 |
 |
 |
| |
Single monoclonal antibody infusion controls
HIV for over 5 months in 2 people
|
| |
| |
Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2019, Seattle
Mark Mascolini
A single infusion of the PGT121 monoclonal antibody (mAb) pushed HIV load below detection and kept it there for over 5 months in 2 study participants not taking antiretroviral therapy (ART) [1]. These 2 people had the lowest pretreatment viral loads in the study, below 1000 copies.
PGT121 is a potent mAb that targets HIV's V3 envelope region critical to HIV binding to cellular CD4. This mAb neutralizes 60% to 70% of global HIV-1 viruses. Previous work in monkeys showed that PGT121 lowers viral load in SHIV-infected animals, delays viral rebound after treatment interruption when combined with a TLR7 agonist, and protects monkeys against SHIV challenge at a concentration below 5 ug/mL. At CROI researchers from Boston's Beth Israel Deaconess Medical Center presented the first trials of PGT121 in humans.
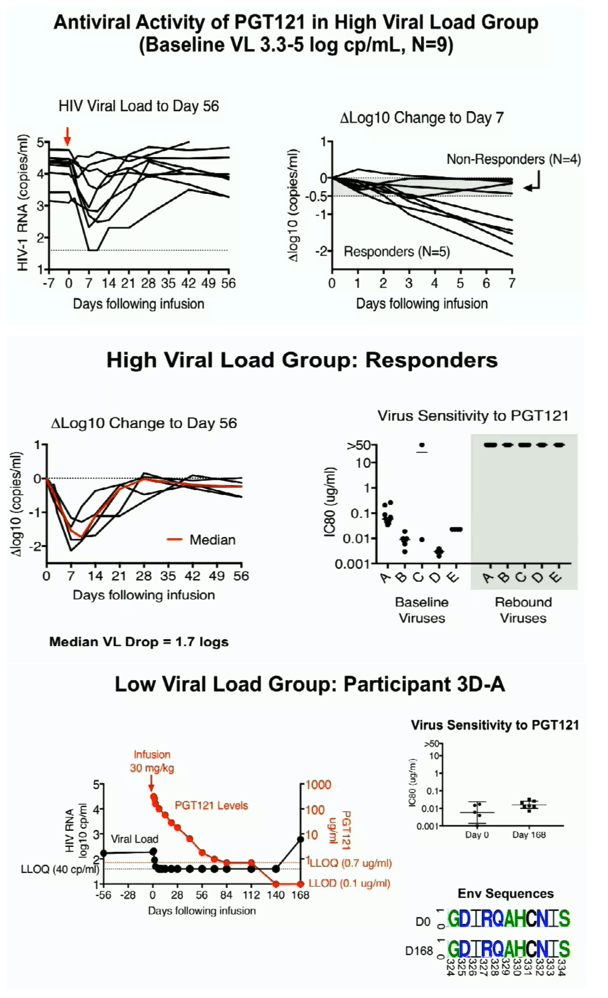
Beth Israel investigators described two studies, the first a randomized, double-blind, placebo-controlled, dose-escalation study in 20 HIV-negative adults and 15 antiretroviral-treated adults with undetectable HIV. Participants received an intravenous (IV) infusion of 3, 10, or 30 mg/kg of PGT121 or 3 mg/kg subcutaneously. There were 5 people per dose group, 1 of whom got placebo. The second part of the study was an open-label trial of a 30 mg/kg intravenous infusion in HIV-positive adults who had not started ART. Nine people had a high viral load (3.3 to 4.8 log10 copies, or about 2000 to 63,000 copies) and 4 had a low viral load (2.2 to 2.6 log10 copies, or about 160 to 400 copies). Nobody in this part of the study took placebo. Together the two studies now have 41 people taking the mAb and 7 taking placebo.
Age averaged 44 years in HIV-positive people on ART, 31 in HIV-positive people not on ART, and 27 in HIV-negative people. Body mass index for those three groups averaged 25 to 26 kg/m2. Half of the HIV-negative group and about 10% of HIV-positive people were women. Blacks and Asians made up about one quarter of the three study groups.
In both studies IV and subcutaneous PGT121 were safe and well tolerated, with low local reactogenicity (usually tenderness or erythema in about 25% of participants) and low systemic reactogenicity (usually headache or malaise in under 25%). There were no mAb-related moderate or severe adverse events. Median PGT121 elimination half-life stood at 23.5 days in HIV-negative people, 19 days in HIV-positive people on ART, and 13 days in HIV-positive people not on ART. CD4 count did not change substantially after PGT121 infusion in people with HIV.
Among HIV-positive people not taking ART, 5 of 9 in the high viral load group responded to the mAb virologically with a median viral load drop of 1.7 log (about 50-fold) by day 7, after which viral loads rebounded in everyone. These participants had PGT121-sensitive virus when starting the mAb, and HIV in all became PGT121-resistant upon viral rebound by day 28. The researchers proposed that HIV rebounded in these people when PGT121 had completely left their system.
Two people in the low viral load group had rapid drops in viral load to below 40 copies after receiving the 30 mg/kg PGT121 infusion. As PGT121 levels declined steadily, viral load remained below the 40-copy limit without ART through 140 days in both participants. By day 140 PGT121 levels become completely undetectable (below 0.1 ug/mL) in 1 person and viral load rebounded above the 40-copy detection limit by day 168. Rebound HIV was still sensitive to PGT121. The other person maintained a sub-40-copy viral load without ART through day 168 as PGT121 levels hovered at the detection limit of 0.7 ug/mL. The Beth Israel team suggested that PGT121 remains effective even at this low level.
The 5-month and longer viral undetectability periods in these 2 people represent the longest observed viral suppression without ART after a single broadly neutralizing antibody infusion in people with HIV. The Beth Israel team saw no evidence that enhanced cellular immune responses explained this sustained suppression. Rather they attributed viral suppression to the "exquisite potency of PGT121, even at levels below the limit of quantitation."
Kathryn Stephenson, the presenting researcher, speculated that PGT121 would have to be combined with another mAb in people with a viral load above 1000 copies. But in those with a lower viral load--perhaps a low load attained with ART--PGT121 may prove effective as a single agent. She suggested that the high potency of PGT121 may also support its use as long-acting preexposure prophylaxis (PrEP).
Reference
1. Stephenson KE, Julg B, Ansel J, et al. Therapeutic activity of PGT121 monoclonal antibody in HIV-infected adults. Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2019. Seattle. Abstract 145LB.
WEBCAST: [endif] http://www.croiwebcasts.org/console/player/41314?mediaType=slideVideo&

|
| |
|
 |
 |
|
|