 |
 |
 |
| |
Adherence to Long-Acting CAB and RPV Injections Through 96 Weeks of Maintenance Therapy in LATTE-2 / Good adherence to injected cabotegravir and rilpivirine in 230 LATTE-2 participants
|
| |
| |
22nd International AIDS Conference, Amsterdam, Netherlands, July 23-27, 2018
Mark Mascolini
A big majority of 230 phase 2 trial participants received their monthly or every-2-month long-acting cabotegravir (CAB) and rilpivirine (RPV) injections on time [1]. Patient questionnaire results confirmed high adherence to this novel every-4-week or every-8-week regimen for chronic HIV infection.
LATTE-2 tested the antiviral activity and safety of combined injections of the integrase inhibitor CAB and the nonnucleoside RPV in previously untreated people with HIV [2]. All participants started with 20 weeks of an oral regimen including CAB plus abacavir/lamivudine (ABC/3TC). Those with a viral load below 50 copies at week 20 were randomized 2-to-2-to-1 to injected CAB/RPV every 4 weeks, injected CAB/RPV every 8 weeks, or continued oral therapy. After 96 weeks, 94% in the 8-week group, 87% in the 4-week group, and 84% in the oral group maintained viral suppression.
Because monthly or every-2-month injections eliminate the need for daily antiretroviral dosing, simplified adherence is a compelling rationale for long-acting injectable therapy. But the need for a monthly or every-other-month dose does not make adherence automatic, as studies of injected hormonal contraceptives show [3]. Thus verifying good adherence to a regimen like injected CAB/RPV is important, especially since a missed monthly dose may have worse consequences than one or two missed daily doses.
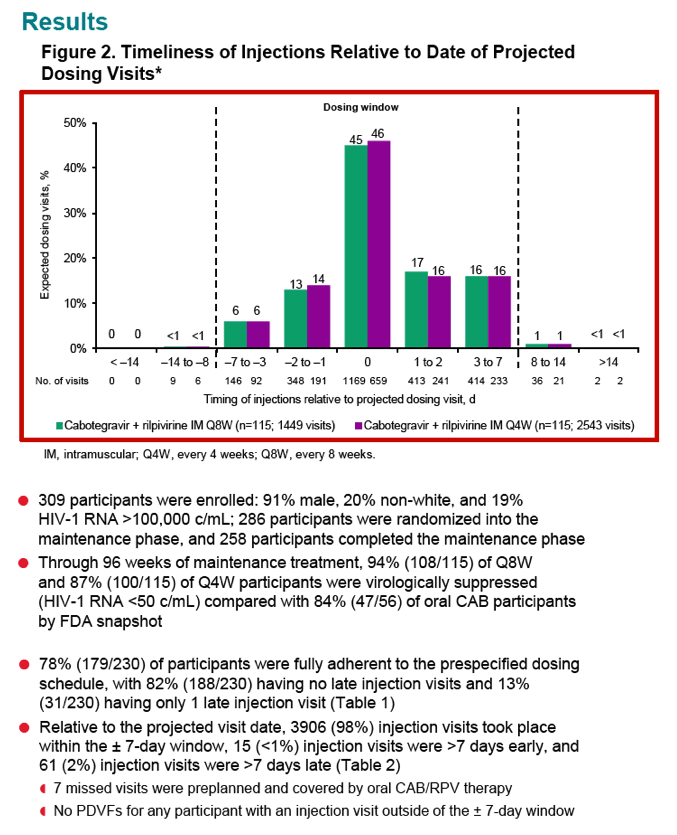
LATTE-2 investigators calculated adherence to injected CAB/RPV as the proportion of injection visits kept within a +/- 7-day window around the expected visit up to week 96. Researchers projected target injection visit dates from the day-1 visit date. Trial participants completed the HIV Medication Questionnaire, which asks 6 questions about each injection and oral dose.
Among 309 adults enrolled in LATTE-2, 91% were men, 20% nonwhite, and 19% had a pretreatment viral load above 100,000 copies. Researchers randomized 286 people after the oral lead-in phase and 258 completed the maintenance phase.
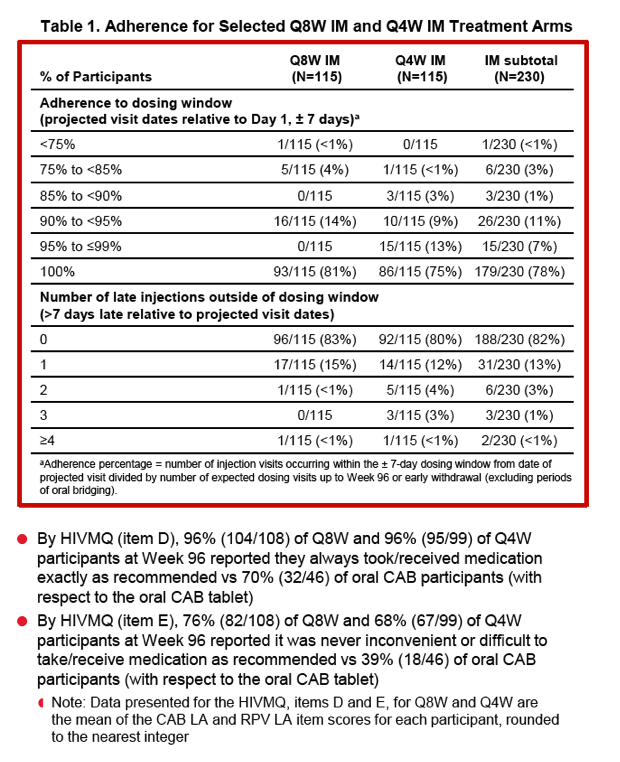
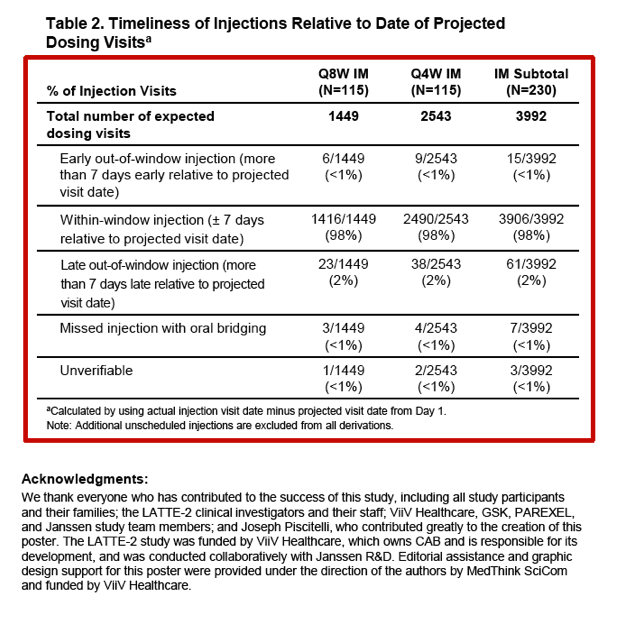
The adherence analysis involved 230 people, of whom 188 (82%) had no late injection visits and 31 (13%) had only 1 late visit. Among 3992 injection visits, 3906 (98%) took place within 7 days of the projected visit date. Seven missed injection visits were preplanned and covered by oral CAB/RPV. No participants with a visit outside the +/- 7-day window had a protocol-defined virologic failure.
The adherence questionnaire showed that 96% receiving every-8-week injections, 96% receiving every-4-week injections, and 70% who stayed with oral dosing reported they always received or took CAB/RPV exactly as recommended. Three quarters of the every-8-week group, two thirds of the every-4-week group, and 39% of the oral group said they never found it inconvenient to receive or take their medication.
The researchers believe these results "suggest that long-acting injectable therapy [with CAB/RPV] may provide an alternate, directly observed therapeutic option" for people with HIV infection.
References
1. Sutton K, Shaefer M, Margolis D, et al. Patient adherence to long-acting CAB and RPV injections through 96 weeks of maintenance therapy in LATTE-2. AIDS 2018: 22nd International AIDS Conference, Amsterdam, Netherlands, July 23-27, 2018. Abstract THPEB084. https://programme.aids2018.org//PAGMaterial/eposters/10659.pdf
2. Margolis DA, Gonzalez-Garcia J, Stellbrink HJ, et al. Long-acting intramuscular cabotegravir and rilpivirine in adults with HIV-1 infection (LATTE-2): 96-week results of a randomised, open-label, phase 2b, non-inferiority trial. Lancet. 2017;390:1499-1510. http://www.natap.org/2017/IAS/IAS_23.htm
3. Murphy PA, Brixner D. Hormonal contraceptive discontinuation patterns according to formulation: investigation of associations in an administrative claims database. Contraception. 2008;77:257-263.



|
| |
|
 |
 |
|
|