 |
 |
 |
| |
Safety of in utero antiretroviral (ARV) exposure:
neurologic outcomes in HIV-exposed, uninfected children
|
| |
| |
In Utero Efavirenz--and Perhaps Dolutegravir--Tied to Neurotoxicity in HIV-Negative Children
IDWeek 2018, October 3-7, 2018, San Francisco
Mark Mascolini
HIV-exposed but uninfected (HEU) children exposed to efavirenz in utero had a 60% higher risk of neurologic abnormalities in a 3747-child observational cohort [1]. The study tentatively linked dolutegravir exposure to neurotoxicity, but these statistical analyses were tenuous because of the small number of cases observed so far.
Pediatric HIV/AIDS Cohort Study (PHACS) researchers who conducted this study noted that prior work detected a higher risk of mitochondrial dysfunction in HEU children than in HIV-unexposed uninfected children. Some HEU children in these analyses had neurologic symptoms including seizures, febrile seizures, and cognitive delay.
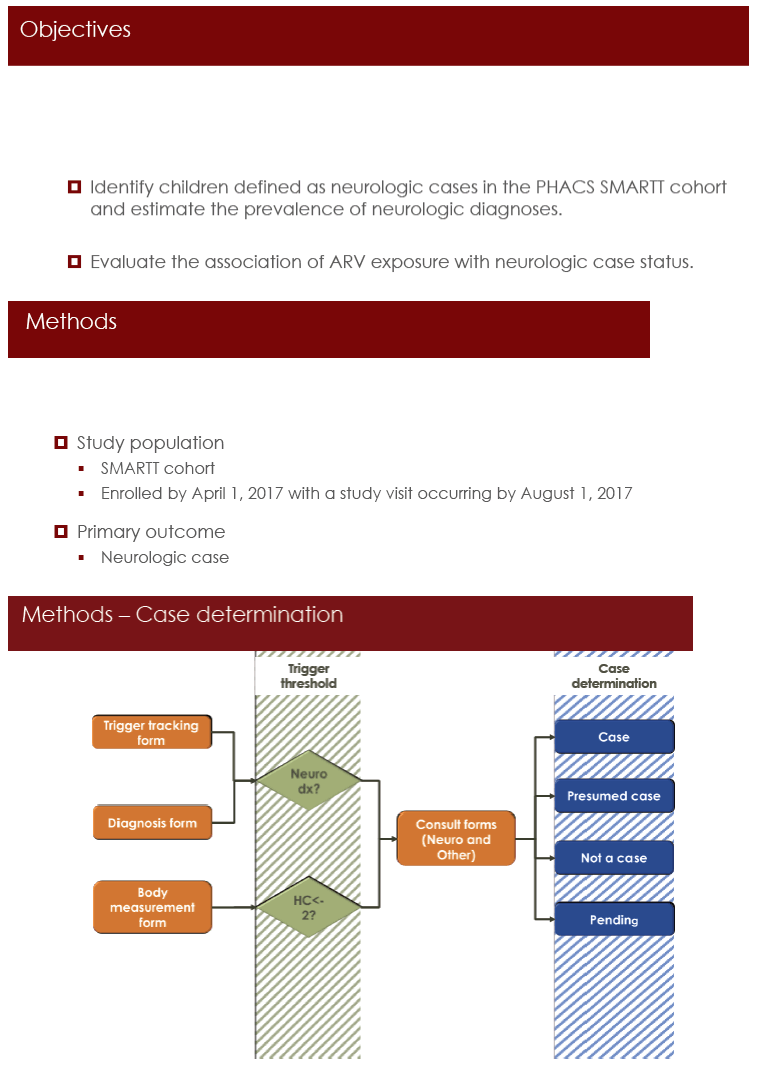
The PHACS team undertook this analysis to identify neurologic problems in HEU children enrolled in the Surveillance Monitoring for ART Toxicities (SMARTT) study, a longitudinal observational cohort. The study group included children enrolled in SMARTT by April 1, 2017 who had a study visit up to August 1, 2017. The primary outcome was a neurologic condition (microcephaly, febrile seizures, seizure disorders, ophthalmologic disorders, other neurologic conditions) determined by reviewers blinded to antiretroviral exposure. Comparison groups consisted of children unexposed to a specific antiretroviral class or specific antiretrovirals. The researchers used log-binomial regression to calculate relative risks of neurologic conditions with antiretroviral exposure after adjustment for relevant maternal and infant factors. Sensitivity analyses considered follow-up time or excluded children with major congenital abnormalities.
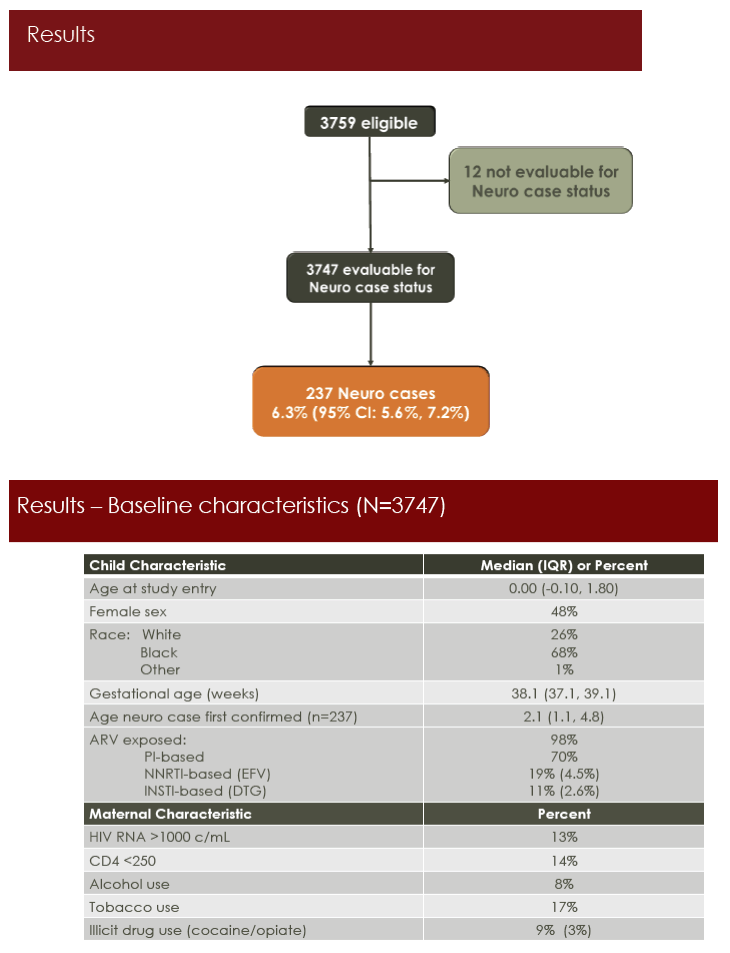
Of the 3747 children evaluable for neurologic conditions, 48% were girls, 68% black, and 26% white. While 70% of children had in utero exposure to protease inhibitors, 19% had exposure to nonnucleosides (4.5% efavirenz) and 11% to integrase inhibitors (2.6% dolutegravir). Maternal risks included tobacco use by 17%, alcohol use by 8%, and illicit drug use by 9%.
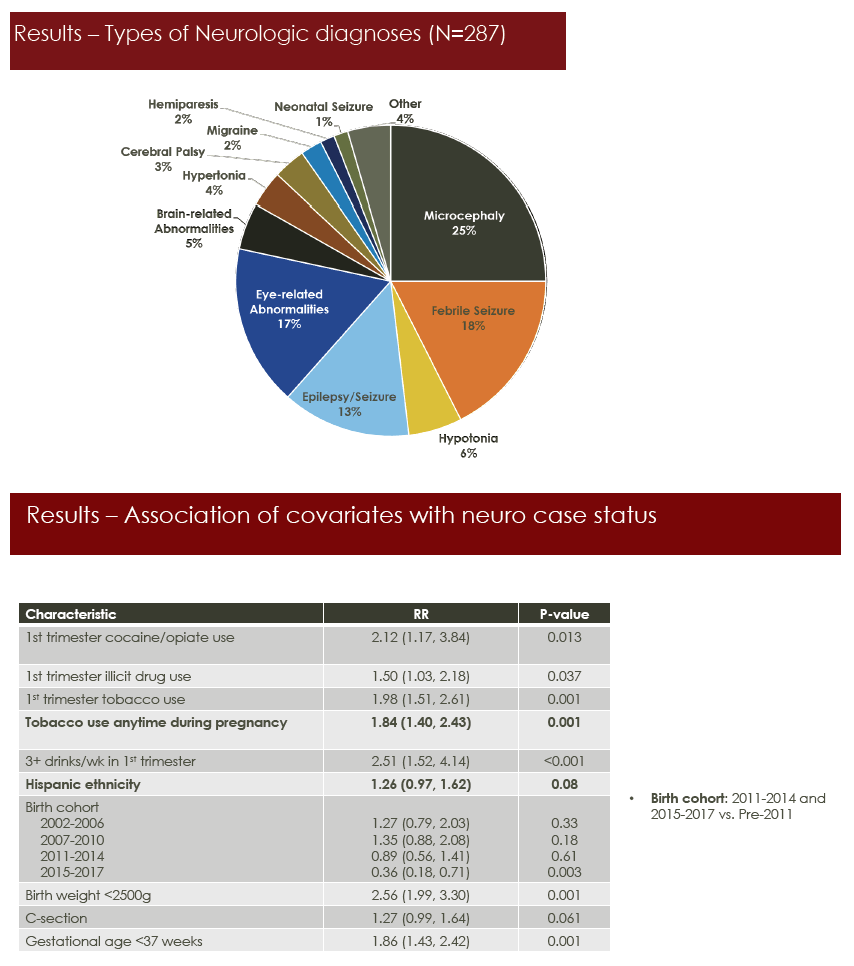
Among evaluable children, 237 (6.3%) had a neurologic abnormality, usually microcephaly (25%), febrile seizure (18%), eye-related abnormalities (17%), or epilepsy/seizure (13%). Tobacco use any time during pregnancy almost doubled the risk of a neurologic condition in HEU children (relative risk [RR] 1.84, 95% confidence interval [CI] 1.40 to 2.43, P = 0.001). There was a trend toward higher neurologic risk with Hispanic ethnicity (RR 1.26, 95% CI 0.97 to 1.62, P = 0.08).
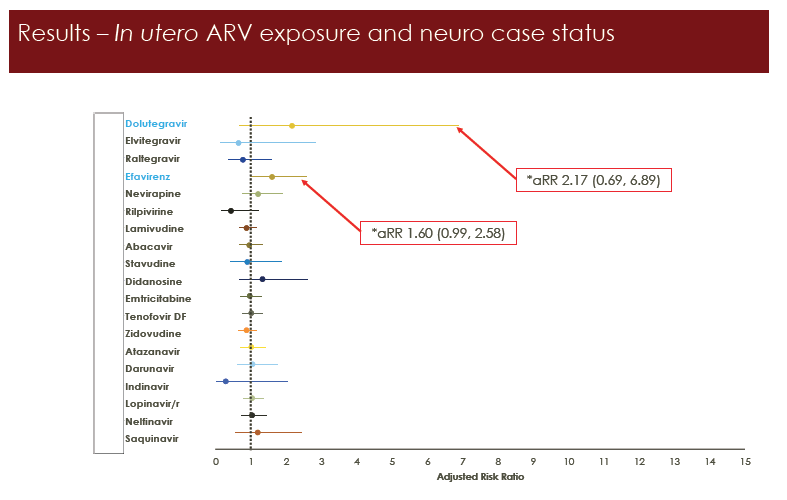
In the primary analysis, adjusted relative risk for neurologic conditions nearly reached significance with efavirenz exposure (adjusted relative risk [aRR] 1.60, 95% CI 0.99 to 2.58). The association with dolutegravir was weaker (aRR 2.17, 95% CI 0.69 to 6.89). In 4 sensitivity analyses, the association with efavirenz exposure reached statistical significance: excluding congenital abnormalities (aRR 1.64, 95% CI 1.02 to 2.64), first 2 years of follow-up (aRR 1.87, 95% CI 1.06 to 3.29), up to age 2 (aRR 2.20, 95% CI 1.21 to 3.99), and exposure before conception versus never* (aRR 1.88, 95% CI 1.07 to 3.29) (all P < 0.05). Associations with dolutegravir did not reach statistical significance in any of 7 sensitivity analyses, but trends always leaned toward a positive association with dolutegravir, at aRRs from 1.52 to 3.15.
The association between in utero efavirenz exposure and neurologic conditions in HIV-negative children--confirmed in several sensitivity analyses--has fading clinical import as efavirenz sees waning overall use and has long been shunned during pregnancy because of potential teratogenicity [2]. The potential link between dolutegravir exposure and neurologic abnormalities in HEU is more troubling because of wide use of this integrase inhibitor. But the hints of an association in this study, the PHACS team said, "were not statistically stable due to [the] small number of cases" and so require confirmation in more HEU children.
*HEU children exposed to the antiretroviral of interest before conception versus those never exposed to that antiretroviral (but possibly exposed to other antiretrovirals).
References
1. Crowell CS, Williams P, Yildirim C, et al. Safety of in utero antiretroviral (ARV) exposure: neurologic outcomes in HIV-exposed, uninfected children. IDWeek 2018, October 3-7, 2018, San Francisco. Abstract LB5.
2. Hsu HE, Rydzak CE, Cotich KL, et al. Quantifying the risks and benefits of efavirenz use in HIV-infected women of childbearing age in the USA. HIV Med. 2011;12:97-108. https://www.ncbi.nlm.nih.gov/pubmed/20561082
The PHACS SMARTT Study: Assessment of the Safety of In Utero Exposure to Antiretroviral Drugs
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4876360/
The Surveillance Monitoring of ART Toxicities Study is a prospective cohort study conducted at 22 US sites to evaluate safety of in-utero antiretroviral drug exposure in HIV-uninfected children born to HIV-infected mothers. Children meeting predefined clinical or laboratory thresholds have more intensive evaluations to determine whether they meet criteria for adverse events. Both cohorts opened to participating sites in the United States including Puerto Rico in March 2007





|
| |
|
 |
 |
|
|