 |
 |
 |
| |
TOCILIZUMAB (monoclonal antibody) ALTERS LIPIDS IN HIV+ INDIVIDUALS IN A RANDOMIZED, DOUBLE-BLIND STUDY
|
| |
| |
Download the PDF here
IL-6 blockade may reduce some indices of inflammation in HIV+ individuals, but also exacerbates lipid levels, potentially limiting benefits in this population. Further study is needed to determine the consequences of TCZ-mediated lipidome alterations on CVD risk.
CROI 2019
Reported by Jules Levin
NATAP
Emily Bowman1, Morgan Cichon1, Kenneth Riedl1, Jane Baum2, Michael L. Freeman2, Michael M. Lederman2, Benigno Rodriguez2, Nicholas Funderburg1
1The Ohio State University, Columbus, OH, USA,2Case Western Reserve University, Cleveland, OH, USA
Abstract
Cardiovascular disease (CVD) risk is increased in HIV infection, despite suppressive antiretroviral therapy (ART). Increased IL-6 levels are linked to CVD, and are predictive of morbidity and mortality in HIV infection. Tocilizumab (TCZ), a monoclonal antibody that inhibits IL-6 activity, can reduce inflammation and improve disease outcomes in individuals with rheumatoid arthritis (RA). Increased serum lipids (total cholesterol, HDL, LDL) were observed following TCZ treatment, but were not significantly linked to CVD risk in the RA population. The effects of TCZ on inflammation, lipid profiles, and clinical outcomes in HIV+ individuals is not known.
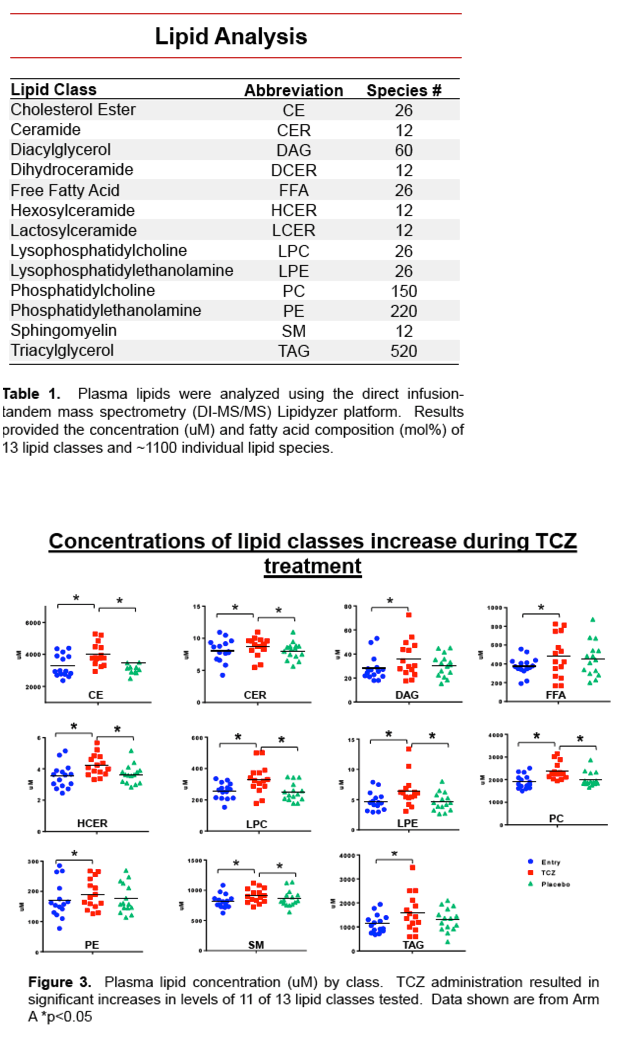
This was a phase I/II double-blind, placebo controlled, crossover trial of TCZ administered intravenously (IV) every 4 weeks for 3 doses. Male and female ART-treated HIV+ study participants were randomized to receive either TCZ or placebo followed by a 12 week washout period and treatment crossover. At each study visit, lipid panels and detailed lipidomics analyses, measuring ∼1200 lipid species across 13 classes, were performed by mass spectrometry.
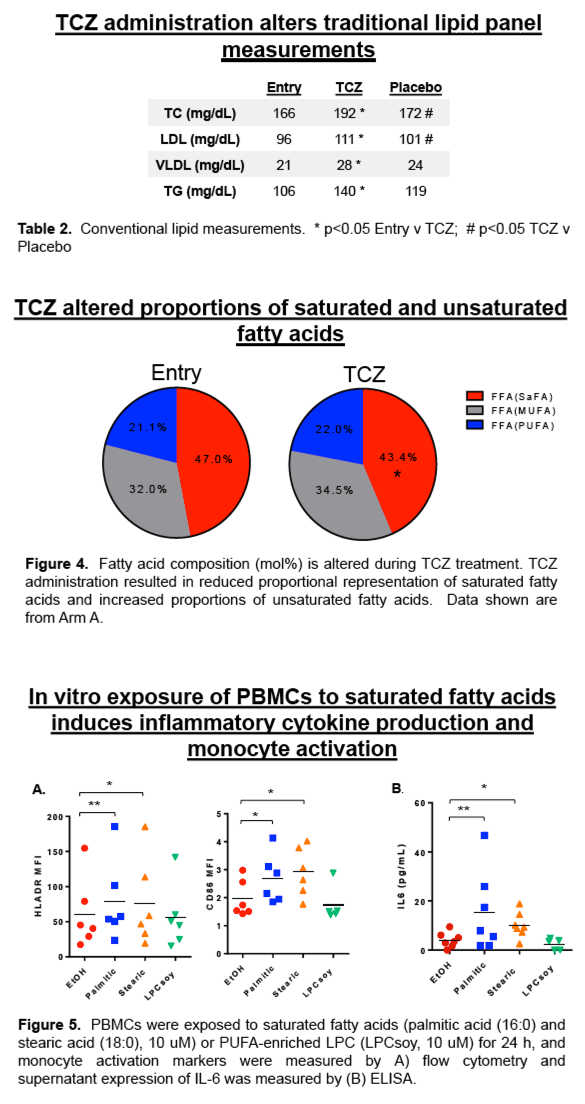
Traditional lipid measurements for total cholesterol, LDL, and VLDL levels were increased following TCZ treatment (p<0.01 for all). Plasma concentrations of total lipids (p=0.0001), and concentrations of the lipid classes, CE, CER, DAG, FFA, HCER, LPC, LPE, PC, PE, SM, TAG, were increased following TCZ treatment compared to baseline and placebo (p<0.05 for all). We also measured significant changes in concentrations of 129 individual lipid species (p<0.05). Additionally, fatty acid composition was altered among lipid species; TCZ treatment reduced the proportion of free saturated fatty acids (SaFAs) (47% vs 43%, p=0.05), and increased the proportion of free monounsaturated fatty acids (MUFAs) (32% vs. 35%, p=0.06) and polyunsaturated fatty acids (PUFAs) (21% vs 22%). In vitro exposure of PBMCs to SaFAs induced inflammatory cytokine production and monocyte activation.
TCZ therapy alters lipid profiles in HIV+ individuals on ART. The concentrations of multiple lipid classes increased during TCZ treatment, however, the SaFA/UFA ratio was improved for some classes. IL-6 blockade may reduce some indices of inflammation in HIV+ individuals, but also exacerbates lipid levels, potentially limiting benefits in this population. Further study is needed to determine the consequences of TCZ-mediated lipidome alterations on CVD risk.
|
| |
|
 |
 |
|
|