 |
 |
 |
| |
Longitudinal Phenotyping of Declarative Memory Among Women
Living with HIV
|
| |
| |
CROI 2019
Reported by Jules Levin
Kathryn C. Fitzgerald1, Pauline M. Maki2, Yanxun Xu3, Wei Jin3, Gayle Springer4, Kathryn Anastos5, Deborah Gustafson6, Seble Kassaye7, Victor G. Valcour8, & Leah H. Rubin1
1Johns Hopkins University School of Medicine, Baltimore, MD, 2University of Illinois at Chicago, Chicago, IL, USA, 3Johns Hopkins University, Baltimore, MD, USA, 4Johns Hopkins Bloomberg School of Public Health, Baltimore, MD, USA,
5Albert Einstein College of Medicine, Bronx, NY, USA, 6SUNY Downstate Medical Center, Brooklyn, NY, USA, 7George Washington University, Washington, DC, USA, 8University of California San Francisco, San Francisco, CA, USA
program Abstract:
There is considerable heterogeneity in patterns of cognitive decline in HIV-infected (HIV+) individuals. Age, sex, and race contribute to individual differences in patterns of cognitive decline, including in the domain of verbal memory. Advanced statistical models can be applied to parse out the heterogeneity in cognitive decline and to identify subgroups of individuals who might benefit from interventions tailored to their particular pattern of change and risk factors. Here, we applied a novel statistical method to identify clusters of individuals with distinct patterns of age-related change in declarative memory in HIV+ and HIV-uninfected (HIV-) women.
We included 1530 women from the Women's Interagency HIV Study who completed the Hopkins Verbal Learning Test at two or more visits. To derive subgroups with similar patterns of decline by HIV-serostatus, we applied a novel modeling strategy that simultaneously considers multiple longitudinal declarative memory outcomes. This model adopts a linear mixed-effects framework to model the trajectory of each cognitive outcome over time, while also jointly clustering individuals via a factor analysis model. We tested for differences in demographic and clinical characteristics between the clusters using a multivariable-adjusted multinomial model.
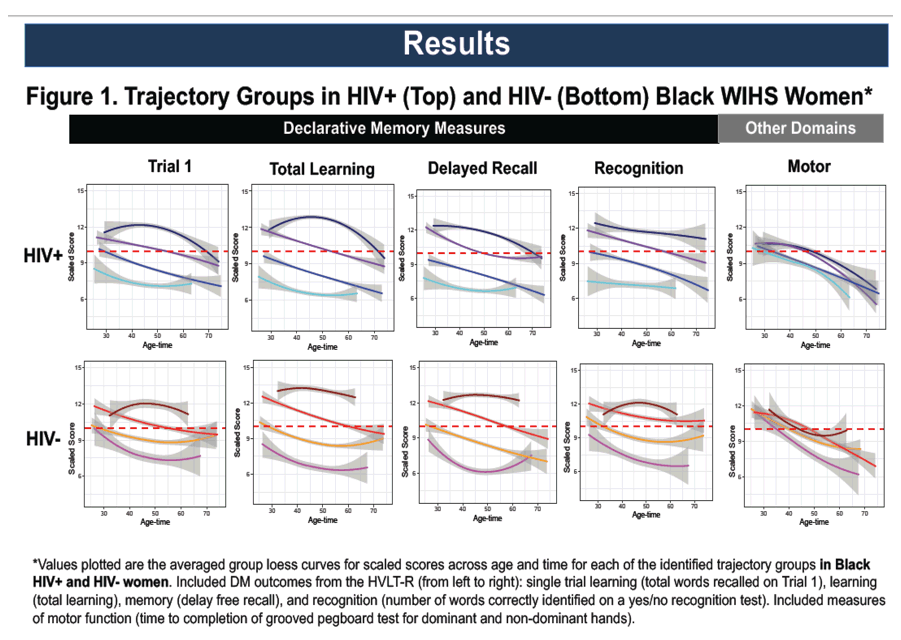
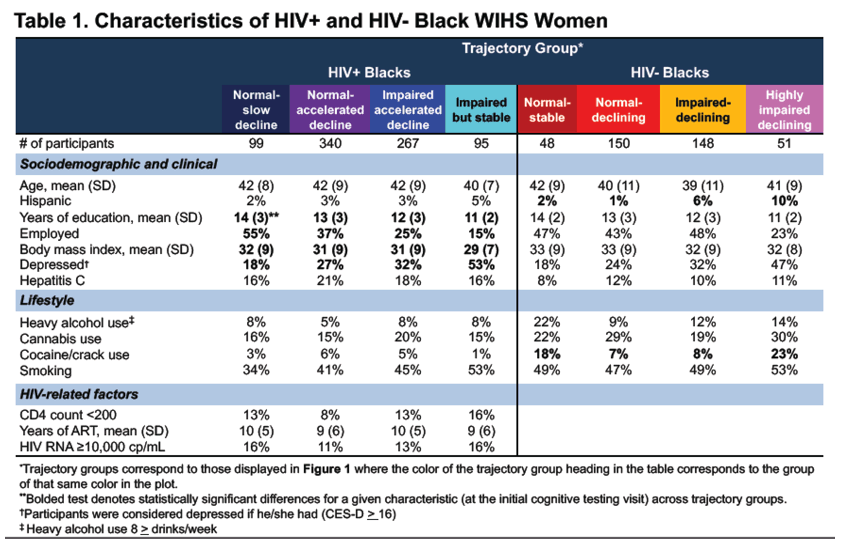
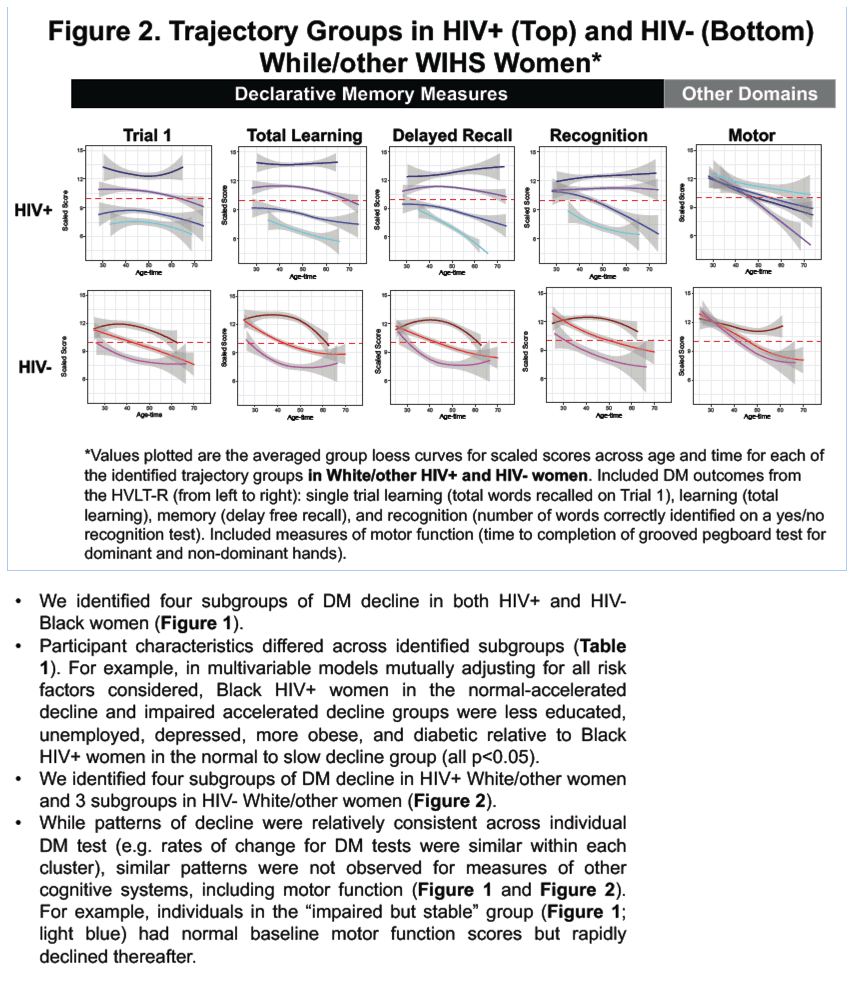
Of the 1530 included participants, 1167 were HIV+ (69% African-American [AA]; 31% white/other [W/O]) and 586 were HIV- (68% AA; 32% W/O). Stratification by race was necessary to optimize clustering. In the HIV+ AA's, we identified four distinct subgroups with differential patterns of change in memory: a subgroup with minimal decline, two with accelerated decline, and a subgroup with stable impairment in learning and memory (Fig 1A). In the HIV- AA's, we identified three subgroups: one with lesser decline and two with accelerated decline (Fig 1B). In multivariable adjusted models, individuals with accelerated decline were more likely to be less educated (P<0.001) and have a history of depression (P<0.001) versus those in the minimal decline subgroups (Fig 1C). Similarly classified subgroups were identified in W/O HIV+ and W/O HIV- participants.
Our data-driven modeling approach successfully identified clinically meaningful subgroups of individuals with distinct phenotypes of declarative memory decline. Depression was a key, potentially modifiable determinant of membership in a subgroup characterized by more rapid decline.


|
| |
|
 |
 |
|
|