 |
 |
 |
| |
PHARMACOKINETIC PROFILE OF IBALIZUMAB FROM A PHASE 3 TRIAL
|
| |
| |
a new type of antiretroviral medication for adult patients living with HIV who have tried multiple HIV medications in the past (heavily treatment-experienced) and whose HIV infections cannot be successfully treated with other currently available therapies (multidrug resistant HIV, or MDR HIV).Trogarzo is administered intravenously once every 14 days by a trained medical professional and used in combination with other antiretroviral medications.
48-Week Safety and Efficacy On-Treatment Analysis of Ibalizumab in Patients with Multi-Drug Resistant HIV-1
FDA approves new HIV treatment (Long-Acting Ibalizumab) for patients who have limited treatment options
IBALIZUMAB SUSCEPTIBILITY & Potency vs ART Resistance - New HIV Drug for Multi-Drug Resistant Patients
IBALIZUMAB FDA Product Insert
Reported by Jules Levin
IDWeek 2018, October 3-7, 2018, San Francisco
P. KUMAR1, S. WEINHEIMER2, Z. COHEN3, C. MARSOLAIS3, K-L. KUO4, S. LEWIS2 1Georgetown University, Washington, DC, 2TaiMed Biologics, Irvine, CA, 3Theratechnologies, Montreal, Canada, 4TaiMed Biologics, Taipei City, Taiwan

program abstract
Background: Ibalizumab (IBA) is a long-acting humanized monoclonal antibody that binds domain 2 of the CD4 receptor and blocks HIV-1 infection of host cells. TMB-301 was a 24-week, Phase 3 clinical trial conducted in 40 heavily treatment-experienced patients with multidrug resistant (MDR) HIV-1 investigating the safety, efficacy and tolerability of IBA. Patients received a 2000 mg IBA loading dose followed by 800 mg every 2 weeks by intravenous infusion plus an optimized background regimen. Viral load <50 and <200 HIV RNA copies/mL was achieved in 43% and 50% of patients, respectively at Week 25.
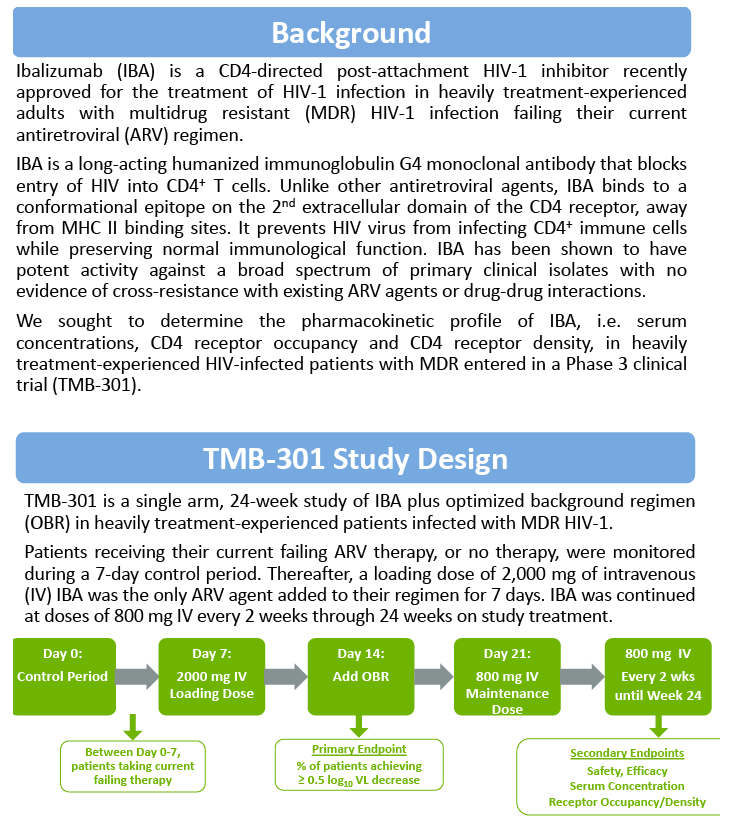
We determined the pharmacokinetic profile of IBA, i.e., serum concentrations, CD4 receptor occupancy (RO), and CD4 receptor density (RD), in these patients with MDR HIV-1.
Methods: Pre- and post-dose blood samples collected at various time points during trial were used to determine IBA serum concentrations, CD4 RO and RD at trough. IBA serum concentrations were measured using a validated ELISA. IBA bound to CD4+ T cells (RO) and cell surface CD4 levels (RD) were measured simultaneously by flow cytometry using the Molecules of Equivalent Soluble Fluorescence approach.
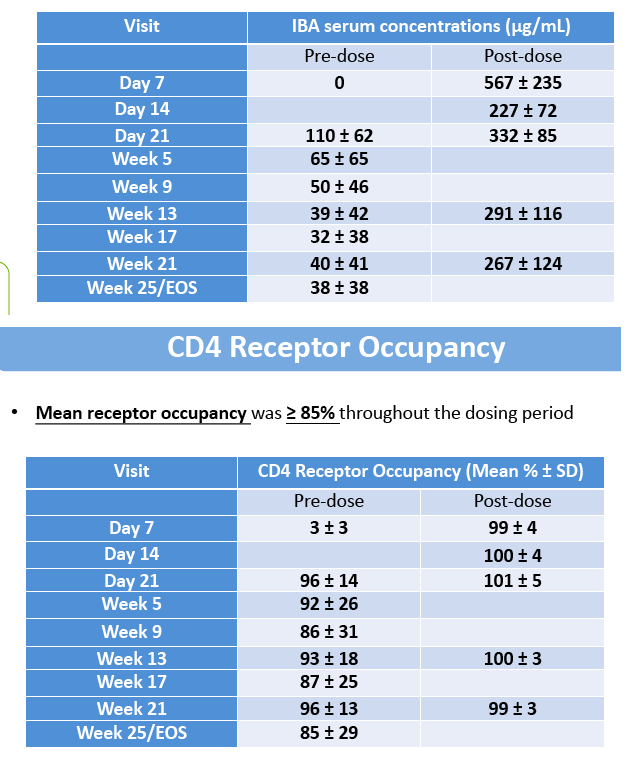
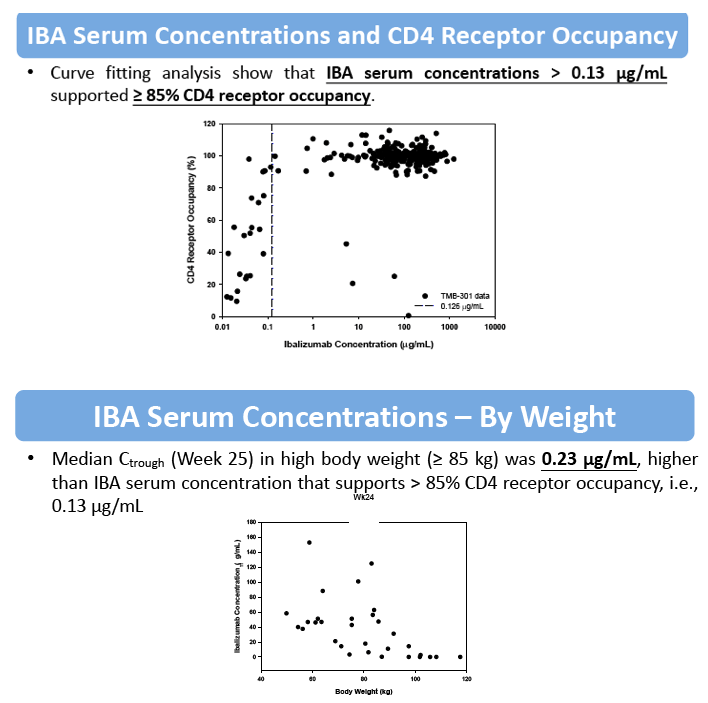
Results: The maximum IBA serum concentrations were observed immediately after the end of the 2000 mg infusion with mean (SD) of 567 (235) μg/mL. Steady state was reached at Week 4 after the loading dose. The mean IBA concentrations were >30 μg/mL throughout the dosing period. Both Cpeak and Ctrough (Day 7 and Week 25 IBA concentrations) were decreased with increased body weight. The median Ctrough in the high body weight group (≥85 kg) was 0.23 μg/mL.
The mean RO was >85% throughout the dosing period. The 2000 mg loading dose helped to reach >85% RO after the initial dose and maintain high levels of RO throughout the dosing period. Elevation in RO was generally associated with increased IBA serum concentrations; concentrations ≥0.13 μg/mL supported ≥85% CD4 RO.
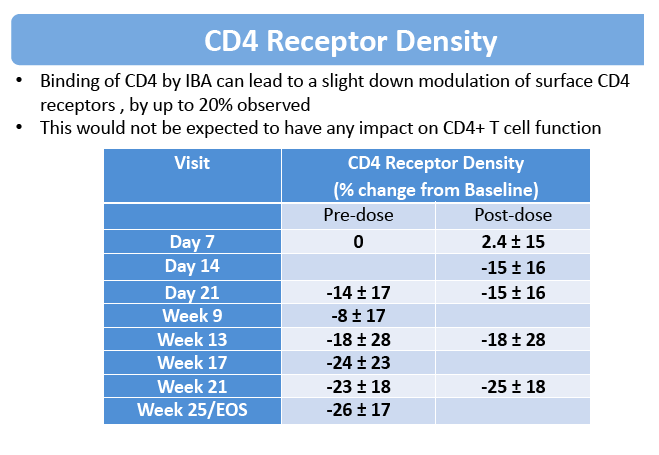
After IBA administration, down-modulation of surface CD4 receptors by up to 20% was observed. There was no apparent association between IBA serum concentration and RD probably due to high inter-individual variation.
Conclusion: Dosing regimen of 2000 mg loading dose followed by 800 mg every 2 weeks was sufficient to support high levels of RO and to maintain the drug concentration above the therapeutic level.

|
| |
|
 |
 |
|
|