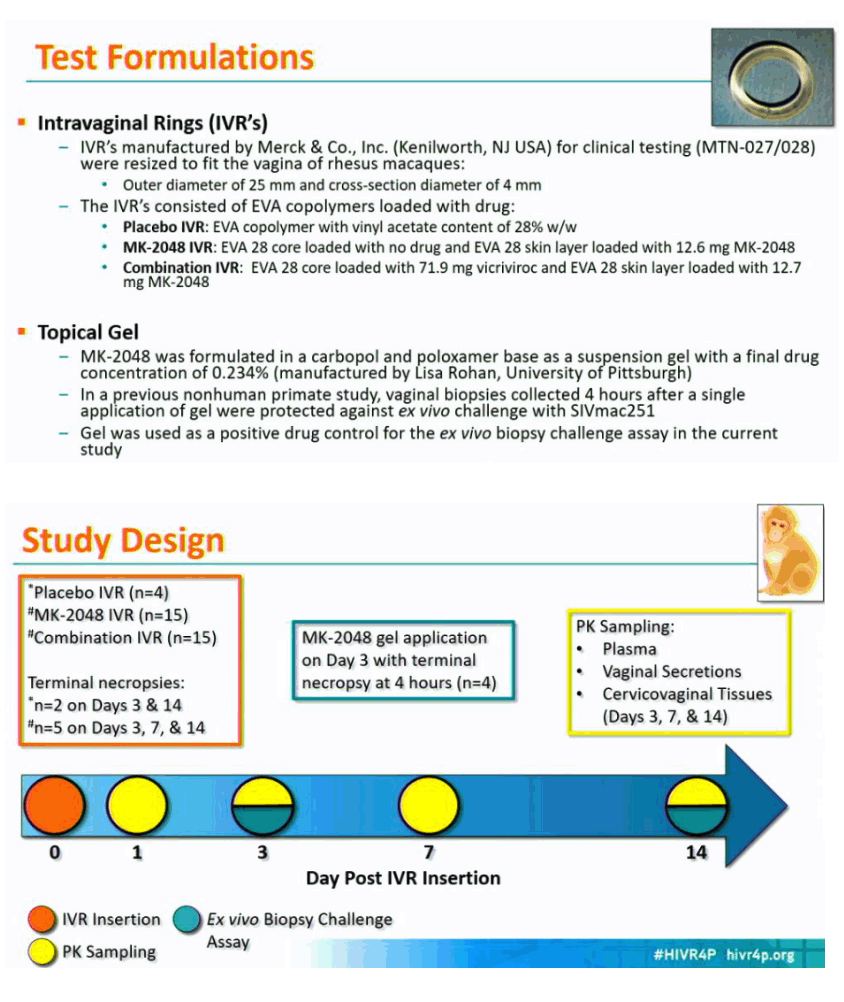
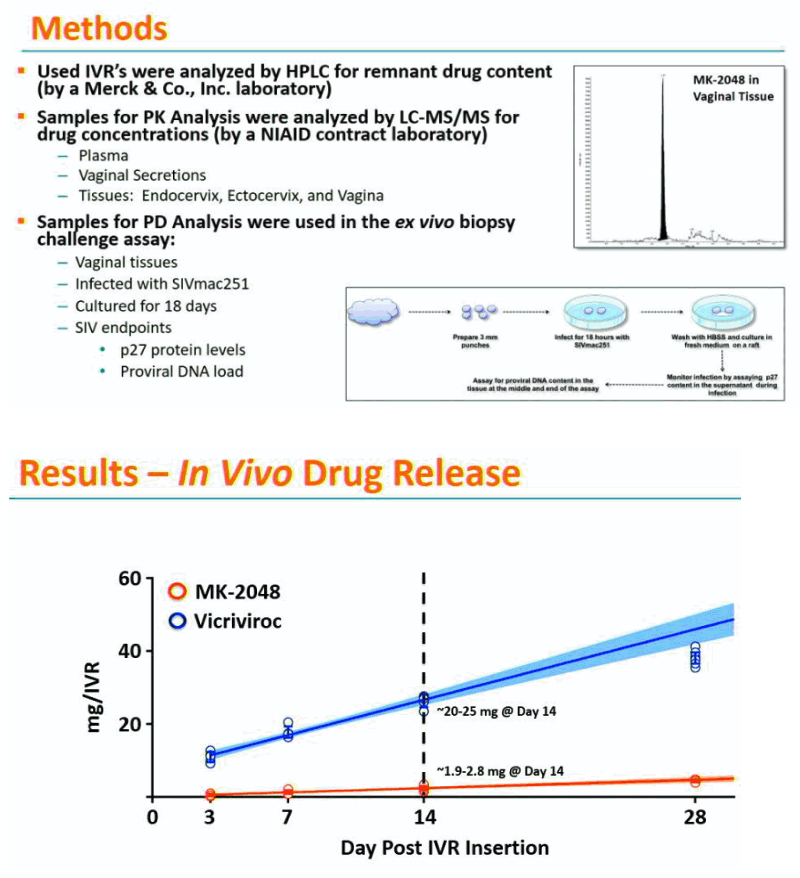
Methods: IVRs containing MK-2048 alone (12.6 mg) or in combination with vicriviroc (VCV; 12.7 mg MK-2048; 71.9 mg VCV) were inserted and retained in the RM for 14 days. Blood, vaginal secretions, and reproductive tract (RT) tissues were collected at multiple time points after insertion. Drug concentrations in study samples and IVR remnant content were determined by LC-MS/MS and HPLC, respectively. Vaginal biopsies were challenged ex vivo with SIVmac251, and viral infection was detected by proviral DNA load and SIV p27 antigen.
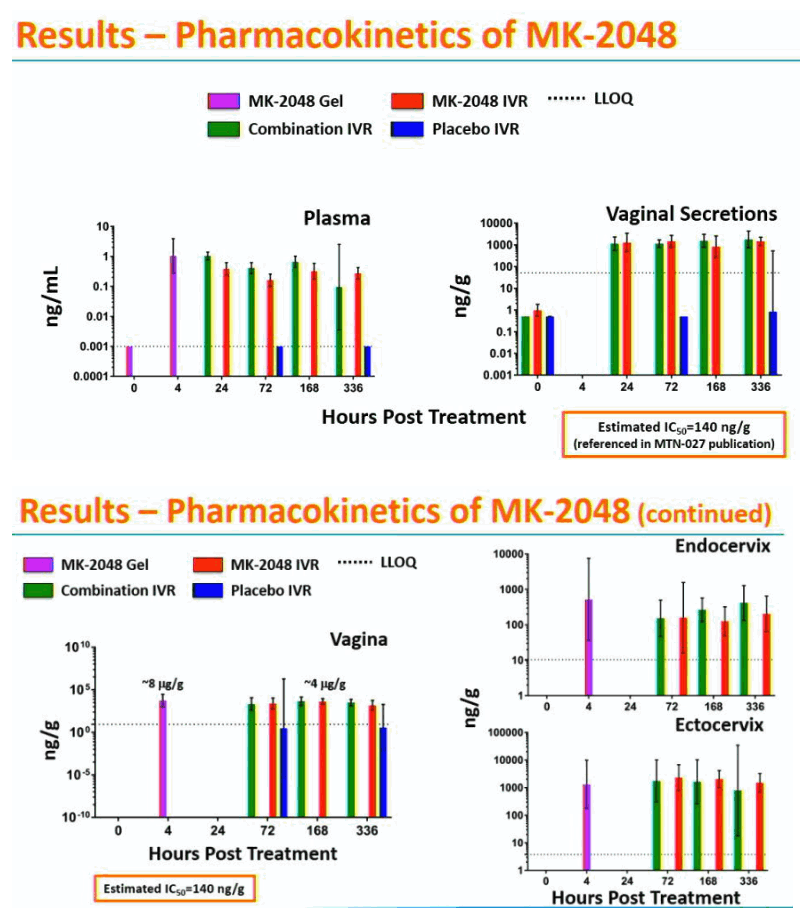
Results: Remnant analyses indicated a linear in vivo release profile for MK-2048 (geomean ±95%CI mg/IVR): 0.49 (0.31-0.79) on Day 3 and 2.33 (1.93-2.83) on Day 14. Plasma MK-2048 levels peaked at 24-168 hours and ranged from 0.04-2.96 ng/mL. Vaginal secretion MK-2048 levels peaked at 24-336 hours and ranged from 198-19435 ng/mL. MK-2048 concentrations in plasma, RT tissues, and vaginal secretions were generally positively correlated (r=0.43-0.90; p< 0.05). Vaginal MK-2048 concentrations were negatively correlated with the viral PD endpoints, demonstrating inhibition of virus replication in the biopsy challenge (r=-0.53 to -0.89; p< 0.05).
Conclusions: These results indicate the utility of the RM model for evaluating the PK of MK-2048 and confirming its antiviral activity in vaginal tissues. Given the limits around tissue sampling in human clinical trials and no surrogate markers of protection, the ability to determine PK-PD properties in RM may prove useful in the preclinical development of IVRs and other biomedical HIV prevention products.



|
| |
|
 |
 |
|
|