 |
 |
 |
| |
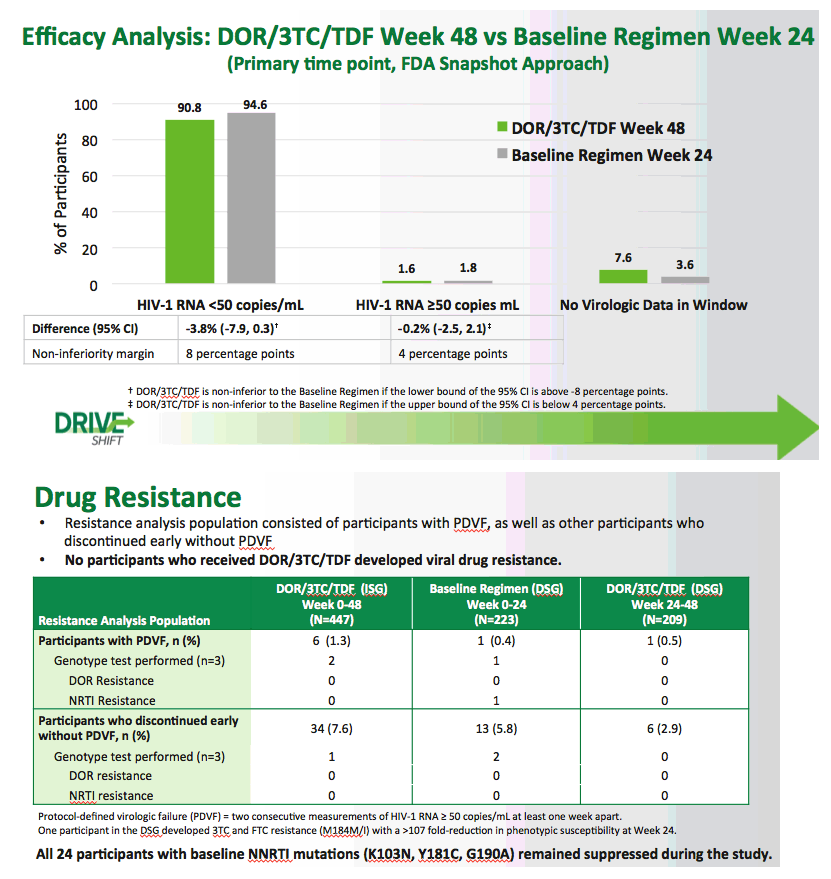
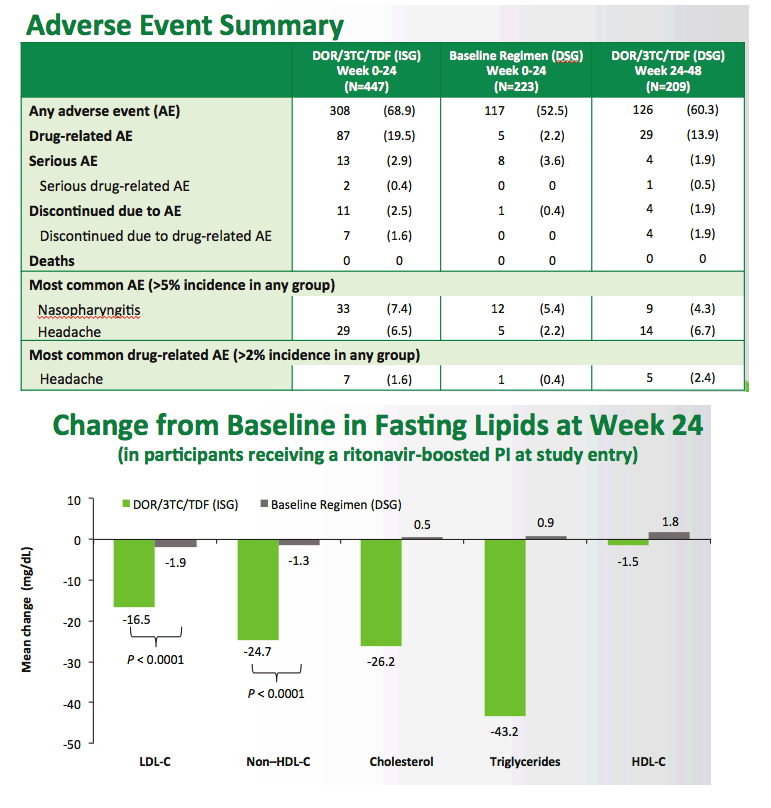
Switch to Doravirine/Lamivudine/Tenofovir Disoproxil Fumarate (DOR/3TC/TDF) Maintains Virologic Suppression Through 48 Weeks: Results of the DRIVE-SHIFT Trial
|
| |
| |
IDWeek 2018, October 3-7, 2018, San Francisco
Princy Kumar1, Margaret Johnson2, Jean-Michel Molina3, Giuliano Rizzardini4, Pedro Cahn5, Markus Bickel6, Josep Mallolas7, Yan Zhou8, Cristiana Morais8, Sushma Kumar8, Peter Sklar8, George J Hanna8, Carey Hwang8, Wayne Greaves8, for the DRIVE-SHIFT Study Group
1Georgetown University, Washington DC; 2Royal Free Hospital, London, UK; 3University of Paris Diderot and Hôpital Saint-Louis, Paris, France; 4Fatebenefratelli Sacco Hospital, Milan, Italy; 5Fundación Huesped, Buenos Aires, Argentina; 6Infektiologikum, Frankfurt, Germany; 7University of Barcelona, Barcelona, Spain; 8Merck & Co., Inc., Kenilworth NJ, USA






|
| |
|
 |
 |
|
|