 |
 |
 |
| |
Only 30% With HCV/HIV in Safety-Net System Get HCV Therapy in DAA Era - Hepatitis C eradication: who is being left behind in the HIV population?
|
| |
| |
IDWeek 2018, October 3-7, 2018, San Francisco
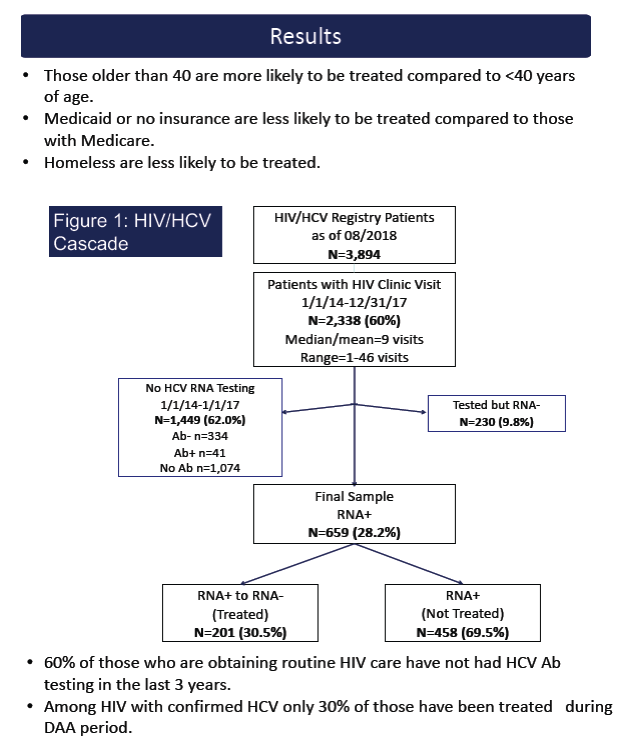
from Jules: in this current era where guidelines say all HIV+ should be screened for HCV this study in a major US city Dallas reports only 30% get DAA therapy, 60% were not screened for HCV, 66% had undetectable HIV viral load, 60% of those who are obtaining routine HIV care have not had HCV Ab testing in the last 3 years, the most vulnerable: Medicaid or no insurance are less likely to be treated compared to those with Medicare.
Mark Mascolini
Only 30% of HIV-positive people with confirmed HCV got treated for HCV during the direct-acting antiviral (DAA) era in a large Texas safety-net hospital system [1]. People under 40 and those without insurance or using Medicaid were less likely to receive DAAs.
Although HCV cure rates with DAAs exceed 90% regardless HIV infection, University of Texas Southwestern Medical Center researchers noted that disparities in access to DAAs persist [2,3]. Because little is known about DAA access in people with HIV, the Dallas investigators conducted this study, retrospectively analyzing all patients with HCV and HIV who received outpatient care in the hospital system from January 2014 through December 2017. Everyone had HIV before January 2014, and all tested positive for HCV antibody or HCV RNA or had an HCV diagnosis code between January 2014 and December 2017. The researchers used multivariable logistic regression to identify predictors of HCV treatment, adjusting for all predictors with a P < 0.20 in univariate models.
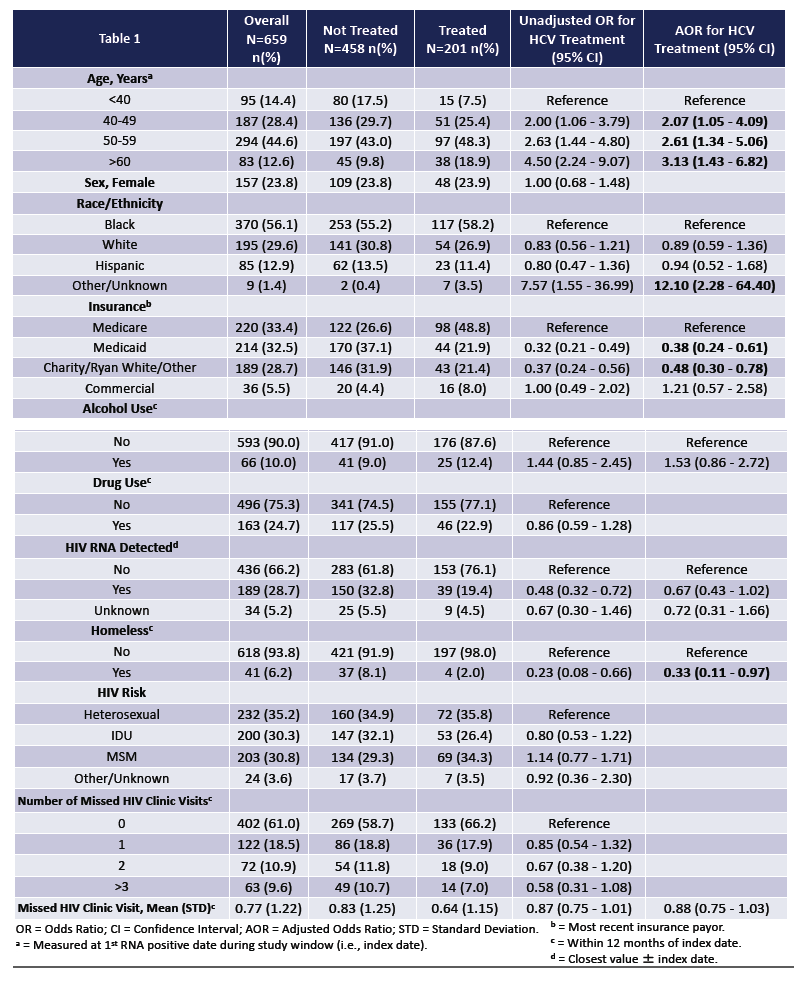
Among all people in care for HIV, 60% had not received HCV antibody testing and 62% had no HCV RNA test in 2014-2017. Among 659 people positive for HCV RNA, 24% were women, 56% black, 30% white, and 13% Hispanic. One third became infected through drug use, one third through gay sex, and one third through heterosexual sex. Only 10% used alcohol, while 25% used other drugs. Forty-one people (6%) were homeless. Only 5.5% of the study group had commercial insurance, one third used Medicare, one third used Medicaid, and 30% had no insurance, relying on Ryan White support or charity.
Of the 659 people positive for HCV RNA, 458 (69.5%) had not received DAA therapy. Multivariate analysis determined that, compared with people younger, those 40 to 49, 50 to 59, or 60 or older had 2 to 3 times higher odds of getting DAA therapy (for 40 to 49, adjusted odds ratio [aOR] 2.07, 95% confidence interval [CI] 1.05 to 4.09). Compared with Medicare recipients, those relying on Medicaid had about 60% lower odds or getting DAAs (aOR 0.38, 95% CI 0.24 to 0.61) and those with no insurance had about 50% lower odds (aOR 0.48, 95% CI 0.30 to 0.78). Homeless people had two thirds lower odds of receiving DAA therapy (aOR 0.33, 95% CI 0.11 to 0.97).
Factors that did not affect chances of receiving DAA therapy in the multivariate analysis included gender, black or Hispanic race/ethnicity, HIV transmission risk, alcohol or drug use, or missed HIV clinic visits.
The researchers concluded that HCV antibody or RNA testing is infrequent in people with HIV infection in this safety-net hospital system. Only 30% of those with confirmed HCV got treated in the DAA era.
References
1. Jain MK, Chavez C, Sanders J, Vysyaraju K. Hepatitis C eradication: who is being left behind in the HIV population? IDWeek 2018, October 3-7, 2018, San Francisco. Abstract 2233. Poster at https://idsa.confex.com/idsa/2018/webprogram/Paper70237.html
2. Yek C, de la Flor C, Marshall J. Effectiveness of direct-acting antiviral therapy for hepatitis C in difficult-to-treat patients in a safety-net health system: a retrospective cohort study. BMC Med. 2017;15:204. doi 10.1186/s12916-017-0969-3.
3. Wong RJ, Jain MK, Therapondos G, et al. Race/ethnicity and insurance status disparities in access to direct acting antivirals for hepatitis C virus treatment. Am J Gastroenterol. 2018;113;1329-1338. doi 10.1038/s41395-018-0033-8
------------------------------------------


|
| |
|
 |
 |
|
|