 |
 |
 |
| |
The ADVANCE trial: the impact of DXA-assessed bone mineral density of TDF/FTC/EFV and TDF/FTC+DTG versus TAF/FTC+DTG South Africa
|
| |
| |
17th European AIDS Conference, November 6-9, 2019, Basel
Mark Mascolini
After 96 weeks in the South African ADVANCE trial, markers of bone or kidney health differed modestly between people randomized to tenofovir alafenamide (TAF)/emtricitabine (FTC) + dolutegravir (DTG) versus tenofovir disoproxil fumarate (TDF)/FTC+DTG, always favoring the TAF arm [1]. The FRAX equation predicted a lower 10-year risk of major osteoporotic fractures with TAF/FTC+DTG than with TDF/FTC+DTG.
ADVANCE randomized 1053 previously untreated South Africans 12 years old or older to TAF/FTC+DTG, TDF/FTC+DTG, or TDF/FTC + efavirenz (EFV). After 48 weeks, proportions in those three groups with a viral load below 50 copies were 84%, 85%, and 79% [2]. At that point, TAF/FTC+DTG affected bone density and kidney function less than the other two regimens. In a 96-week analysis reported separately by NATAP, weight continued rising through the last follow-up point in people taking TAF/FTC+DTG, and metabolic syndrome developed more in that group than in the others [3].
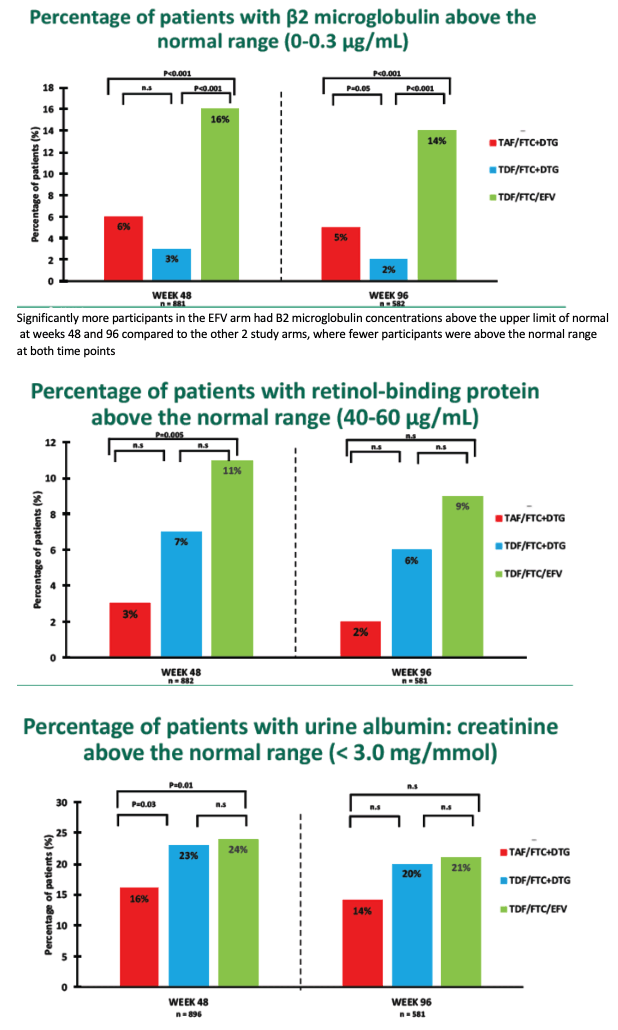
Researchers used DXA scans to measure bone mineral density (BMD) for the whole body, spine, and hip at study entry and after 48 and 96 weeks. They used the FRAX tool to estimate fracture risk in all participants older than 40. Kidney lab tests included uric acid, phosphate, beta-2 microglobulin, urine retinol-binding protein, serum creatinine, creatinine clearance, and urine albumin-to-creatinine ratio.
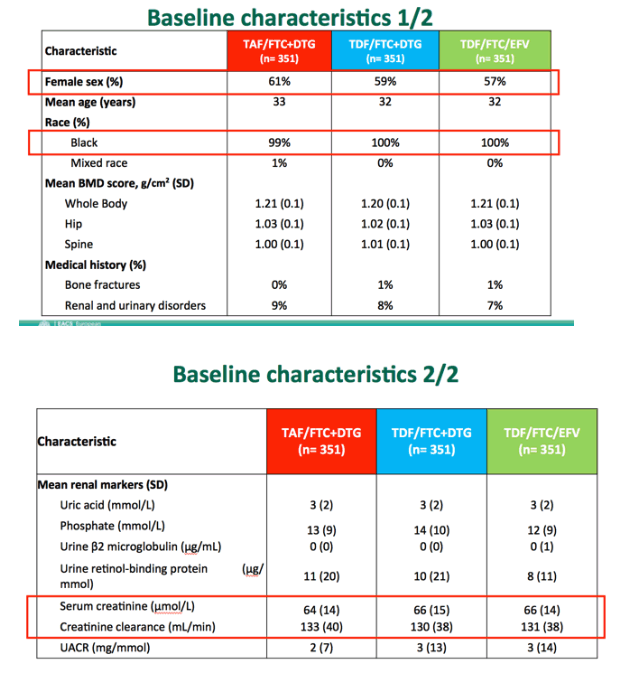
In the TAF/FTC+DTG, TDF/FTC+DTG, and TDF/FTC+EFV arms, 61%, 59%, and 57% were women, age averaged 33, 32, and 32, and nearly everyone was black. Almost no one had a previous fracture, and only about 8% had a kidney or urinary disorder. Everyone entered the trial with creatinine clearance above 60 mL/min (above 80 mL/min if younger than 19 years). Pretreatment creatinine clearance averaged 133, 130, and 131 mL/min in the three study arms.
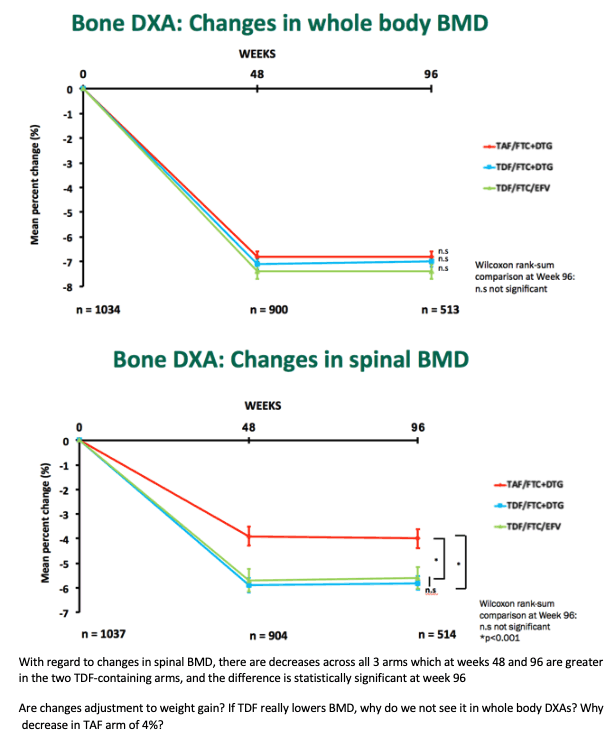
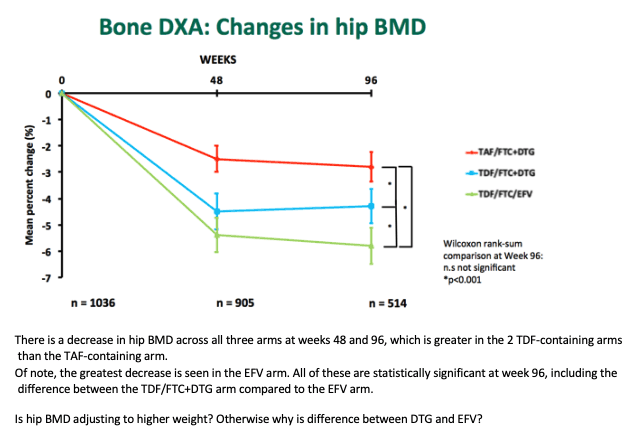
Through 96 weeks whole-body BMD fell about 7% in all three treatment arms. Spine BMD dropped about 6% with the two TDF regimens but only about 4% with the TAF regimen, a significant difference (P < 0.001). Through 96 weeks hip BMD fell almost 3% with TAF/FTC+DTG, about 4% with TDF/FTC+DTG (P < 0.001 versus TAF/FTC+DTG), and about 6% with TDF/FTC+EFV (P < 0.001 versus either DTG arm).
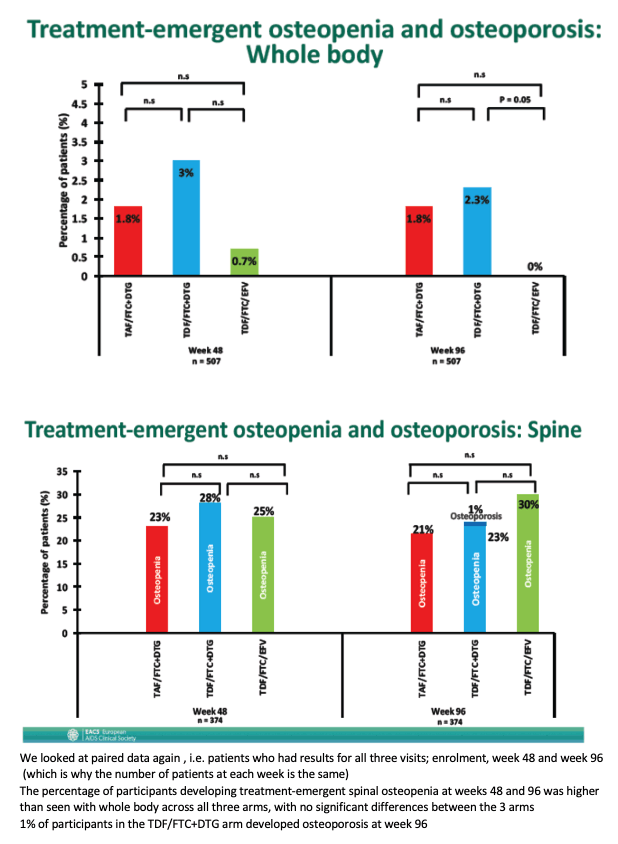
At week 96 WHO-defined treatment-emergent whole-body osteopenia arose in 1.8% taking TAF/FTC+DTG, 2.3% taking TDF/FTC+DTG (difference not significant from TAF arm), and 0% with TDF/FTC+EFV (P = 0.05 versus TDF/FTC+DTG). Treatment-emergent osteopenia of the spine did not differ significantly between the three study arms at week 96. At the same point treatment-emergent osteopenia of the hip was significantly lower with TAF/FTC+DTG (6.3%) than with TDF/FTC+EFV (30%, P < 0.001) but did not differ significantly from TDF/FTC+DTG (11.1%).
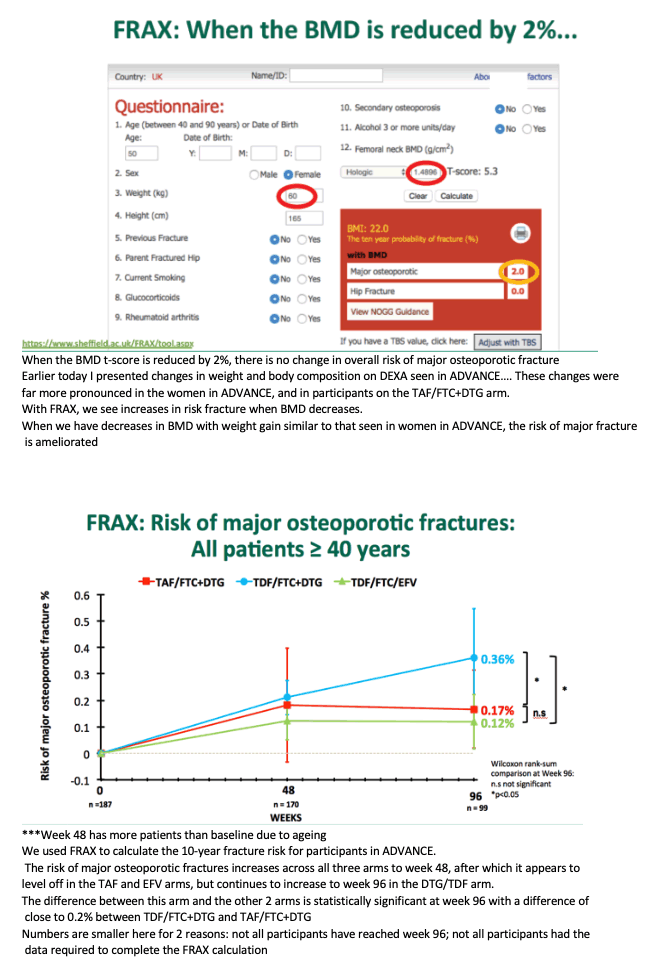
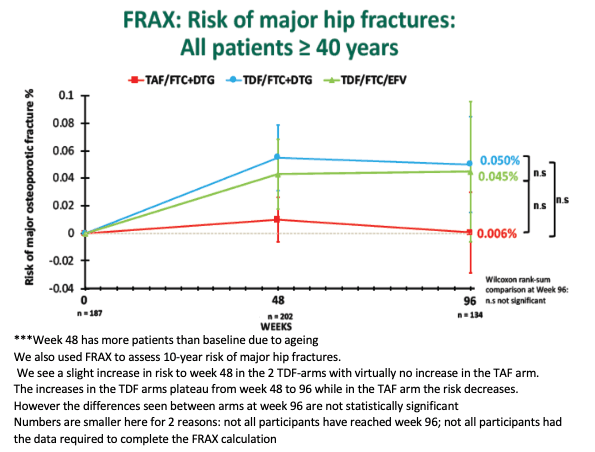
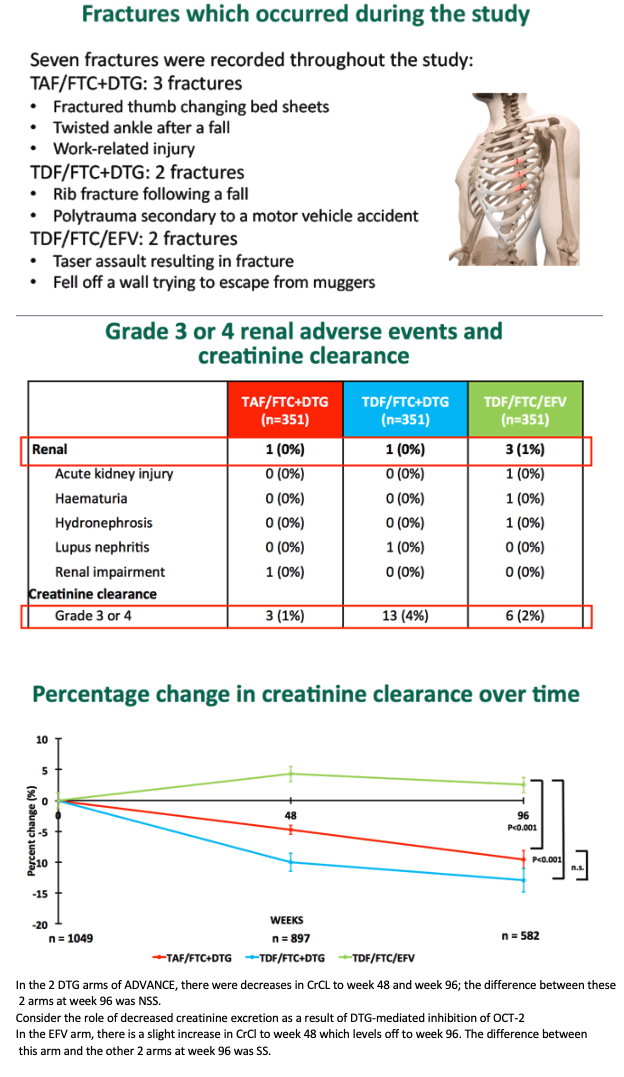
FRAX-determined 10-year risk of major osteoporotic fracture in participants 40 or older averaged 0.36% with TDF/FTC+DTG, a rate significantly greater than with TAF/FTC+DTG (0.17%, P < 0.05) or TDF/FTC+EFV (0.12%, P < 0.05). But the three treatment arms did not differ significantly in risk of major hip fractures (all 0.05% or less). Seven fractures occurred during the study, none related to study drugs.
Through 96 weeks grade 3 or 4 creatinine clearance affected 1% randomized to TAF/FTC+DTG, 4% randomized to TDF/FTC+DTG, and 2% randomized to TDF/FTC+EFV. Creatinine clearance fell about 10% on average with TDF/FTC+DTG, about 7% with TAF/FTC+DTG (not significant versus TDF/FTC+DTG), and did not change with TDF/FTC+EFV (P < 0.001 versus either of the DTG arms).
References
1. Qavi A, Moorhouse M, Sokhela S, et al. The ADVANCE trial: the impact of DXA-assessed bone mineral density of TDF/FTC/EFV and TDF/FTC+DTG versus TAF/FTC+DTG South Africa. 17th European AIDS Conference, November 6-9, 2019, Basel. Abstract 4/3.
2. Venter WDF, Moorhouse M, Sokhela S, et al. Dolutegravir plus two different prodrugs of tenofovir to treat HIV. N Engl J Med. 2019;381:803-815.
https://www.nejm.org/doi/full/10.1056/NEJMoa1902824
3. McCann K, Moorhouse M, Sokhela S, et al. The ADVANCE clinical trial: changes from baseline to week 96 in DXA-assessed body composition in TAF/FTC+DTG compared to TDF/FTC+DTG, and TDF/FTC/EFV. 17th European AIDS Conference, November 6-9, 2019, Basel. Abstract 3/3.
EACS: The ADVANCE clinical trial: changes from baseline to week 96 in DXA-assessed body composition in TAF/FTC+DTG compared to TDF/FTC+DTG, and TDF/FTC/EFV Weight Keeps Rising Through 96 Weeks With TAF/FTC+DTG in Black Africans - (11/12/19)
Editorial: Is modern antiretroviral therapy causing weight gain? - (11/12/19) Sara H. Bares Oct 2019
"Weight Gain Following Initiation of Antiretroviral Therapy: Risk Factors in Randomized Comparative Clinical Trials". Sax et al.
EACS: Are new antiretroviral treatments increasing the risks of obesity? - (11/08/19)





|
| |
|
 |
 |
|
|