 |
 |
 |
| |
Summary from CROI 2019 for viral Hepatitis
Curing viral hepatitis: what are the clinical implications and what are the remaining barriers to overcome?
|
| |
| |
Jürgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Correspondence:
Prof. Dr. J.K. Rockstroh
Department of Medicine I
University of Bonn
Sigmund-Freud-Str. 25
53105 Bonn
Germany
Introduction
At this year CROI, well in line with the obvious excitement around the two new potential patient cases of HIV cure, the program committee conducted an HBV session which was titled "HBV: racing HIV for the cure, which in retrospect could not have been chosen more accurately. Beyond, the overview on current HBV cure research presented by Fabien Zoulim in the HIV associated tumor viruses session, this themed poster discussion included work on HBV seroconversion rates in HIV/HBV coinfected individuals as well as an analysis of possible prediction factors for HBs-Ag loss (1-3). Understanding why HBs-seroconversion occurs much more frequently in HIV-coinfected subjects under tenofovir containing antiretroviral therapy than in HBV-monoinfected patients receiving tenofovir therapy, as well as evaluating which factors may play a relevant role here, could be instrumental for further HBV cure research. Other relevant topics were risk of HBV reactivation under/or following DAA-based HCV therapy (4), and factors associated with lack of HBV replication control under suppressive tenofovir therapy in HIV/HBV coinfected subjects starting dual active anti-HIV/HBV therapy (5).
In the hepatitis C space new data was presented on a possible recent reduction in new incident acute HCV infections among HIV-seropositive men who have sex with men (MSM), following the increased roll-out of DAA-based treatment strategies in London (6). Of note, HCV-reinfections however remain a concern for jeopardizing HCV elimination in HIV/HCV coinfected MSM. Alarming data on HCV-reinfections was also presented from New York reemphasizing the need for special interventions in this high-risk patient population but also wider access to HCV treatment during acute infection (7). Other presentations focused on the clinical outcome of successful DAA therapy in particular on non-hepatic comorbidities such as cardiovascular events and diabetes (8-10). This is of great interest as it supports the notion that every chronic HCV infection should be treated regardless of fibrosis stage in order to positively impact the outcome of concomitant comorbidities as well as prevent new incident cases for example of diabetes mellitus. Finally, for the first time data on DAA therapy in pregnant women with chronic hepatitis C was presented (11). Under consideration of the alarming increase in HCV-infection rates in pregnant women because of the recent opioid crisis in the US, as well as a perinatal HCV transmission risk of 4-7% in women with viremia, new treatment strategies are urgently needed for this growing patient group. Obviously, with much safer HCV treatment options with short treatment durations, HCV eradication during pregnancy emerges as an attractive option to prevent perinatal HCV transmission. In summary, although no new drugs for HBV or HCV therapy were presented a whole range of important clinical studies were discussed which all have important clinical implications for daily practice.
HBsAg seroconversion in HIV/HBV coinfection
Loss of hepatitis B surface antigen (HBsAg) or HBV functional cure only occurs in 1% of HBV-monoinfected patients per year on TDF (12). HBsAg loss however, remains clinically desirable, as it is associated with a reduction in the risk of developing hepatocellular carcinoma (13). In contrast to HBV monoinfected subjects, successful long-term tenofovir-containing combination antiretroviral therapy leads to HBsAg loss in 5-15% of HBV HIV coinfected patients. However, data on determinants of HBsAg loss in this setting is still missing. Boesecke and colleagues evaluated factors associated with HBsAg loss in a non-interventional retrospective cohort of 7 German HIV care centers assessing rates of HBV seroconversion defined as HBsAg loss in 359 HBV/HIV coinfected patients under HBV active (TDF or TAF containing) combination antiretroviral therapy (cART) (1). The baseline demographics of this cohort are depicted in table 1.
Table 1: Baseline characteristics and region of origin
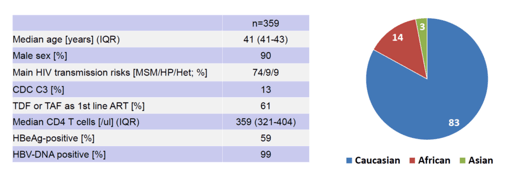
Overall, 18.4% of patients developed an HBsAg loss in the median follow-up time of 11 years (see figure 1).
Figure 1: HBsAg loss (absolute)
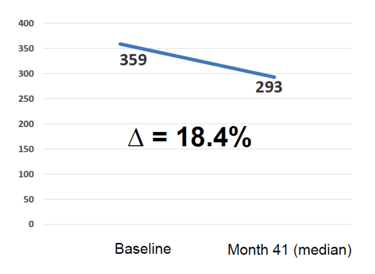
Patients with stage CDC C (p≤0.001), lower CD4 gain (p=0.043) and not receiving TDF/FTC (p=0.008) were less likely to lose HBsAg. While long-term TDF-containing cART leads to higher rates of HBsAg loss when compared to published data for HBV monoinfected subjects, late presentation for HIV and poor immune recovery significantly impair HBV seroconversion rates in HBV/HIV coinfected patients.
Higher seroconversion rates were also reported from an observational HIV/HBV cohort from Zambia (2). This prospective cohort was established in 2013 at 2 primary care clinics in Lusaka, Zambia and enrolled HIV/HBV coinfected adults ≥18 years at ART initiation (fixed-dose combination including TDF). Overall, 284 dually infected subjects were enrolled with a median age, of 34 years (IQR:27-45). 32% of the participants were female. Median CD4 count was 202/μl (IQR: 94-339). Median HBV DNA was 3.29 log10IU/ml (IQR:1-6.42). 41.6% were HBeAg-positive. 10% of study participants developed an HBsAg loss after 2 years on TDF-based therapy. Figure 2 shows the factors associated with HBsAg-loss.
Figure2: Factors associated with HBsAg-loss.
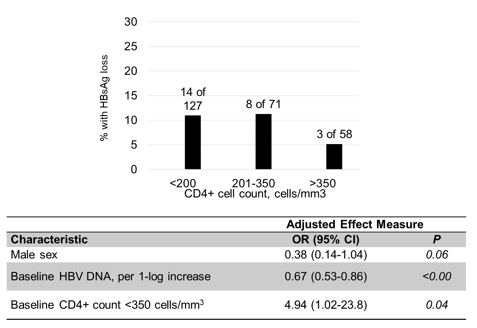
*Factors associated at p<0.2 in univariable analysis were included in the multivariable logistic model
Besides level of HBV-viremia, CD4-count at baseline was the strongest predictor of HBsAg-loss. This African cohort suggests that ART-induced immune reconstitution may enhance anti-HBV immune responses in the liver.
The third cohort looking at HBsAg-loss rates comes from Texas (3). This retrospective analysis of HIV/HBV patients who were seen in three centers in Texas from 2010-2019 included 501 individuals. 98% of the cohort had started combined antiviral therapy (cART) and 96% of the cohort had evidence of HBV specific cART. 59% were black with 51% being MSM. Overall, 10.6% of patients achieved HBsAg-loss during follow-up. Figure 3 shows the relevant factors influencing HBsAg loss in this particular cohort.
Figure 3: Relevant factors for HBsAg-loss.
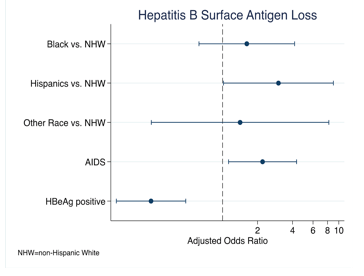
Overall, hepatitis B surface Antigen loss was more likely in Hispanics and in patients with AIDS diagnosis at baseline, as well as those who were HBeAg negative. Again, levels of HBV-viremia (lower in HBeAg patients) and CD4-counts (lower in AIDS-patients) appear to have an impact. Indeed, in patients with HIV who acquire HBV infection, six time higher chronification rates have been reported for HIV-patients. Apparently, ART-induced immune reconstitution may enable patients who potentially would have cleared HBV in the first place if there had not been HIV-associated immunodeficiency, to achieve HBV seroconversion.
Hepatitis B Virologic Failure to Tenofovir-Based Therapies in Patients with HIV/HBV
Combination antiretroviral therapy (cART) regimens containing tenofovir (TDF or TAF) are preferred for treatment of HIV/HBV co-infection. Treatment goal is to achieve undetectable HBV DNA. There is however, a small subset of HIV/HBV co-infected patients which exhibits persistent HBV viremia or virologic breakthrough despite HIV viral suppression while on tenofovir therapy. Prior studies of tenofovir adherence among patients with concurrent HBV viremia and HIV suppression have not been able to evaluate long-term drug exposure due to the relatively short half-life of tenofovir in plasma. The current study aimed at measuring Tenofovir-diphosphate (TFV-DP) concentrations in dried blood spots (DBS) in order to determine long-term adherence (Half-life ≈ 17 days) and Emtricitabine-triphosphate (FTC-TP) concentrations as a marker for short-term adherence (Half life ≈ 35 hours) (5). For this case-control study cases were defined as HIV/HBV co-infected adults on a stable tenofovir prodrug-containing regimen with HBV virologic breakthrough or persistent HBV viremia and controls as HIV/HBV co-infected adults with evidence of HIV viral suppression for greater than 6 months and HBV viral suppression on most recent assay. TFV-DP and FTC-TP levels were quantified by liquid chromatography/tandem mass spectrometry. Overall, 4 cases and 5 controls were included who were on TDF/FTC based therapy. Among patients on TDF, median (range) TFV-DP levels were 516 (215-1176) fmol/punch among unsuppressed patients (n=4) and 1456 (1089-3108) fmol/punch among suppressed patients (n=5). Levels were significantly lower among unsuppressed patients (p=0.03). FTC-TP levels were detectable among 3 of 4 unsuppressed and all of the suppressed patients. HBV genotyping assays performed on 4 unsuppressed patients did not detect TFV resistance. The main results are summarized in table 2.
Table 2: Main results
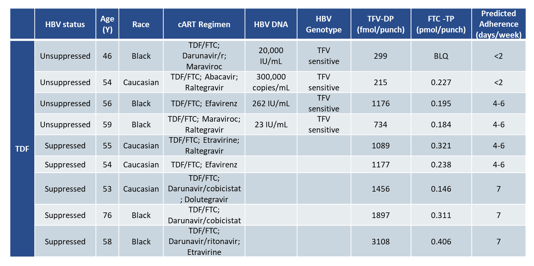
In conclusion, TFV-DP concentrations in DBS were lower among HBV unsuppressed patients than in suppressed patients. Adherence issues are the main reason for the lower drug levels. Detectable FTC-TP levels among the majority of HBV unsuppressed patients reflect recent drug adherence relative to the clinic visit ("white coat adherence"). Interim findings support the concern of poor long-term adherence to TDF therapy among patients with concurrent HBV viremia and HIV viral suppression.
Risk of HBV-reactivation in HIV/HCV coinfected subjects with anti-HBc-Antibodies receiving DAA-therapy.
In 2016 the FDA issued a Black box warning on possible HBV reactivation during DAA therapy in HCV-infected subjects. So far little data is available for HIV/HCV coinfected patients with past HBV infection and sole anti-HBc-Antibodies. As often anti-HIV and HBV dual active antiretroviral therapy is used, this risk may be much lower in HIV/HCV coinfection. The poster by Cerrone and colleagues evaluated the risk of HBV reactivation in 271 HIV/HCV coinfected subjects undergoing DAA therapy and receiving 48 weeks of follow-up (4). HBV reactivation was defined: ALT elevation of >2x ULN OR > 2x baseline + detectable HBV-DNA. Overall, 35% of included patients were HBcAb positive, 6% of them had chronic hepatitis B and were also HBsAg positive. 30% had low or no anti-HBs-Ab (<10 IU/l). 98% of the patients were receiving at least one HBV-active drug prior to DAA initiation. ARV switch to tenofovir (12%) or Entecavir addition (35%) were measures adopted to prevent HBV reactivation. Overall, no cases of HBV reactivation were detected, regardless of HBsAb titre. HBV-active ARV and ETV addition appear to be an effective strategy to prevent HBV reactivation in HIV patients undergoing anti-HCV treatment with DAAs. Further studies are warranted to assess the need of anti-HBV prophylaxis during DAA-based therapy in HIV co-infected patients.
HCV elimination in HIV-seropositive MSM: do we need even more evidence?
Early HCV treatment intervention in acute HCV infection could be very instrumental in helping to achieve microelimination in HIV-infected MSM by preventing further HCV transmissions. At the last two CROI conferences, first countries reported a 50% reduction in new incident cases of acute hepatitis C following identification of HIV-positive MSM and ongoing HCV replication and subsequent antiviral HCV therapy (14-18). At this year CROI, data from London was presented which was able to demonstrate similar trends for London after expansion of access to DA therapy (6). The authors used real world experience to examine trends in incidence of acute HCV in HIV+ MSM between 2013-2018 (pre and post DAAs) from three central London HIV clinics (Imperial College, Mortimer Market, and Royal Free) which provide care for over 6000 HIV+ MSM. Public healthcare was provided via the National Health Service (NHSE) and its DAA treatment program which began in 2015 with decompensated cirrhotics as priority. Access was expanded to all HCV disease stages in 2016 and there were capped monthly allocations per region, with lengthy waiting lists initially in London. Notable exceptions to DAA eligibility included acute infection until 6 months from presentation and HCV reinfection. All 3 sites were also involved in HCV coinfection trials during the study period including for acute and chronic non-cirrhotic disease. Over the 5 year period, peak incidence was observed in late 2015 with an incidence of 17/1000 PYFU and first infections at 15/1000 PYFU. Following the peak in the second half of 2015, the authors observed a 68% overall reduction of acute hepatitis C and nearly an 80% reduction in first infections (see figure 4).
Figure 4: Results: Incidence Rate Reduction
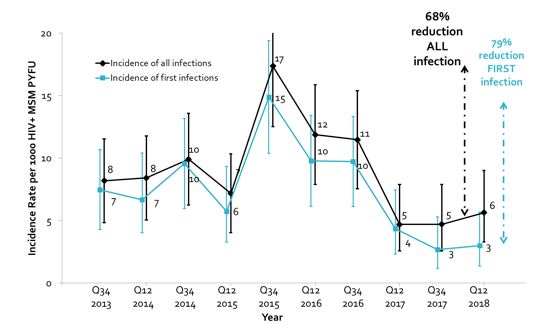
There was a constant rate of HCV reinfections with an increase to 40% in the last 12 months, clearly emphasizing that additional intervention will be needed for complete HCV elimination. Indeed, the large PROBE-C has reported HCV reinfection rates of around 25% within 2 years after first acute HCV episode (19).
This is the third report from HIV+MSM, where a sharp decline in new acute HCV diagnoses is reported since 2015 with no change in screening practices. The observed peak in 2015 is likely to represent a fall-off in rates of IFN-based treatments as DAAs were awaited; this 'warehousing effect' may have contributed to increased HCV transmission by longer duration of viremia. Wider prescribing of HCV therapies via NHSE DAA programs as well as reduction in time to treatment of acute HCV cases, which was largely driven by clinical trial availability, seem to account for the overall fall in incidence of 68%.
CROI: Fall in HCV incidence in HIV+ MSM in London following expansion of access to DAA therapy - (03/12/19)
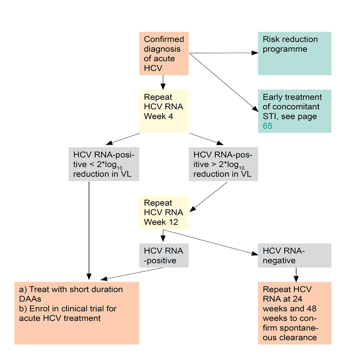
Currently, no direct acting antiviral therapy is licensed for the treatment of acute hepatitis C. Boesecke and colleagues analysed in the European Probe-C cohort in how far this impacts HCV treatment initiation rates in Europe among HIV+ MSM with acute hepatitis C (20). Overall, 464 acute hepatitis C episodes were included from 2007-2017. Impressively, IFN-containing therapy was no longer used for the early treatment of AHC coinfection in this pan-European cohort after 2015 (see figure 5). This was also observed in the presentation from London (see above) where a peak in acute HCV cases was noted in 2015. Although available and recommended by guidelines for treatment during the early acute phase of infection, DAA-based therapy was mostly deferred to post six-months after initial HCV diagnosis (see figure 6). The lack of a 2 log drop in HCV-RNA four weeks after first presentation however, was highly significantly associated with developing chronic HCV infection and may offer a new rule for deciding when to start DAA therapy (see current guideline recommendation in EACS algorithm figure 6). The delay in HCV treatment initiation increases the risk for further HCV transmissions. Clearly, guidelines will have to push stronger for earlier treatment of acute hepatitis C in order to eliminate HCV among HIV+ MSM.
Figure 5:Annual rates of treatment initiation within 6 months for new AHC diagnoses from 2007 to 2017
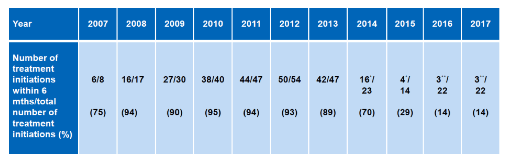
*Including 1 DAA-based treatment
**DAA-based treatments only
Figure 6: Management of acute HCV in HIV-coinfection from the EACS guidelines (see www.eacssociety.org)
Risk of HCV reinfections
One of the biggest challenges for HCV microelimination is the high rate of HCV reinfections particularly in MSM. Whereas free distribution of syringes and clean needles as well as access to opioid substitution therapy can dramatically reduce reinfections in active drug users, it appears much more difficult to change sexual risk behavior. Indeed, within the European PROBE C study a reinfection rate after two years of follow-up of up to 25% has been presented (19). Patients with a second episode of acute HCV were at even higher risk for developing a third episode of acute HCV following spontaneous clearance or successful HCV treatment. Data from the US on HCV reinfection risk in HIV-coinfection were missing so far. Colleagues from New York closed this gap by presenting data from the New York Acute Hepatitis C Surveillance Network. HIV-infected MSM with HCV infection were referred to practice (DSF) at Mount Sinai and enrolled in a longitudinal study which assessed for HCV clearance between 1 Jan 2000 and 31 Dec 2018 (7). Overall 305 HIV-seropositive individuals with cleared infection (33=spontaneously cleared, 106=cleared under IFN based therapy, 166=cleared under DAA therapy). During a median follow-up of 2.2 years (845 person years (PY)), 38 (12%) reinfections were noted. The overall incidence was 4.4/100 PY (95% CI 3.1, 6.0). Figure 7 depicts the time until first infection from this New York cohort.
Figure 7: First HCV-reinfection by time
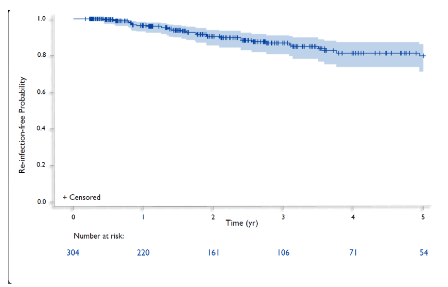
Within the 33 reinfections which cleared infection, 6 further reinfections were noted leading to an even higher incidence of 8.7/100 PY (95% CI 3.5, 18.1). Overall, Latinos had an almost twofold increased risk of reinfection alluding to disparities in the American Healthcare system.
In conclusion, a high re-infection rate similar to the one reported from Western Europe was also seen in NYC which demonstrates the inadequate level of HCV treatment among MSM in NYC, despite DAA "availability" in NYC since Dec'13. The difficulties in obtaining approval for treatment remain a major barrier to treatment access and microelimination.
CROI: Primary HCV in HIV+ MSM: US Epidemic Incidence Reinfections/NYC - (03/12/19)
Impact of SVR on non-hepatic disease manifestations
It is well known that HCV infection is associated with a higher risk of insulin resistance and eventually diabetes, although the underlying mechanism is not well understood. Inflammation caused by chronic HCV infection could be a factor or HCV may have a direct effect on glucose metabolism. Studies during the interferon era produced mixed results, with some showing that successful hepatitis C treatment reduced diabetes risk, while others linked interferon and ribavirin with new-onset diabetes (22,23). With the availability of all oral DAA therapies, much more patients can be successfully treated. Moreover, DAAs are considered metabolically neutral. Indeed first reports have shown an improvement in glycemic control of type 2 diabetes after successful treatment of hepatitis C virus (24). Butt and colleagues aimed at comparing the effect of different HCV treatments on the risk and incidence of diabetes among more than 52,000 US veterans, using data from the Electronically Retrieved Cohort of HCV Infected Veterans (ERCHIVES), a longitudinal national cohort of veterans with hepatitis C (8). After excluding individuals with HIV or HBV, those treated with both interferon-based therapy and DAAs, and those missing follow-up HCV viral load data to determine SVR, the analysis included 4,764 chronic hepatitis C patients treated with pegylated interferon plus ribavirin and 21,279 treated with DAAs, along with the same number of matched untreated control subjects.
Most study participants (96%) were men so the results of the study cannot be transferred to HCV infected women. About 54% were white, about 29% were black and the median age was approximately 60 years. Around 20% had advanced fibrosis or cirrhosis according to the non-invasive FIB-4 index. Among the treated patients, 79% achieved SVR, or continued undetectable HCV RNA 12 weeks after finishing treatment. During the follow-up period, 1,679 untreated veterans, 633 of those treated with pegylated interferon/ribavirin and 255 of those treated with DAAs were diagnosed with diabetes. The corresponding diabetes incidence rates were 20.6, 19.8 and 9.89, respectively, per 1,000 person-years. HCV treatment was associated with a larger reduction in diabetes incidence, particularly among people with more advanced fibrosis stages or cirrhosis. While diabetes incidence did not differ significantly between untreated and interferon-treated patients, it was substantially lower among those treated with DAAs. People who achieved SVR using either type of treatment were less likely to develop diabetes than those without SVR (13.3 vs 19.2 per 1,000 person-years). DAAs reduced the risk of diabetes by 52%, while achieving SVR lowered the risk by a more modest 19%. Figure 8 shows the Kaplan Meier analysis for probability of diabetes free survival by HCV treatment used (panel A) and between untreated and treated patients (panel B).
Figure 8: Kaplan Meier Curves.
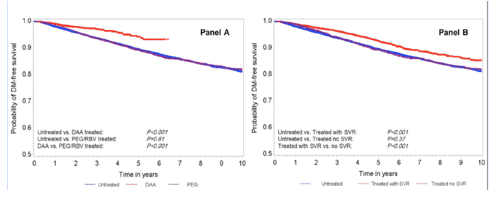
In summary, DAA therapy halved the risk of incident diabetes, but PEG/RBV had no significant impact on diabetes risk (HR 0.96, 95% CI 0.86 to 1.06 P = 0.38). The researchers concluded that treatment of HCV with DAA regimens confers benefits beyond virologic control and may be useful in controlling or mitigating some of the extrahepatic complications of HCV.
CROI: INCIDENT DIABETES AND GLUCOSE CONTROL AFTER HCV TREATMENT WITH DAAS IN ERCHIVES - (03/07/19)
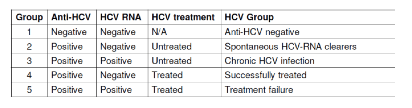
Interesting somewhat different results were reported from the large EuroSIDA cohort, which investigated the impact of HCV coinfection and HCV treatment outcome on risk of cardiovascular disease (CVD), non-AIDS defining malignancies (NAMD) and end-stage liver disease (ESLD) in HIV-1 infected individuals (9). All persons from the pan-European HIV cohort study EuroSIDA with known anti-HCV and HCV-RNA status after January 2001 were included and were divided into five groups based on time-updated HCV-RNA and HCV treatment status (see table 3).
Table 3: Studied patient groups
Separate analyses were performed for three clinical events; ESLD (including hepatocellular carcinoma [HCC]), NADM (excluding HCC) and CVD (myocardial infarction, stroke, angioplasty, coronary bypass, carotid endarterectomy). Poisson regression was used to compare incidence rates between HCV groups. Of 22,826 persons who were enrolled in EuroSIDA, 18,736 persons had known anti-HCV and HCV-RNA status, and were therefore eligible for inclusion into this analysis. The largest group was those anti-HCV negative (n=10433, 62.0%). Overall, most were male (74%), of white ethnic origin (85.2%), ever exposed to antiretroviral therapy (83.9%) and current smokers (54.3%) with a median age of 41(interquartile range [IQR] 35 – 49) and CD4 cell count of 438 (IQR 281 – 630 cells/μl) but as expected, there was considerable heterogeneity between the 5 groups at baseline. During a median follow-up of 8.3 (IQR 3.1 – 13.7) years, there was a total of 887 CVD, 902 NADM and 436 ESLD events (see figure 9).
Figure 9: Type of Clinical Events by Hepatitis C Strata
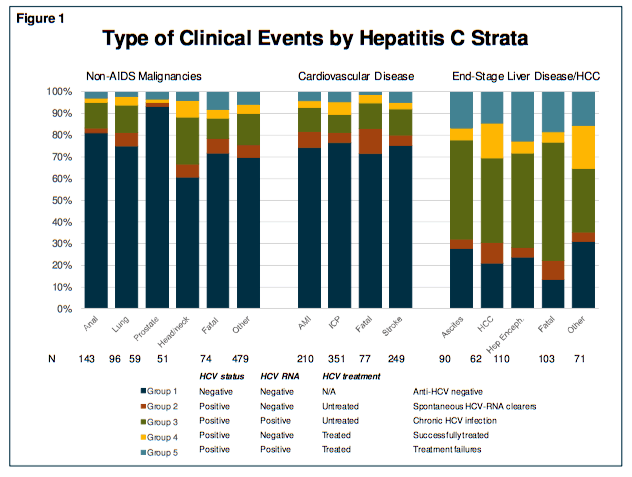
Figure 10 shows the univariate and adjusted rate ratios of CVD, NADM and ESLD, using those who were successfully treated for HCV (group 4) as the reference group. Persons with chronic infection (group 3; adjusted incidence rate ratio [aIRR] 1.47; 95% CI 1.02 – 2.13; p=0.041) or treatment failure (group 5; aIRR 1.80; 95% CI 1.22 – 2.66; p=0.0033) had significantly increased incidence rates of ESLD compared to those successfully treated (group 4). Spontaneous clearers also had marginally significantly lower incidence rates of ESLD compared to those successfully treated (group 3; aIRR 0.61; 95% CI 0.36 – 1.02; p=0.058). There were no statistically significant differences in incidence of CVD or NADM across the five groups. There was also no evidence that the association between HCV strata and each of the clinical events differed according to age (above or below 50, all p-interaction >0.15). In sensitivity analyses there were no differences between the 5 groups and CVD and NADM when excluding persons from Central East and Eastern Europe who tend to be younger and therefore have been infected with HCV for a shorter period of time. Analyzing myocardial infarctions and strokes separately also showed no differences between the HCV groups.
Figure 10: Univariate and Multivariate Incidence Rate Ratios of Clinical Events
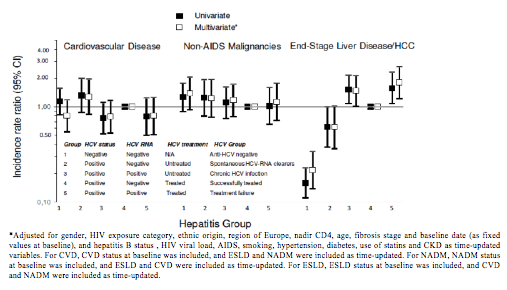
*Adjusted for gender, HIV exposure category, ethnic origin, region of Europe, nadir CD4, age, fibrosis stage and baseline date (as fixed values at baseline), and hepatitis B status , HIV viral load, AIDS, smoking, hypertension, diabetes, use of statins and CKD as time-updated variables. For CVD, CVD status at baseline was included, and ESLD and NADM were included as time-updated. For NADM, NADM status at baseline was included, and ESLD and CVD were included as time-updated. For ESLD, ESLD status at baseline was included, and CVD and NADM were included as time-updated.
In conclusion, this study had no evidence of any impact of HCV infection status or HCV treatment on incidence of both non-AIDS defining malignancies and cardiovascular disease in HIV coinfected persons. In contrast, successful HCV treatment lowered the incidence of end-stage liver disease and hepatocellular carcinoma compared to those chronically infected or those failing HCV treatment. The results underline the importance of early and effective HCV treatment for reducing liver-related complications in HIV/HCV infected persons.
CROI: Clinical Outcomes in Persons Coinfected with HIV and HCV: Impact of HCV Treatment - (03/07/19)
Spanish colleagues studied the effect of HCV eradication on bone mineral density in HIV/HCV coinfected subjects (10). Whereas it is common sense that cirrhosis is associated with osteoporosis, there is however, still some debate about the association between non-cirrhotic chronic HCV infection and osteoporosis. In HIV-coinfection HCV infection has been associated with an increased risk of bone loss and fracture (25). The mechanism is not well understood and may involve severity of liver disease, a reduction in bone mineral density, or microstructural abnormalities associated with HCV infection. BMD was prospectively analyzed at baseline and 96 weeks after initiation of anti-HCV therapy (Rx) in HIV/HCV-coinfected patients. Patients were recruited during 2012 – 2014 in 13 centers. Osteoporosis was defined as a T score ≤−2.5 SD and Osteopenia as a T score between −1 and −2.5 SD. Cirrhosis was assessed by FibroScan and defined as LS >12.5 kPa. Overall, 160 subjects (102 with SVR, 58 with no SVR) were included. The change in femoral neck categories is shown below in figure 11.
Figure 11: Change in femoral neck BMD categories
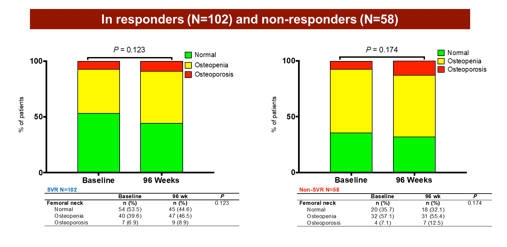
In the medium term, eradication of HCV following anti-HCV therapy was not associated with significant changes in BMD in HIV/HCV-coinfected persons. These findings so far do not support a causal association between HCV infection and reduced BMD in HIV/HCV-coinfected persons.
CROI: Effects of eradication of HCV on bone mineral density in HIV/HCV-coinfected patients - Bone density stable over 96 weeks after DAA cure in HIV/HCV group - - (03/07/19)
In summary, the data on outcome of non-hepatic diseases and events remains inconclusive. There was an over 50% reduction in new onsite diabetes in DAA treated and cured HCV patients but this did not seem to translate into a reduction in cardiovascular events in HIV/HCV coinfected patients, in a different cohort analysis. Indeed, as HIV induced inflammation persists, even after HCV cure, this may lead to differences in outcome of cardiovascular disease and non-hepatic other conditions in coinfection versus HCV monoinfection. More data and probably longer follow-up with more events are needed to come to conclusions. Disappointingly, despite an increased osteoporosis rate and fracture risk in HIV/HCV coinfection, no positive impact on BMD measurements was obtained after successful HCV eradication.
What are the risk factors for development of hepatocellular cancer (HCC) in HIV-coinfected individuals without advanced fibrosis?
The incidence of hepatocellular carcinoma (HCC) has increased 4-fold since 1996 with a 4x greater risk in HIV+ versus uninfected. Despite the rising incidence of HCC in HIV-coinfected subjects, few studies have assessed determinants of HCC during the antiretroviral treatment era. The Veterans Aging cohort study therefore, in a retrospective cohort study from 1999-2015, evaluated traditional as well as HIV-related risk factors for HCC development (21). The analysis included 2,497 people—about 8 percent—with advanced fibrosis or cirrhosis (FIB-4 > 3.5) and 29,836 people with absent, mild or moderate fibrosis (FIB-4 ≤3.5). In both groups, 51 and 52% were men respectively, and about half were African American. Those with advanced liver disease had a higher median age (50 versus 46) and were more likely to have hepatitis B (10 % versus 5 %), hepatitis C (59 % versus 30 %) or an alcohol-related diagnosis (47 % versus 29 %). In addition, they were more likely to have an HIV viral load above 500 copies/ml (63 %versus 56 %) and a CD4 count below 200 cells/mm³ (39 percent versus 26 %). During follow-up, 278 people were diagnosed with HCC. Most strikingly, 43 % of those with HCC did not have advanced fibrosis or cirrhosis according to FIB-4 scores. This clearly questions current HCC screening algorithms which concentrate on HCC screening in advanced fibrosis. The characteristics at HCC diagnosis are shown below in table 4.
Table 4: Baseline Characteristics at HCC diagnosis
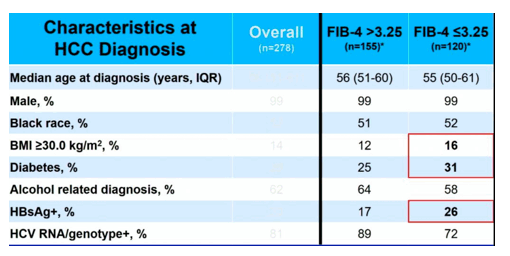
Patients with new HCC diagnosis with no or little fibrosis were more likely to have a BMI ≥ 30.0 kg/m², diabetes or HBV coinfection. HIV viral load and CD4 count had no significant effect on liver cancer risk among people with advanced fibrosis or cirrhosis. In contrast, in individuals without advanced fibrosis a higher viral load was associated with a 24 percent greater HCC risk. Having an HIV RNA level of 500 copies/ml or more conferred a 57 percent greater risk of liver cancer and having a CD4 count below 200 cells/mm³ was associated with a 58 to 78 percent higher risk. In summary, this study provides strong evidence that HIV viremia and a low CD4 cell count contribute to risk of HCC.
First study of HCV treatment in pregnant women: can this become a new treatment strategy to prevent perinatal HCV transmission?
Perinatal transmission of Hepatitis C virus (HCV) infection is a growing concern. Chappel and colleagues reported at this meeting for the first time results from DAA HCV treatment during pregnancy. In this open-label, phase 1 study, HIV-negative pregnant women with chronic genotype 1 HCV infection were enrolled between 23-24 weeks of gestation and began a 12-week course of LDV/SOF. Of 29 pregnant women with chronic HCV who were screened, 20 were excluded because of genotype 2 or 3 infection (n=10), ongoing illicit drug use (n=4), declining study participation (n=3), intensions to delivery off-site (n=2), and an APRI score of >1 (n=1). Nine women were enrolled, 8 achieved SVR one patient is still in follow-up. In this first study of HCV treatment in pregnant women, response to LDV/SOF was similar to the viral response observed in non-pregnant individuals without any safety concerns identified. However, larger studies are needed before this strategy can be recommended. Moreover, there is a need for treatment strategies in non-GT1 pregnant women as well.
CROI: A PHASE ONE STUDY OF LEDIPASVIR/SOFOSBUVIR IN PREGNANT WOMEN WITH HEPATITIS C VIRUS - (03/12/19)
Summary
•Long-term TDF-containing cART leads to higher rates of HBsAg loss (10-18%) in HIV/HBV coinfected subjects when compared to published data for HBV monoinfected subjects.
•ART-induced immune reconstitution may enhance anti-HBV immune responses in the liver.
•Poor long-term adherence to TDF therapy among HIV/HBV coinfected patients can lead to concurrent HBV viremia despite HIV viral suppression.
•HBV-active ARV and ETV addition appear to be an effective strategy to prevent HBV reactivation in HIV patients undergoing anti-HCV treatment with DAAs.
•Wider prescribing of DAA-based HCV therapies as well as reduction in time to treatment of acute HCV cases seem to account for the overall fall in incidence of acute HCV infections by 68%.
•IFN-containing therapy is no longer used for treatment of AHC coinfection in Europe after 2015. Although available and recommended by guidelines during the acute phase, DAA- based therapy is mostly deferred to the early chronic phase of HCV-infection. With more patients, being viremic now than in the interferon- era, drug labels need to be urgently amended to allow usage of DAA during the acute phase to limit HCV transmission in high-risk populations.
•HCV re-infection rate in NYC is 4.4/100 PY which is more than 7 times the primary infection rate in NYC, similar to the reinfection pattern seen in Western Europe. Higher reinfection rate in Latinos suggests healthcare disparities in the US.
•HCV treatment significantly reduces the incidence and risk of subsequent diabetes. The largest impact on new onset diabetes is seen under DAA-based therapy.
•Within the EuroSIDA cohort no evidence of any impact of HCV infection status or HCV treatment on incidence of both non-AIDS defining malignancies and cardiovascular disease in HIV coinfected persons was found. In contrast, successful HCV treatment lowered the incidence of end-stage liver disease and hepatocellular carcinoma compared to those chronically infected or those failing HCV treatment. The results underline the importance of early and effective HCV treatment for reducing liver-related complications in HIV/HCV infected persons.
•Eradication of HCV following anti-HCV therapy was not associated with significant changes in BMD in HIV/HCV-coinfected persons. These findings do not support a causal association between HCV infection and reduced BMD in HIV/HCV-coinfected persons.
•Among HIV-seropositive subjects without advanced liver fibrosis, higher HIV-RNA levels and longer duration of viremia, lower CD4-counts, morbid obesity, and diabetes in addition to viral hepatitis B and C coinfection, increased the risk of HCC.
•In a first study of HCV treatment in pregnant women, response to LDV/SOF was similar to the viral response observed in non-pregnant individuals without any safety concerns identified. However, larger studies are needed before this strategy can be recommended in a wider scale. Moreover, there is also a need for treatment strategies in non-GT1 pregnant women as well.
References
1. Boesecke C et al. Late presentation and its impact on HBV seroconversion in HBV/HIV. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0627
2. Chihota BV et al. High rates of HBV functional cure among HIV/HBV-coinfected patients on ART in Zambia. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0625
3. Jain MK et al. Hepatitis B cure in HIV patients is more likely in Hispanics and those with AIDS. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0624
4. Cerrone M et al. Absence of HBV reactivation in HIV/HCV/HBcAB coinfected patients treated with DAA. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0628
5. Zhang HL et al. Hepatitis B virologic failure to Tenofovir-based therapies in patients with HIV/HBV. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0626
6. Garvey LJ et al. Fall in HCV incidence in HIV+MSM in London following wider acess to DAA therapy. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0085
7. Corollo JR et al. HCV reinfection among HIV-infected MSM in New York City. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0086
8. Butt AA et al. Incident diabetes and glucose control after HCV treatment with DAAs in ERCHIVES. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0088
9. Peters L et al. Clinical outcomes in persons coinfected with HIV and HCV: impact of HCV treatment. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0565
10. Carrero A et al. Effects of Eradication of HCV on Bone Mineral Density in HIV/HCV-Coinfected Patients. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0687
11. Chappell CA et al. A Phase-1 study of ledipasvir/sofosbuvir in pregnant women with hepatitis C virus. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0087
12. Buti M, et al. Long-term safety and efficacy of nucleo(t)side analogue therapy in hepatitis B. Liver Int. 2018 Feb;38 Suppl 1:84-89.
13. Dusheiko G, et al. HBsAg loss in chronic hepatitis B: pointers to the benefits of curative therapy. Hepatol Int. 2016 Sep;10(5):727-9.
14. Braun D et al., for the Swiss HIV Cohort Study. A treatment as prevention trial to eliminate HCV in HIV+ MSM: The Swiss HCVREE trial. 25th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2018, Boston; abstract LB87
15. Boerekamp A et al. Unrestricted DAA access in the Netherlands: Rapid therapy uptake in HIV+HCV+ patients. 24th Conference on Retroviruses and Opportunistic Infections, February 13-16, 2017, Seattle; abstract 136
16. Boerekamp A et al.: Substantial decline in acute HCV infections among Dutch HIV+MSM after DAA roll out. 24th Conference on Retroviruses and Opportunistic Infections, February 13-16, 2017, Seattle; abstract 137LB
17. Boerekamps A et al.: NVHB-SHM hepatitis working group and the Netherlands ATHENA HIV observational cohort. High treatment uptake in HIV/HCV-coinfected patients after unrestricted access to direct-acting antivirals in the Netherlands. Clin Infect Dis. 2017 Nov 23. doi: 10.1093/cid/cix1004. [Epub ahead of print]
18. Boerekamps A et al.: Declining HCV incidence in Dutch HIV positive men who have sex with men after unrestricted access to HCV therapy. Clin Infect Dis. 2017 Nov 23. doi: 10.1093/cid/cix1007. [Epub ahead of print]
19. Ingiliz P, et al. NEAT study group. HCV reinfection incidence and spontaneous clearance rates in HIV-positive men who have sex with men in Western Europe. J Hepatol. 2017 Feb;66(2):282-287.
20. Boesecke C et al. Can`t buy me love? Obstacles to microelimination of acute HCV coinfection in Europe. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0576
21. Torgersen J et al. HIV viremia and low CD4+ increase HCC risk in those without advanced liver fibrosis. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0090
22. Pandya P, et al. Impact of sustained virological response to chronic hepatitic C antiviral therapy on new onset diabetes mellitus type 2 after controlling for metabolic syndrome. J Investig Med. 2017;65:765-771.
23. Zornitzki T, et al. Interferon therapy in hepatitis C leading to chronic type 1 diabetes. World J Gastroenterol. 2015;21:233-9.
24. Hum J, et al. Improvement in Glycemic Control of Type 2 Diabetes After Successful Treatment of Hepatitis C Virus. Diabetes Care. 2017;40:1173-1180.
25. Bedimo R et al. Hepatitis C virus coinfection as a risk factor for osteoporosis and fracture. Curr Opin HIV AIDS 2016;11:285-293
|
| |
|
 |
 |
|
|