 |
 |
 |
| |
Efficacy and safety of bictegravir/emtricitabine/tenofovir alafenamide vs comparators in cis-women and girls (living with HIV): an analysis of 5 clinical trials
|
| |
| |
High Response Rates, Good Safety with B/F/TAF in Women and Girls in 5 Trials
17th European AIDS Conference, November 6-9, 2019, Basel
Mark Mascolini
Combined analysis of five phase 2 or 3 trials documented high 48-week response rates with single-tablet bictegravir/emtricitabine/tenofovir alafenamide (B/F/TAF) in cis-women and girls [1]. Only 1 person in these trials stopped B/F/TAF because of an adverse event.
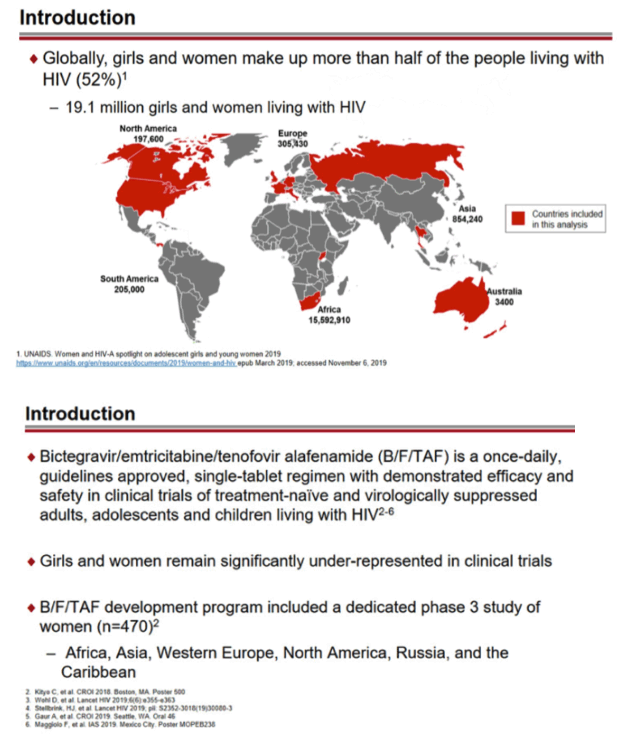
Women and girls account for more than half of people living with HIV globally, yet most clinical trials still enroll low proportions of females. To get a better understanding of B/F/TAF efficacy and safety in women and girls, researchers conducted a combined analysis of five trials comparing the once-daily combination with dolutegravir-based regimens in previously untreated people or with an ongoing regimen in three switch studies.
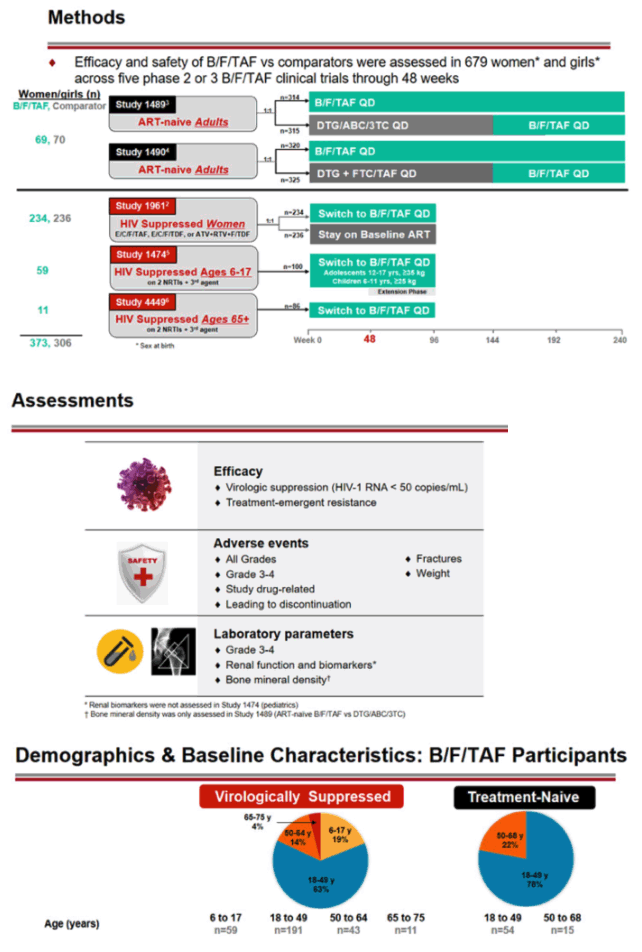
The combined analysis included 373 women and girls randomized to B/F/TAF and 306 randomized to a comparator regimen. The primary efficacy analysis was the proportion of participants with a viral load below 50 copies at week 48 by FDA snapshot analysis.
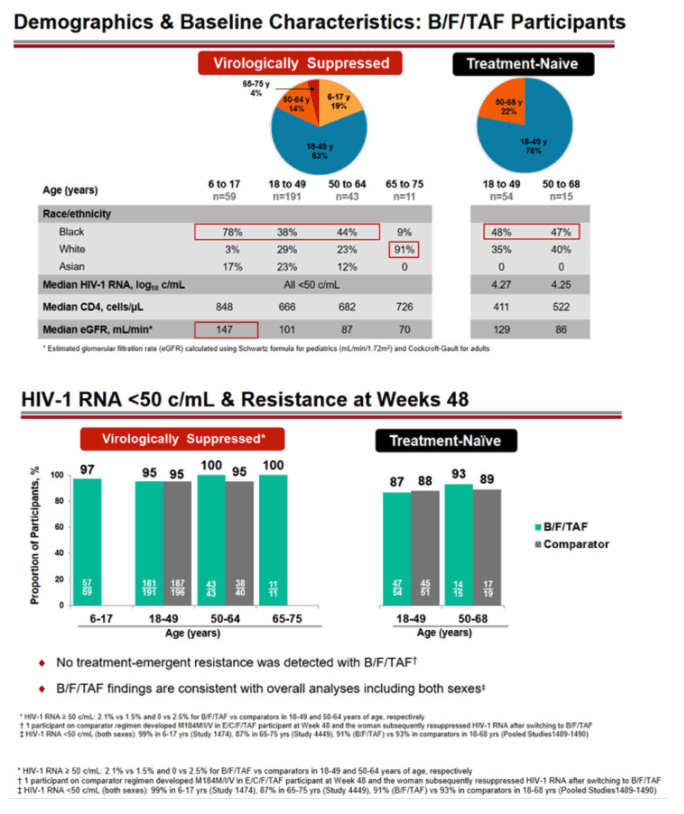
Among virologically suppressed people in switch trials, 19% were 6 to 17 years old, 63% were 18 to 49, 14% were 50 to 64, and 4% were 65 to 75. Across those four age groups, 78%, 38%, 44%, and 9% were black and 3%, 29%, 23%, and 91% white. Median CD4 counts across those four groups were 848, 666, 682, and 726. Among treatment-naive participants starting their first regimen, 78% were 18 to 49 and 22% were 50 to 68. In those two age groups, 48% and 47% were black, and median CD4 counts stood at 411 and 522.
Among women and girls switching to B/F/TAF from another regimen, at 48 weeks virologic suppression rates were 97% in 6-to-17-year-olds, 95% in 18-to-49-year-olds (and 95% in comparator participants this age), 100% in 50-to-64-year-olds (and 95% in comparator participants this age), and 100% in 65-to-75-year-olds. In women starting their first antiretroviral regimen, 48-week response rates were 87% in 18-to-49-year-olds (and 88% in comparator participants that age) and 93% in 50-to-68-year-olds (and 89% in comparator participants that age). Treatment-emergent resistant virus developed in no one randomized to B/F/TAF and in 1 person taking elvitegravir/cobicistat/F/TAF.
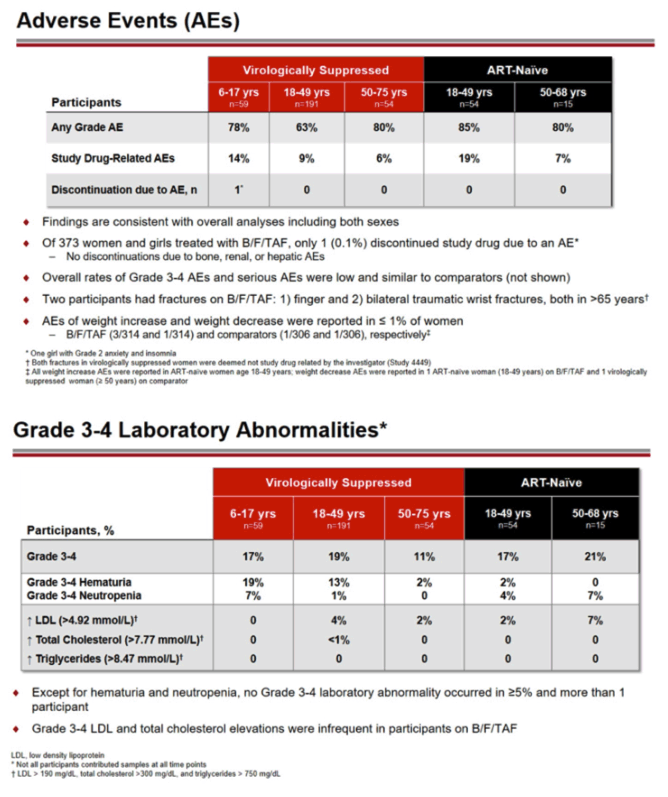
Among virologically suppressed women and girls switching to B/F/TAF, rates of study drug-related adverse events were 14% in 6-to-17-year-olds, 9% in 18-to-49-year-olds, and 6% in 50-to-75-year-olds. Among 373 women and girls taking B/F/TAF, 1 girl withdrew because of an adverse event, grade 2 anxiety and insomnia. In antiretroviral-naive trial participants, 19% of 18-to-49-year-olds had drug-related adverse events, as did 7% of 50-to-68-year-olds. About 1% of women had adverse events described as increased or decreased weight.
In the switch trials, grade 3 or 4 lab abnormalities affected 17% of 6-to-17-year-olds, 19% of 18-to-49-year-olds, and 11% of 50-to-75-year-olds. Among antiretroviral-naive women starting B/F/TAF, 17% of 18-to-49-year-olds and 21% of 50-to-68-year-olds had grade 3 or 4 lab abnormalities. Except for hematuria and neutropenia, no grade 3 or 4 lab abnormality affected more than 1 participant.
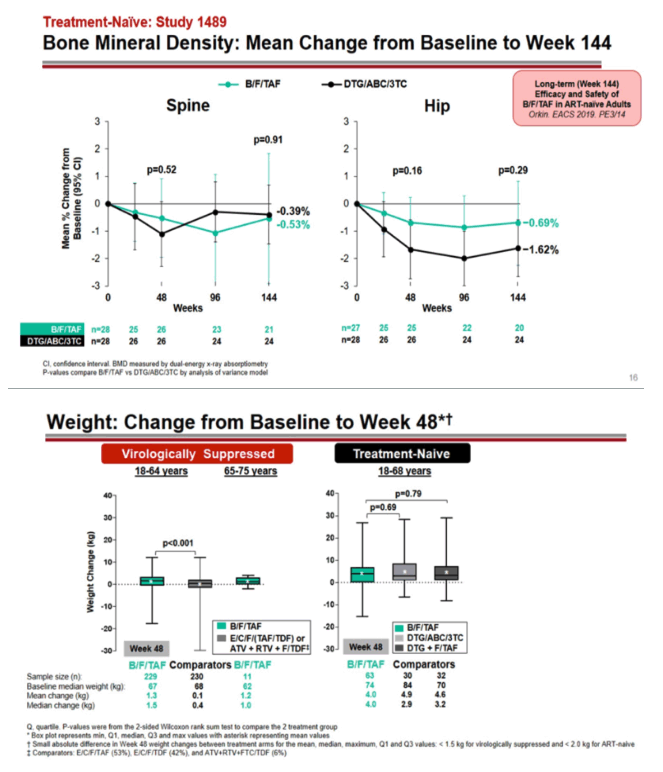
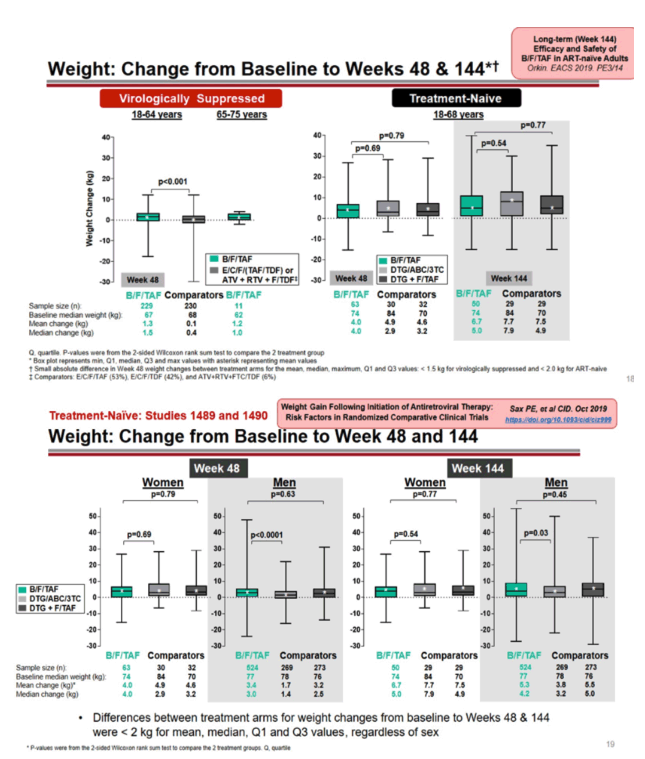
Among virologically suppressed 18-to-64-year-olds, those switching to B/F/TAF gained an average 1.3 kg in weight (median 1.5 kg) during 48 weeks, a small gain significantly higher than in participants maintaining the same regimen (average 0.1 kg, median 0.4 kg) (P < 0.001). In treatment-naive 18-to-68-year-old trial participants, weight gain with B/F/TAF did not differ significantly from gains with dolutegravir/abacavir/lamivudine or dolutegravir plus F/TAF (median 4.0, 2.9, and 3.2 kg). Those comparisons did not become significant through 144 weeks (median 5.0, 7.9, and 4.9 kg).
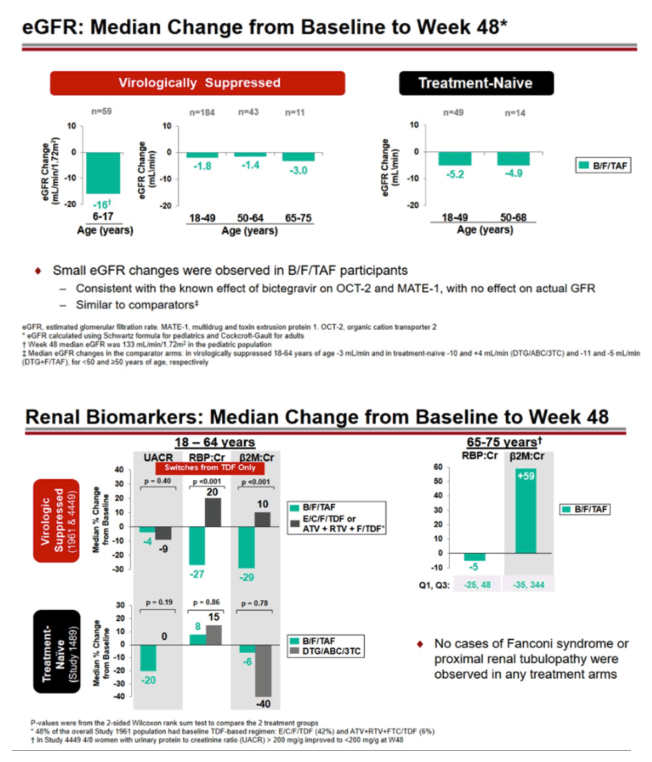
In the switch trials, though 48 weeks the kidney function marker estimated glomerular filtration rate (eGFR) dropped a median 16 mL/min in 6-to-17-year-olds taking B/F/TAF, 1.8 mL/min in 18-to-49-year-olds, 1.4 mL/min in 50-to-64-year-olds, and 3.0 mL/min in 65-to-75-year-olds. In treatment-naive women taking B/F/TAF for 48 weeks, median declines in eGFR were 5.2 mL/min in 18-to-49-year-olds and 4.9 mL/min in 50-to-68-year-olds.
In the trial comparing B/F/TAF with dolutegravir/abacavir/lamivudine, spine bone mineral density fell slightly in both arms with no significant difference at week 48 or 144 (-0.53% with B/F/TAF, -0.39% with the dolutegravir regimen, P = 0.91). Through 144 weeks, hip bone mineral density fell nonsignificantly less with B/F/TAF than with the dolutegravir regimen (-0.69% versus -1.62%, P = 0.29).
Reference
1. Orkin C, Ajana F, Kityo C, et al. Efficacy and safety of bictegravir/emtricitabine/tenofovir alafenamide vs comparators in cis-women and girls (living with HIV): an analysis of 5 clinical trials. 17th European AIDS Conference, November 6-9, 2019, Basel. Abstract PS7/6.


|
| |
|
 |
 |
|
|