 |
 |
 |
| |
Global burden of NAFLD and NASH: trends,
predictions, risk factors and prevention
|
| |
| |
Download the PDF here
Nature Reviews Gastroenterology & Hepatology 20 September 2017 - Zobair Younossi1,2, Quentin M. Anstee3,4, Milena Marietti5, Timothy Hardy3,4, Linda Henry1,2, Mohammed Eslam6, Jacob George6 and Elisabetta Bugianesi5
Abstract
NAFLD is one of the most important causes of liver disease worldwide and will probably emerge as the leading cause of end-stage liver disease in the coming decades, with the disease affecting both adults and children. The epidemiology and demographic characteristics of NAFLD vary worldwide, usually parallel to the prevalence of obesity, but a substantial proportion of patients are lean. The large number of patients with NAFLD with potential for progressive liver disease creates challenges for screening, as the diagnosis of NASH necessitates invasive liver biopsy. Furthermore, individuals with NAFLD have a high frequency of metabolic comorbidities and could place a growing strain on health-care systems from their need for management. While awaiting the development effective therapies, this disease warrants the attention of primary care physicians, specialists and health policy makers.
http://www.natap.org/liver.htm
Key points
⋅ As a consequence of the pandemic spread of obesity, NAFLD is one of the most important causes of liver disease worldwide in adults and children, although some patients are lean
⋅ Global prevalence of NAFLD is estimated at 24%; the highest rates are reported from South America and the Middle East, followed by Asia, the USA and Europe
⋅ The large volume of patients sets NAFLD apart from other liver disease, meaning the major focus of clinical care is discerning those at highest risk of progressive liver disease
⋅ Being overweight in childhood and adolescence is associated with increased risk of NAFLD later in life; consequently, the threshold of liver-related morbidity and/or mortality is reached at a younger age
⋅ Patients with NAFLD have a high risk of liver-related morbidity and mortality along with metabolic comorbidities and might place a growing strain on health-care systems
⋅ While awaiting effective therapies, NAFLD warrants the attention of primary-care physicians, specialists and health policy makers, starting with prevention of excessive weight gain during childhood
Main
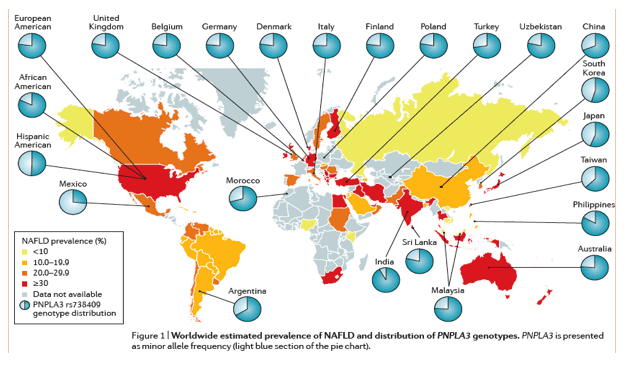
During the past century, dramatic modifications in lifestyle have radically changed the health priorities in most areas of the world, owing to a growing incidence of noncommunicable disease. The new epidemic in chronic liver disease is related to the burden of NAFLD, paralleling the worldwide increase of obesity. The global prevalence of NAFLD is currently estimated to be 24%1. Community surveys utilizing ultrasonography or proton NMR spectroscopy have assessed the prevalence of NAFLD across geographical locales (Fig. 1), whereas studies based on elevated liver enzymes systematically underestimated the true prevalence1. NAFLD is highly prevalent in all continents, but the highest rates are reported from South America (31%) and the Middle East (32%), followed by Asia (27%), the USA (24%) and Europe (23%), whereas NAFLD is less common in Africa (14%)1.
NAFLD, particularly its histological phenotype NASH, can potentially progress to advanced liver disease, cirrhosis and hepatocellular carcinoma (HCC)2. The prevalence of NAFLD is constantly increasing (15% in 2005 to 25% in 2010) and similarly the rate of NASH in the same timeframe has almost doubled (59.1% versus 33%)1. NASH is now considered the second most common indication for liver transplantation in the USA after chronic hepatitis C, and is still growing2. This Review will provide evidence of the global burden of NAFLD and its clinical and economic implications, which should be considered by health policies to secure a better future for coming generations.
NAFLD in the USA and South America
Prevalence of NAFLD in the USA. Over the past two decades, there has been extensive research to accurately determine the prevalence of NAFLD in the USA3,4,5,6,7,8,9,10. These data were summarized in a meta-analysis published in 2016 reporting the worldwide prevalence of NAFLD1. In most of these studies, the prevalence of NAFLD in the general population was determined by imaging or other indirect methods. In this context, the prevalence of NAFLD in the USA diagnosed by ultrasonography was estimated to be 24.13% (95% CI 19.73-29.15%). On the other hand, the prevalence of NAFLD as determined by any other noninvasive methods (such as the Fatty Liver Index, International Classification of Diseases (ICD) 9 or ICD 10 coding) was reported to be ∼21.09% (95% CI 15.0-28.8%), suggesting that diagnosis of NAFLD solely based on blood testing or ICD coding can lead to under-reporting of its true prevalence1.
In the USA, the prevalence of NAFLD can vary by the ethnicity. In this context, the prevalence of NAFLD is reported to be highest in Hispanic Americans, followed by Americans of European descent and then African Americans3,4,5,6,7,8,9,10,11. Although still not fully resolved, a number of factors could explain these reported ethnic disparities in the prevalence of NAFLD in the USA, including genetic factors, environmental factors, access to health care and presence of chronic diseases such as the metabolic syndrome1,6,7,8. The lower prevalence of NAFLD among African Americans than Hispanic Americans5,8 is especially surprising because of the higher prevalence of obesity and hypertension in African American patients5,8.
By contrast, a study using data from the US Third National Health and Nutrition Examination Survey (1988-1994) reported that the metabolic syndrome was the primary driver of NAFLD among non-Hispanic black patients and Mexican Americans, but not for white Americans. Despite some contradictory data regarding the interaction of NAFLD and components of the metabolic syndrome in African Americans, this study suggests that the association of the metabolic syndrome with NAFLD might be influenced by ethnicity12.
In another study using data from the Nonalcoholic Steatohepatitis Clinical Research Network (NASH CRN), investigators compared Latino patients with NASH with non-Latino white patients with NASH13. The study found that Latino patients with NASH were younger (mean 44.2 years of age versus 50.9 years of age), undertook less physical activity, but had a higher carbohydrate intake than non-Latino white patients. Furthermore, they reported that the effect of insulin resistance as defined by the homeostatic model assessment (HOMA) on the risk of NASH was modified by ethnicity. In this context, HOMA was not a risk factor for NASH among Latino patients (odds ratio (OR) 0.93, 95% CI 0.85-1.02), but was an important risk factor among non-Latino white patients with NASH (OR 1.06, 95% CI 1.01-1.11). These data confirm that factors associated with NAFLD can be influenced by the ethnic background of the patient13.
It is also important to recognize that even within certain ethnic groups in the USA there might be differences in the prevalence of NAFLD. In fact, the prevalence of NAFLD among Hispanic Americans can vary according to the country of origin8,14,15. In one study, investigators compared the prevalence of NAFLD in Hispanic individuals of Mexican origin and Hispanic individuals of Dominican and Puerto Rican origins (Caribbean area). Using data from the Multi-Ethnic Study of Atherosclerosis (MESA) cohort, the overall prevalence of NAFLD in Hispanic people was 29%14. However, Hispanic individuals of Mexican origin had a higher prevalence of NAFLD, at 33%, whereas Hispanic individuals of Dominican origin had a prevalence of only 16% and Hispanic individuals of Puerto Rican origin had a prevalence of 18%. After multivariate analysis controlling for age, sex, BMI, waist circumference, hypertension, level of education, HOMA and serum HDL, triglyceride and C-reactive protein levels, Hispanic individuals of Mexican origin continued to remain at higher risk of having NAFLD than individuals of Dominican and Puerto Rican origin14.
Although the ethnic and country of origin data regarding the prevalence of NAFLD is interesting, the exact explanation for these ethnic differences remains unknown. Some of these differences can be explained by the genetic factors that are described later in this Review (nature), whereas others can be explained by environmental factors (nurture), such as diet, exercise and alcohol consumption. Finally, the prevalence of NASH in the general population remains unknown. Nevertheless, there are indirect estimates for these rates, derived by calculating prevalence of NASH in NAFLD and prevalence of NAFLD in the general population. In this context, the prevalence of NASH among patients with NAFLD in the USA is reported to be 21% (95% CI 19.85-22.95%)1. Using this rate, prevalence of NASH in the US population is estimated to be around 3-4%1. The corresponding prevalence of comorbid conditions associated with NASH in these individuals has been reported to be: obesity in 82%; type 2 diabetes mellitus (T2DM) in 48%; hyperlipidaemia in 82%; the metabolic syndrome in 76%; and hypertension in 70%1.
Prevalence of NAFLD in South America. The prevalence of NAFLD in South America seems to be higher than the rate reported for the USA. Specifically, NAFLD prevalence (assessed by ultrasonography) for South America has been estimated to be ∼30.45% (95% CI 22.74-39.4%)1. The majority of studies reporting the prevalence of NAFLD from South America have been performed in Brazil15,16,17. Nevertheless, in a study reported from Chile, the prevalence of NAFLD (diagnosed using ultrasonography) was estimated to be 23%18. Another study from Columbia, also using ultrasonography, reported a prevalence of 26.6% in men19.
Furthermore, the same investigators have estimated that the prevalence of 'probable NAFLD', inferred from the rates of obesity, in Peru, Argentina, Ecuador, Paraguay, and Uruguay, could be as low as 13% (Peru) to as high as 24% (Uruguay)19. Although there are estimates for the prevalence of NAFLD in South America, the data on the prevalence of NASH is even more scarce. Nevertheless, in one study, 61% of the morbidly obese patients with NAFLD in South America were found to have NASH, which could make the prevalence of NASH 6-18%20. Again, these rates can be influenced by genetic predisposition, as described later.
In summary, NAFLD prevalence rates differ by ethnicity within the USA1,18,19,20,21. The Hispanic population has the highest prevalence whereas African Americans are reported to have the lowest prevalence, despite having higher prevalence of hypertension and obesity, both NAFLD risk factors. Ethnic differences are also noted within South America, with Brazil reporting the highest NAFLD prevalence and Peru the lowest.
Incidence of NAFLD and future projections in the USA and South America. Longitudinal studies of the general population are lacking in both the USA and South America. As a result, there are no true population-based incidence data reported for NAFLD. However, Kanwal and colleagues10have suggested that the annual incidence of NAFLD based on liver enzyme levels in veterans from the USA who were younger than 45 years old increased from 2.32% to 4.26% (P <0.001). Furthermore, NAFLD is known to be highly associated with several metabolic conditions (T2DM, obesity, the metabolic syndrome, hypertension and hyperlipidaemia)22,23. Thus, it is expected that the incidence of NAFLD should rise in parallel to the increasing incidence of obesity and T2DM24. In addition to obtaining data about the true incidence of NAFLD, it is also important to determine the long-term outcomes of patients with NAFLD. In this context, developing algorithms to define which patients with NAFLD will develop the progressive form of NASH, cirrhosis, liver-related mortality or cardiovascular mortality will be of great importance.
NAFLD in Europe
Epidemiology of NAFLD in Europe. In Europe, a gradient of increasing prevalence from north to south has been described for most forms of chronic viral or non-viral hepatitis, but this observation does not hold true for NAFLD. Rather, the globalization of NAFLD runs parallel to the prevalence of obesity and varies accordingly, with degree of hepatic triglyceride accumulation being directly proportional to the severity of each element of the metabolic syndrome25. Although prevalence varies according to the modality used to detect NAFLD, approximately one-quarter of the European population is affected by this liver disease. A meta-analysis published in 2016 reported an average prevalence of 23.71% in Europe, varying from 5% to 44% in different countries1.
Data from the Study of Health in Pomerania (SHIP) cohort in northeastern Germany estimates the prevalence of NAFLD to be ∼30% when diagnosed by ultrasonography26. A UK-based community study determined that NAFLD was the most common aetiology for asymptomatic abnormal liver biochemistry, accounting for 26.4% of cases (of whom 7.6% were predicted to have advanced liver disease)27. Similarly, a series of liver biopsies in individuals with unexplained abnormal liver tests conducted in France reported simple steatosis in 26.8% of patients, of whom 32.7% had NASH28. In the Dionysos Study, the prevalence of NAFLD assessed by ultrasonography in northern Italy was similar in those with and without suspected liver disease (25% versus 20%, P = 0.203), defined as altered circulating liver enzyme levels or hepatitis B surface antigen (HBsAg) and/or anti-HCV positivity29.
Notably, only 54% of NAFLD cases occurred in patients with elevated serum alanine aminotransferase (ALT) levels, but the vast majority of these individuals had many features of the metabolic syndrome. Epidemiological data from Spain describe similar rates, with a NAFLD prevalence of 25.8% in the adult population30. Only a few studies are available from eastern Europe. In Romania, NAFLD (assessed by ultrasonography) was found in 20% of 3,005 hospitalized patients without liver disease31, whereas a study on healthy Hungarian adults confirmed a 22.6% overall prevalence of ultrasonographically detected fatty liver32.
As expected, the prevalence of NAFLD increases substantially in 'at risk' groups such as patients with T2DM2. The two major available studies, conducted on a large cohort of Italian patients with T2DM, reported NAFLD prevalence of 60-70%33,34; data from the UK suggests that NAFLD, detected ultrasonographically, is present in 42.6% of patients with T2DM35. Prevalence of NAFLD also increases with BMI, such that in an unselected Italian population sample 91% of patients who were obese (BMI �30 kg/m2), 67% of those who were overweight (BMI 25-30 kg/m2) and 25% of individuals at a normal weight (BMI 18-25 kg/m2) had ultrasonographical evidence of NAFLD36. The prevalence of NAFLD among patients matching at least one of the Adult Treatment Panel III (ATP III) criteria for serum lipid level alterations is similarly high at 78.8%33.
Data regarding the prevalence of advanced forms of NAFLD and NASH in the general European population are more limited. A community-based study from the Netherlands using a transient elastography reading of �8 kPa for the diagnosis of liver fibrosis estimated that clinically significant fibrosis was present in 5.6% (169 of 3,041) of total, nonselected individuals and 8.4% (69 of 822) of those with NAFLD, and was positively associated with steatosis and T2DM37. In this respect, the influence of T2DM on risk of progressive NAFLD is supported by a UK-based paired-biopsy study that showed incident T2DM to be the strongest predictor of progressive disease38. A postmortem study conducted in Greece on 498 cases of ischaemic heart disease or traffic accident deaths revealed simple steatosis in 31.3% and NASH in 39.8% of individuals39. In a study of Spanish patients with gallstone disease scheduled for cholecystectomy, 51.6% of them had histological evidence of NAFLD and 19.8% of NASH40. Of note, in this cohort, ultrasonography confirmed a fatty liver in only 67.6% of the histologically diagnosed patients with NAFLD. Similar rates of NAFLD are reported in healthy people evaluated in transplantation units as potential living liver donors. In a single retrospective study performed in a mixed US and Italian cohort in this setting, the histological prevalence of steatosis and steatohepatitis was 48.5% and 15.5%, resepctively41. However, both NAFLD and NASH were more frequently found in Americans compared with Italians (54% versus 34% for NAFLD and 17.6% versus 16.2% for NASH, respectively). The rates of NASH are clearly increased in patients referred to tertiary centres for NAFLD. In a meta-analysis published in 2016, the pooled NASH prevalence in Europe among patients with NAFLD with an indication for biopsy was 69.25% (95% CI 55.93-79.98%)1.
Incidence of NAFLD and future projections in Europe. Only a small number of studies explored the incidence of NAFLD in the general population. Over a follow-up of 8.5 years, the incidence of ultrasonographically detected NAFLD was 18.5 per 1,000 person-years in a sample representative of the general Italian population36. Data gathered in the past few years are clarifying the natural history of histologically diagnosed NAFLD. In a Swedish cohort of 229 patients with biopsy-proven NAFLD with a mean longitudinal follow-up of 26.4 ± 5.6 years, patients with NAFLD had increased all-cause mortality (hazard ratio (HR) 1.29, 95% CI 1.04-1.59), exhibited an increased risk of cardiovascular disease (HR 1.55, 95% CI 1.11-2.15), HCC (HR 6.55, 95% CI 2.14-20.03) and cirrhosis (HR 3.2, 95% CI 1.05-9.81), compared with a matched reference population sample42. The presence of fibrosis was found to be the strongest prognostic factor for liver-related events and mortality42. Consistent with this finding, the burden of NAFLD-related HCC is also increasing dramatically. A study conducted in northeast England found that NAFLD-associated HCC accounted for 35% of all HCC cases (41 of 118) in 2010, representing a greater than tenfold increase in 10 years43. Finally, results from the UK National Health Service Blood and Transplant Agency show that decompensated NASH cirrhosis accounted for an increased proportion of patients undergoing liver transplantation (12% in 2013 compared with 4% in 1995)44.
NAFLD in the Asia-Pacific and Africa
NAFLD prevalence varies widely within the Asia-Pacific region, as would be expected from a region comprising at least 55 countries with marked disparities in levels of economic, political and educational development, and which are associated with variations in nutrition, lifestyle and sedentary behaviour. Furthermore, there is a bias towards reporting studies that emanate from more affluent economics in the region with better health-care systems. Unlike data from Europe and North America, data from the Asia-Pacific region and Africa are not as comprehensive both between and within countries, and there is a total absence of this information from many countries.
Although there are no nationwide epidemiological surveys that include an assessment of liver fat, there can be striking differences in the prevalence of NAFLD between different subnational regions and over time. As an example, NAFLD prevalence in the populations of Chengdu (southwest China), Shanghai (east China), Guangdong (south China) and central China was 12.5%, 15.0%, 17.0% and 24.5%, respectively45,46,47. On the other hand, an ultrasonography study of 7,152 workers in Shanghai published in 2012 suggested that NAFLD prevalence was as high as 38.17%48. In Hong Kong, a community-based study employing state-of-the-art proton-magnetic resonance spectroscopy to quantify liver fat estimated a NAFLD prevalence of 28.8%: 19.3% in individuals who were not obese and 60.5% among those who were obese49. Similarly, in Taiwan, the prevalence of NAFLD was reported to be 11.4% in the general community50 but higher in certain subpopulations, including the elderly (mean age 70.3 ± 4.6 years, 50.1% prevalence)51 and in those with a typically inactive lifestyle (66.4% prevalence in taxi drivers)52.
The community prevalence of NAFLD was ∼25% in Japan in 2005, increasing from 12.6% before 1990 to 30.3% in 1998 (Ref. 53). A report published in 2005 suggests that 23-26% of individuals undergoing routine health screening have fatty liver diagnosed by abdominal ultrasonography54. Using similar methodology, the prevalence of NAFLD in South Korea in 141,610 individuals was 27.3% when reported in 2013 (Ref. 55).
South Asia and the Indian subcontinent are currently in the throes of rapid economic and social change, with trends towards urbanization and an urban-rural economic divide. Not unexpectedly, in rural India, a region characterized by traditional diets and lifestyles, the prevalence of NAFLD is low (∼9%), whereas it mimics Western prevalence trends in urban populations, with rates varying between 16% and 32%56,57,58. A similar dramatic variation in NAFLD prevalence between urban and rural regions and along racial, economic and cultural boundaries (5-30%) was observed from smaller surveys in Sri Lanka, Malaysia, Singapore and Indonesia59,60,61,62. Overall, NAFLD prevalence varies but is increasing across Asia. Given that this region is subject to the same global forces of change towards increased energy-dense food consumption and reduced physical activity, NAFLD rates in the East and West are more similar than different, in the context of a similar obesogenic environment.
The scant available data on the prevalence of NAFLD in Africa suggests that Africans tend to have lower prevalence of the condition, consistent with what has been reported in African Americans. In Nigeria, NAFLD prevalence of 9.5-16.7% in individuals with diabetes and 1.2-4.5% in individuals without diabetes has been reported63,64. Similarly, in South Africa, the prevalence in individuals who were obese or overweight was 45-50%65. A small population-based study published in 2014 suggested a prevalence of 20% in the Sudanese population66.
Lean NAFLD
Initially described in Asian populations, NAFLD in the absence of obesity, so-called 'lean NAFLD', can develop in ∼10-20% of Americans and Europeans5,67 (Fig. 2; see Supplementary information S1, S2 (tables))45,49,50,56,67,68,69,70,71,72,73,74,75,76,77,78. This manifestation of NAFLD deserves clinical attention as many physicians have a perception that lean NAFLD is more benign in nature than NAFLD in individuals who are obese. Lean NAFLD encompasses a heterogeneous spectrum of disease arising from different aetiologies (Box 1). Increased visceral obesity (as opposed to general obesity), high fructose and fat intake and genetic risk factors, including congenital defects of metabolism, might be associated with lean NAFLD. A very large proportion of lean NAFLD cases probably belong to the 'metabolically obese, normal weight' phenotype79, described in at least 5% of the population in the West. This subgroup comprises individuals who are non-obese, frequently sedentary, and who have impaired insulin sensitivity, increased cardiovascular risk and increased liver lipid levels, the consequence of decreased capacity for storing fat and reduced mitochondrial function in adipose tissue and increased hepatic de novo lipogenesis79. When compared with patients who have NAFLD and are overweight or obese, patients with NAFLD who are lean are younger and have a lower prevalence of the metabolic syndrome (2-48% versus 22-64% in patients who are overweight or obese)67,80. However, these patients are usually insulin-resistant and have higher plasma triglyceride levels when compared with matched controls without NAFLD74,80. In a cohort of patients with biopsy-proven NAFLD who were not obese and not diabetic, the metabolic milieu was similar to that observed in patients with NAFLD who are obese, with adipose tissue insulin resistance having an important role81.
As lean NAFLD is usually present with fewer obesity-related comorbidities, it is commonly believed that this subgroup would follow a relatively benign clinical course. Within the cohort of the National Health and Nutrition Examination Survey III79, mortality of patients with metabolically normal NAFLD was similar to the cohort without liver disease. Unfortunately, most reports are limited by the use of imaging modalities rather than liver biopsy to confirm the diagnosis of fatty liver5,74,79,80,81,82,83,84. In a study conducted in Italy by Fracanzani et al.82 that included 430 cases of biopsy-proven NAFLD, 55% of patients without visceral obesity had NASH and a fibrosis stage �F2 despite milder metabolic alterations than patients who were obese with biopsy-proven NAFLD. In a study published in 2017 (Ref. 83), similar proportions of patients who were obese and non-obese had NASH (51.9% versus 43.5%, respectively, P = 0.217), although the latter group had a lower degree of hepatic steatosis and hepatocyte ballooning. Consistent with a report from a decade earlier79, the proportion of patients with advanced fibrosis at baseline was not different between those who were obese and non-obese, suggesting that once an individual has been diagnosed with NASH, obesity might not be the main driver of fibrosis progression. Genetic factors might be involved in risk of lean NAFLD, however, the presence of NASH in these patients was not explained by mutations in genes that can influence either insulin resistance (ENPP1 and IRS1 polymorphisms) or the severity of steatosis (PNPLA3 and TM6SF2 polymorphisms)82.
The longitudinal risk of mortality in lean NAFLD has not been explored in any detail. In the study by Fracanzani et al.82 discussed earlier, clinical events occurred in 11.9% of patients who were obese and 8.3% of patients who were not obese (P = 0.190) after a median follow-up of 49 months. Cardiovascular events accounted for about two-thirds of all major events in both groups. All deaths (n = 6) occurred in the obese group, but definitive conclusions are difficult to make as follow-up was short. An international cohort study including 483 patients with biopsy-diagnosed NAFLD with a mean follow-up period of over 11 years85, published in abstract form, challenged the concept that the prognosis of patients with NAFLD who have a normal BMI is benign. Despite presenting with a healthier metabolic profile and less advanced liver fibrosis, median survival free of liver transplantation was shorter in patients who were lean than in patients who were not lean (18.1 versus 26.6 years, respectively, P <0.001).
The final question is how to manage patients who are lean and diagnosed with NAFLD, given that it might be harder to correct the underlying risk factors in these individuals. Careful identification and correction of environmental causes, such as substantial fructose consumption, might be particularly effective in children and adolescent patients, in whom the intake of sugar-sweetened drinks can be high. Weight loss remains the background therapy in all patients with NAFLD and overweight or obesity, but habitual physical activity should also be emphasized in patients with NAFLD who are lean. More studies are urgently needed to understand the natural history of the disease, but also to promote greater awareness among practitioners about the potential health risks associated with lean NAFLD.
The future effect of paediatric NAFLD
The obesity rate in children has risen from 5.0% in 1960 to 16.9% in 2009-2010 (Ref. 86). The obesity-related risk of future liver disease is alarming, as weight gain during school-years carries a higher risk of NAFLD than weight-gain in late adulthood. In a longitudinal study conducted in Denmark87, a weight increase during childhood and early adolescence was related to all histological features of adult NAFLD, even after adjusting for initial as well as attained BMI. Among children with similar attained BMIs at 13 years of age, the risk of cirrhosis in adulthood was increased by 16% per 1-unit gain in BMI z-score at every age from 7 through 13 years87. Similarly, weight gain during late adolescence is able to induce an increased susceptibility to developing NAFLD later in life. Another study tested the association of basal BMI on the development of end-stage liver disease or liver-related death in a general population cohort of 44,248 men aged 18-20 years that attended military service in Sweden between 1969 and 1970 (Ref. 88). After a follow-up of almost 38 years, being overweight in late adolescence increased the risk of liver-related outcomes by 64% compared with those with a low to normal BMI, with risk increased 5% for each unit of BMI above the normal range (BMI 18-25 kg/m2)88. Obesity early in life also increases the risk of HCC in adulthood. Another study conducted in Denmark that included schoolchildren aged from 7 to 13 years old89 showed that each unit increase in BMI z-score increased the risk of liver cancer by 20-30% 30 years later. In other words, compared with an average height and weight 13-year-old boy, a boy of similar height but who weighed 6 kg more would have a 30% increased risk of HCC89. Besides body weight trajectory, other mechanisms seem to influence the spectrum of liver damage in NAFLD later in life. In the Cardiovascular Risk in Young Finns Study90, after a follow-up of 31 years, adult NAFLD was predicted by modifiable as well as non-modifiable risk factors during childhood, including BMI and plasma insulin levels, male sex, genetic background (that is, PNPLA3 and TM6SF2 variants) and low birth weight, an emerging risk factor for adulthood NAFLD. Overall, these observations mean that NAFLD and its complications, including HCC, are more likely to be anticipated at younger age, yielding a possible reduction of life expectancy and an additional societal burden.
Risk factors: nature or nurture?
Evidence from patients that have undergone serial liver biopsies over an interval of several years demonstrates that the progression of NAFLD from steatosis to NASH and fibrosis is not linear and is probably more dynamic than previously thought; the fibrosis progression rate in simple steatosis is estimated to be 14 years per stage of fibrosis, and the fibrosis progression rate in NASH is estimated at 7 years per stage of fibrosis. Data published in the past few years suggest that risk of liver-related mortality in NAFLD grows exponentially as the stage of fibrosis increases38,91,92. Furthermore, evidence from familial aggregation and twin studies have shown a heritable component to NAFLD93,94. Interestingly, the genetic susceptibility for the development of steatosis and fibrosis might be shared95. Different ethnic groups have disparate propensities to advanced disease, with Hispanic individuals being more susceptible than white patients, whereas the lowest susceptibility is observed in black individuals96. An interesting systematic review suggested that the leading explanations for the lowest incidence and prevalence of both NAFLD and NASH in African Americans in the USA is related to genetic differences in lipid metabolism; that is, this group have lower serum triglyceride levels and higher serum HDL cholesterol levels than patients with NAFLD who are Hispanic or white97.
Genetic associations with NAFLD. In NAFLD, genome-wide association studies have identified novel loci associated with disease severity phenotypes. A full discussion is beyond the scope of this article, but the available literature has recently been reviewed elsewhere98. To date, non- synonymous SNPs in two genes in particular, PNPLA3 (encoding patatin-like phospholipase domain-containing protein 3) and TM6SF2 (encoding transmembrane 6 superfamily member 2), have most consistently been validated as associated with NAFLD in separate large cohorts99,100. Fig. 1 shows the geographical distribution of PNPLA3 genotypes in patients with NAFLD. PNPLA3 is presented as minor allele frequency (that is, the frequency at which the second most common PNPLA3 allele occurs in a population). Among the emerging newly discovered risk loci, variants near the genes encoding for membrane-bound O-acyltransferase domain-containing 7 (MBOAT7) and transmembrane channel-like 4 (TMC4) have been shown to be associated with development and severity of NAFLD in patients of European descent101. Similarly, within the Latino population in South America, the TM6SF2 Glu167Lys and PNPLA3 Ile148Met protein variants seem to confer susceptibility to progressive NASH102.
Epigenetic factors and NAFLD. Although there have been major advances uncovering the genetic basis for the heritability of NAFLD, heritable mechanisms other than those encoded within the nucleotide sequence of genes are emerging. Discordant NAFLD in genetically identical twins has been shown to be explained by microRNAs103. Epigenetic factors might also be a mechanism through which environmental exposures exert a heritable effect on disease risk. Remodelling of DNA methylation at key fibrosis modifier genes underpinned protection against carbon tetrachloride (CCl4)-induced liver fibrosis in male offspring of male mice that were themselves subjected to CCl4-mediated liver injury104. Remarkably, DNA methylation remodelling occurred in the same genes in patients with NASH with mild fibrosis and intriguing data suggests that epigenetic signatures present on circulating cell-free DNA could be a potential biomarker of this effect and, therefore, disease severity104,105.
Role of environmental factors. Genetic predisposition must be placed in the context of environmental factors that also have an important role. The most relevant factors are dietary habits, activity and socioeconomic factors. Although large amounts of data suggest that dietary composition might predispose individuals to NAFLD, evidence at the population level is less well characterized. In this context, a study published in 2014 reported that patients with NAFLD tended to reside in areas with many food source options including grocery stores, restaurants and fast food places. Furthermore, those with NAFLD were more likely to report having the unhealthiest eating habits (eating processed foods and/or foods with a high content of fat, salt and sugar or corn syrup) and reported eating more frequently at restaurants106. Other studies focused on the nutritional assessments of patients with NAFLD have further documented increased consumption of low-nutrient, high-sodium and high-fat foods, especially diets high in meat-derived fats and lower amounts of fresh fruits107,108. In addition to these dietary habits, individuals with fatty liver were found to have very low physical activity levels and increased sitting times compared with healthy individuals109,110,111.
The prevalence of NAFLD is also related to socioeconomic factors, but their exact role is debated. In a study exploring the role of environmental factors in different ethnic groups with NAFLD, acculturation, education level, health-care use and income, along with dietary and lifestyle factors and sleep, were not found to be independently associated with risk of developing NAFLD, suggesting that environmental factors might have a role on a background of genetic predisposition16.
Alcohol consumption in the context of NAFLD should be carefully considered. Data from the SHIP study demonstrates that the presence of obesity and alcohol consumption are not mutually exclusive. In patients with radiologically diagnosed hepatic steatosis, 27.3% of men and 9.7% of women fulfilled criteria for both obesity and high alcohol consumption (that is, dual aetiology fatty liver disease)112. Prospective data from the UK in 9,559 men with up to 42-years of follow up unequivocally show that alcohol consumption and the presence of obesity act synergistically to increase the risk of liver disease morbidity and mortality113. Furthermore, the risk of incident HCC is multiplied (HR 7.19, P <0.01) in individuals who consume alcohol and are obese relative to non-obese, non-drinkers114. To fully address the effect of even moderate alcohol consumption on NAFLD will require prospective, longitudinal studies recording cumulative lifetime alcohol consumption.
Overall, these data confirm the concept that NAFLD is a complex disease that is affected by inter-related environmental factors and genetic predisposition. The exact contribution of each of the genetic or environmental components in the promoting the burden of NAFLD is not known and could vary in different regions of the world. Therefore, future studies need to focus on this knowledge gap to better determine treatment and improve patient outcomes.
Global perspectives
Challenges for health care. With an estimate of 64 million individuals affected in the USA and 52 million in European countries1,115, what clearly sets NAFLD apart from other common liver diseases is the sheer volume of patients. In this context, the major focus of clinical care is discerning patients with NAFLD at highest risk for liver-related complications. Joint guidelines on the management of NAFLD released by the European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD) and the European Association for the Study of Obesity (EASO) recommend that individuals with obesity or any component of the metabolic syndrome should undergo ultrasonography and have steatosis biomarkers and serum liver enzyme levels measured116. Furthermore, a burning issue is the development of HCC in NASH even in the absence of cirrhosis, for which no indication is currently provided by guidelines116.
Economic burden of NAFLD. As the clinical consequences of NAFLD grow, the economic consequences will also increase. A model on the population of US and of four European countries (Germany, France, Italy and the UK) published in 2016 estimated the annual burden associated with all incident and prevalent NAFLD cases at US$103 billion in the USA ($1,613 per patient) and at $35 billion in the four Europe countries (from $354 to $1,163 per patient)115. In a study of Medicare patients with NAFLD, the mean yearly inflation-adjusted charges from the outpatient setting increased from $2,624 ± 3,308 in 2005 to $3,608 ± 5,132 in 2010 (Ref. 117). If the annual rate of increase in the costs related to NAFLD is assumed to parallel the annual growth in the prevalence of obesity, the expected 10-year burden of NAFLD could increase to an estimated $1.005 trillion in the USA and $334 billion in the Europe115.
The main question is whether the enormous cost of screening and management would be justified, provided it will be affordable. Cost-utility analysis of NASH screening is hampered by the lack of evidence for benefit during the early stages of disease progression, uncertainties around the accuracy of noninvasive markers of liver damage and the lack of effectiveness data relating to treatment in patients with NASH. Steatosis testing has not been recommended by the UK National Institute for Health and Care Excellence (NICE) NAFLD Guideline Committee owing to the uncertainty both in the cost-effectiveness results for all tests and in the clinical evidence base118. On the other hand, the EASL-EASO-EASD 2016 guidelines and the UK NAFLD Guideline Committee recommend biomarkers (NAFLD fibrosis score, FIB-4) and transient elastography or acoustic radiation force impulse imaging to screen patients with NAFLD for advanced fibrosis and cirrhosis116,118. Ultimately, screening for NASH will probably be cost-effective when medications with reasonable efficacy and adverse effects are available.
Conclusions
NAFLD is now the leading cause of chronic liver disease in the USA and Europe and is increasing worldwide, but there is a paucity of prospective population-based cohort studies from other geographical areas, including South America, Asia, Australia and Africa, which are needed to better understand the global burden of disease (Box 2). Understanding the genetic and environmental risk factors of NAFLD and NASH and their distribution across different countries is of paramount importance to develop strategies to implement a multipronged public health policy and deal with this important chronic liver disease (Box 2).
|
| |
|
 |
 |
|
|