 |
 |
 |
| |
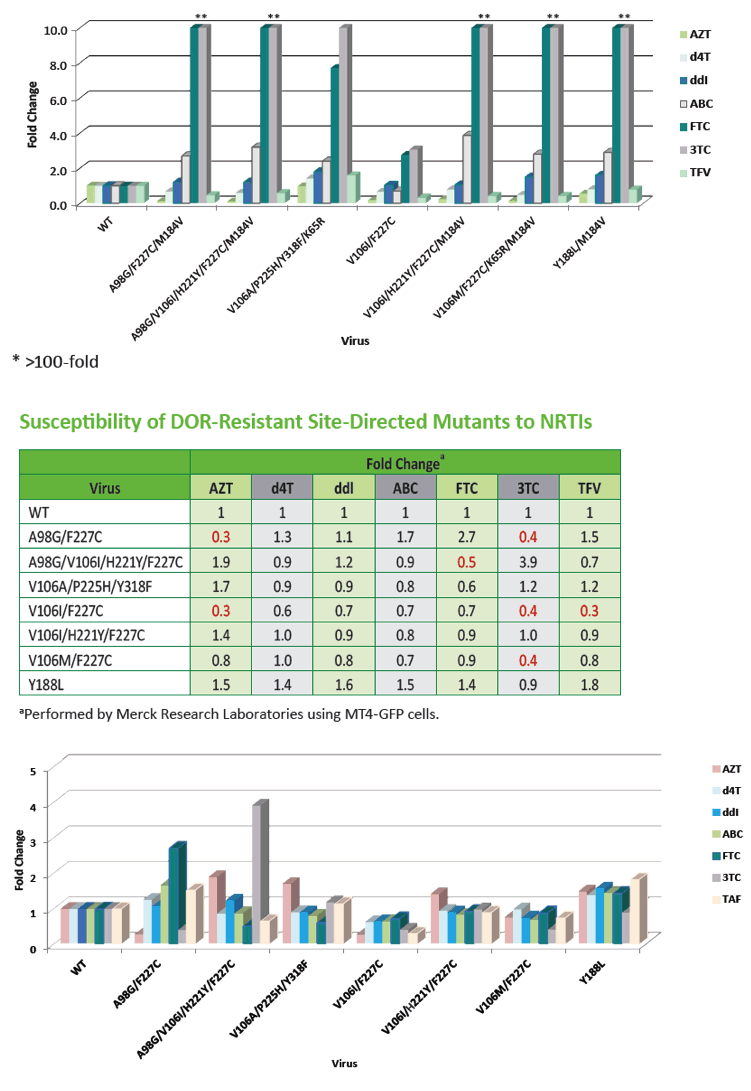
Characterization of Doravirine-Selected Resistance Patterns
From Participants in Treatment-Naïve Phase 3 Clinical Trials
|
| |
| |
Reported by Jules Levin
22nd International AIDS Conference; Amsterdam, The Netherlands; 23-27 July 2018
Ming-Tain Lai1; Min Xu2; Winnie Ngo1; Meizhen Feng1; Donald Graham1; Jinqing Wang1; Daria J. Hazuda1; George J. Hanna3; Sushma Kumar4; Xia Xu5; Elizabeth Martin3; Carey Hwang3
1Department of Antiviral Research, 2Department of Pharmacology, 3Department of Clinical Research, 4Department of Clinical Sciences and Study Management, 5Department of Biostatistics, Merck Research Laboratories (West Point, PA), Merck & Co., Inc., Kenilworth, NJ, USA
IAC: Doravirine (DOR) Versus Ritonavir-Boosted Darunavir (DRV+r): 96-Week Results of the Randomized, Double-Blind, Phase 3 DRIVE-FORWARD Noninferiority Trial - (07/25/18)
Doravirine Studies - efficacy, safety / FDA NDA Accepted
FDA Accepts New Drug Applications for Merck's Doravirine, the Company's Investigational Non-Nucleoside Reverse Transcriptase Inhibitor (NNRTI), for Treatment of HIV-1 Infection

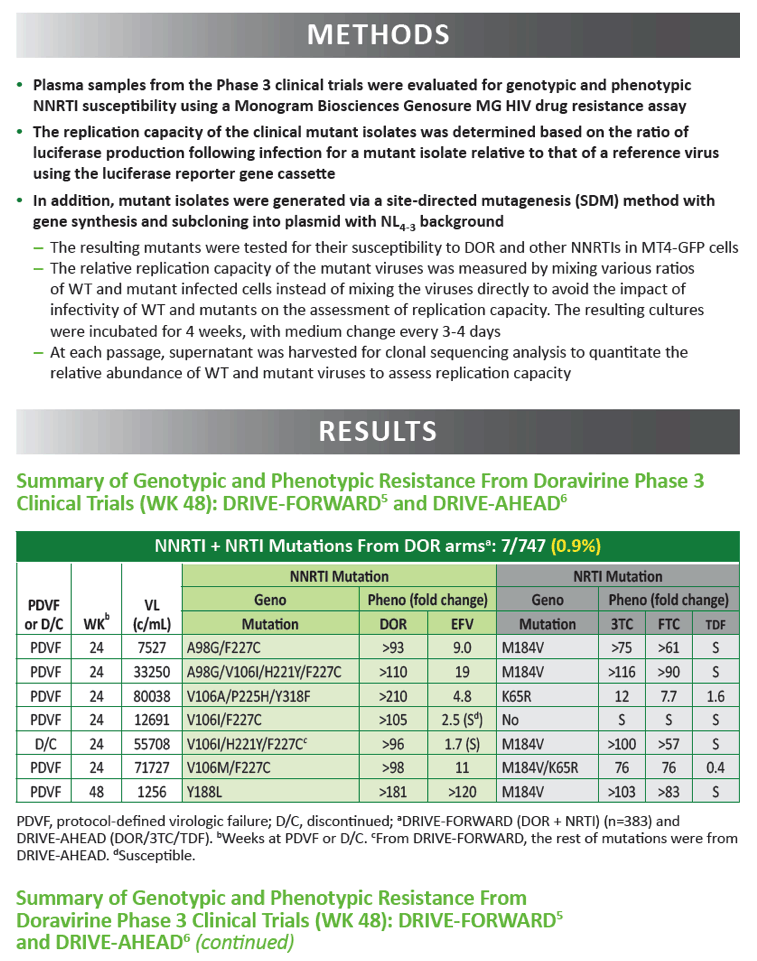
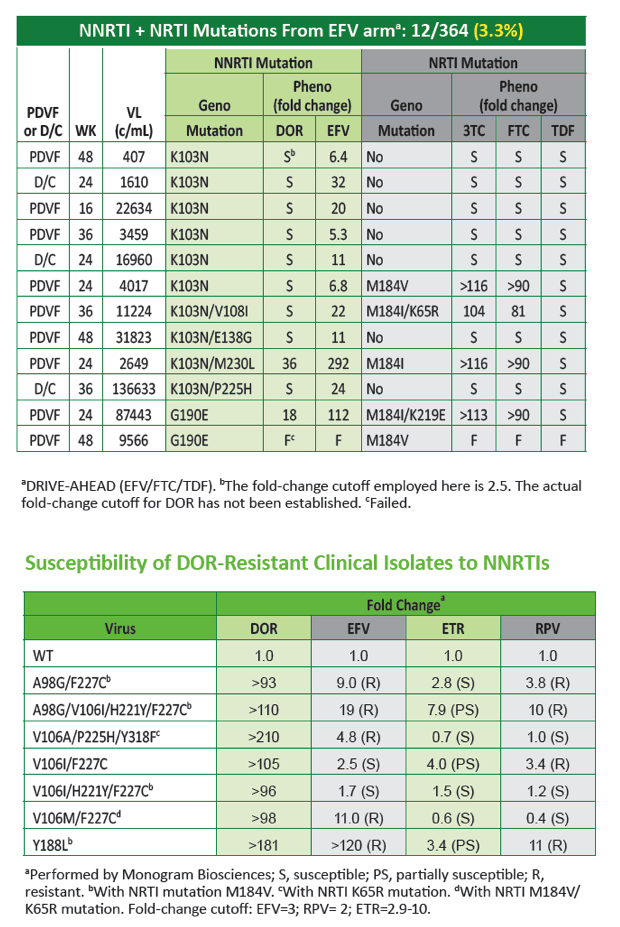
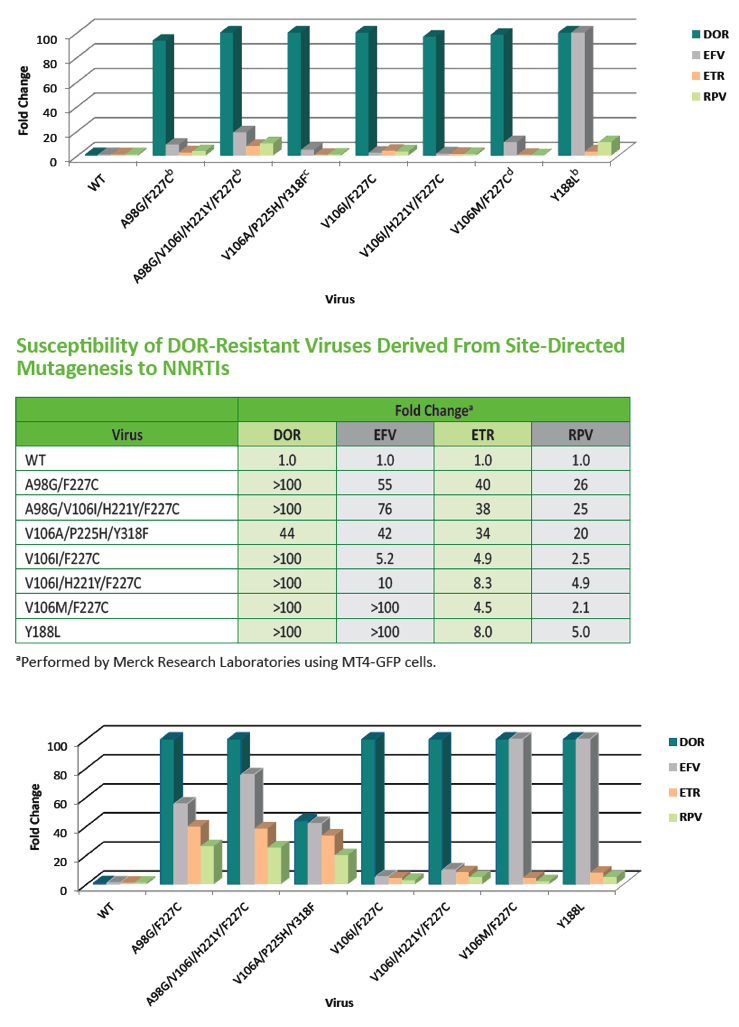
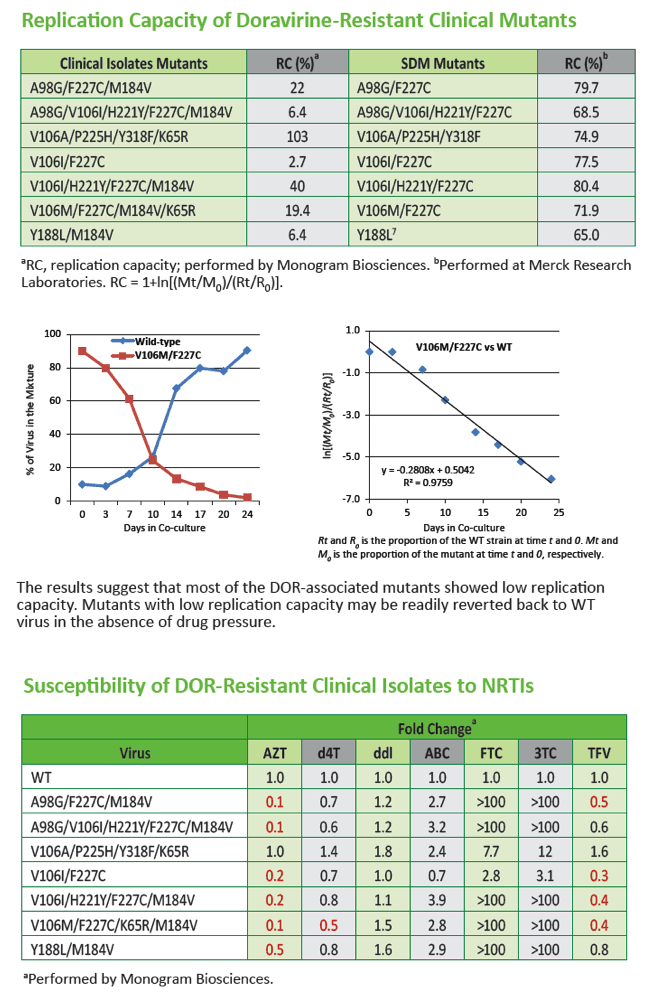

|
| |
|
 |
 |
|
|