 |
 |
 |
| |
Tenofovir treatment has lower risk of hepatocellular carcinoma
than entecavir treatment in patients with chronic hepatitis B
|
| |
| |
Reported by Jules Levin
EASL 2019 April 10-14 Vienna (home of gelato)
Terry Cheuk-Fung Yip1,2, Vincent Wai-Sun Wong1,2,3, Yee-Kit Tse1,2,
Henry Lik-Yuen Chan1,2,3, Grace Lai-Hung Wong1,2,3
1Institute of Digestive Disease,
2Department of Medicine and Therapeutics, and
3State Key Laboratory of Digestive Disease,
The Chinese University of Hong Kong
Download the PDF here
Download the PDF here

--------------------------------------------
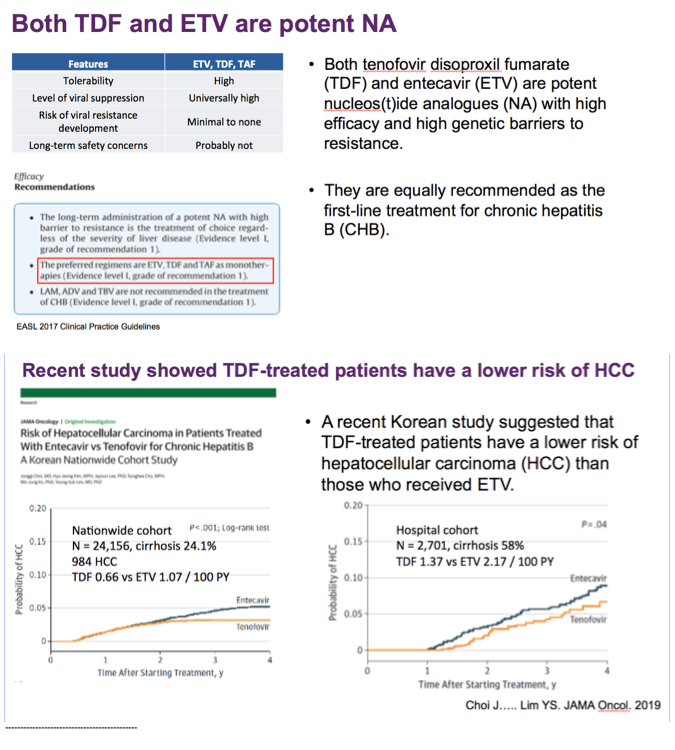
Risk of Hepatocellular Carcinoma in Patients Treated With Entecavir vs Tenofovir for Chronic Hepatitis BA Korean Nationwide Cohort Study
JAMA Oncol. 2019
Question Are entecavir and tenofovir disoproxil fumarate, the first-line antiviral agents for chronic hepatitis B infection, associated with similar risks of hepatocellular carcinoma during long-term treatment?
Findings In a Korean nationwide population cohort of 24 156 treatment-naive adult patients with chronic hepatitis B infection, tenofovir treatment was associated with a significantly lower risk of hepatocellular carcinoma and mortality compared with entecavir treatment. The data were validated in a hospital cohort of 2701 treatment-naive adult patients with chronic hepatitis B infection.
Meaning Given the poor prognosis of patients who developed hepatocellular carcinoma, this study's findings may have considerable clinical implications in the prevention of this cancer in patients with chronic hepatitis B infection.
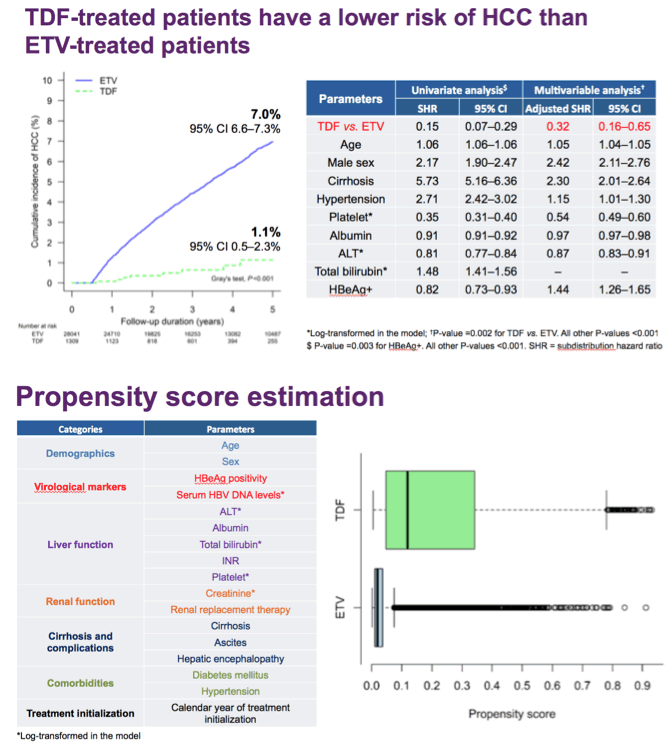
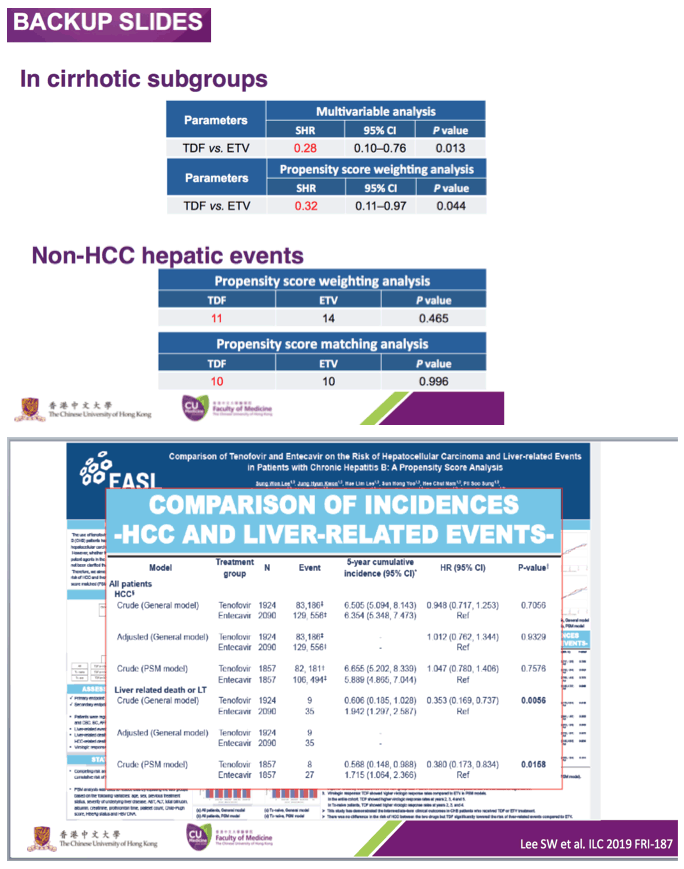
Results Among the population cohort of 24 156, the mean (SD) age was 48.9 (9.8) years, and 15 120 patients (62.6%) were male. Among the hospital cohort of 2701, the mean (SD) age was 48.8 (10.5) years and 1657 patients (61.3%) were male. In the population cohort, the annual incidence rate of HCC was significantly lower in the tenofovir group (0.64 per 100 person-years [PY]) than in the entecavir group (1.06 per 100 PY). By multivariable-adjusted analysis, tenofovir therapy was associated with a significantly lower risk of HCC (hazard ratio [HR], 0.61; 95% CI, 0.54-0.70) and all-cause mortality or transplant (HR, 0.77; 95% CI, 0.65-0.92) compared with entecavir. The tenofovir group also showed a significantly lower risk of HCC in the 10 923-pair propensity score-matched population cohort (HR, 0.62; 95% CI, 0.54-0.70) and 869-pair propensity score-matched hospital cohort (HR, 0.68; 95% CI, 0.46-0.99) compared with the entecavir group.
The mechanism of our main finding on the association of tenofovir with a significantly lower risk of HCC compared with entecavir might be explained in part by the better VR profiles of the tenofovir group as shown in the hospital cohort, which are in line with the results of previous studies.16-18 However, considering that VR was not an independent risk factor for HCC, the differences in HCC risk between the 2 treatments cannot be fully explained by their antiviral potency. A recent study showed that higher serum interferon λ3 levels were induced in patients treated with nucleotide analogues (adefovir dipivoxil and tenofovir), but not in those treated with nucleoside analogues (lamivudine and entecavir).19 Interferon lambda showed potent antitumor activity in murine models of cancer, including hepatoma,20,21 and this antitumor activity could presumably contribute to the difference in the HCC risk that we observed.
Entecavir was shown to be carcinogenic in mice and rats when administered at doses higher than those used in humans.22 It was also shown to potentially incorporate into the human genome, which may contribute to a putative mechanism of carcinogenicity, especially if the embedded genomes have higher error rates during subsequent rounds of replication.23-25 These data raise concern about the carcinogenic potential of entecavir even at clinical doses during long-term treatment, especially in patients with cirrhosis who have increased chromosomal instability of hepatocytes.26,27
Nevertheless, given that HBV replication itself is a strong independent risk factor for HCC, assessing the carcinogenic potential of entecavir should be balanced by considering its protective role against HCC development via suppression of HBV replication. Many observational studies and meta-analyses have shown that in patients with CHB, entecavir therapy significantly reduced HCC risk compared with no treatment.18,22,28 However, a previous cohort study demonstrated that HCC risk with entecavir was not lower than that with lamivudine.7 Nonetheless, the effect of entecavir on HCC risk would be better compared with tenofovir because both drugs have similar potencies in suppressing HBV replication.16,18,28 Based on our data, 185 HCC cases were estimated to have been preventable at 4 years' follow-up if the 11 464 patients in the entecavir group had used tenofovir (eTable 15 in the Supplement).
Pdfs attached above
Editorial
January 2019 JAMA Oncol. 2019
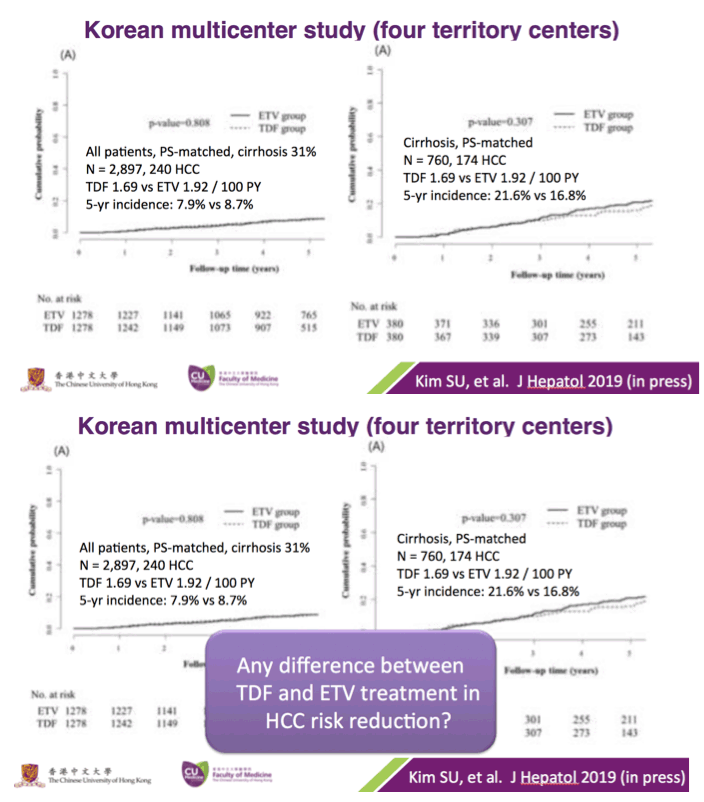
Current guidelines for the treatment of chronic hepatitis B (CHB) recommend as first-line line therapy entecavir (ETV), tenofovir disoproxil fumarate (TDF), or tenofovir alafenamide fumarate.1-3 Both ETV and TDF achieve similar rates of hepatitis B virus (HBV) DNA suppression and alanine aminotransferase normalization over time and have an excellent safety record. However, ETV and TDF have not been directly compared except in patients with decompensated cirrhosis4; to our knowledge, no other head-to-head randomized trials exist. Yet, there is strong interest in understanding if one drug might be superior to the other in specific clinical settings or subpopulations. In the absence of randomized comparator trials, high-quality "real world" cohorts can be quite informative. In this issue of JAMA Oncology, Choi and colleagues5 report the results of 2 retrospective cohort studies of patients with CHB from Korea that evaluated the association between the type of nucleoside or nucleotide analogue and the development of hepatocellular carcinoma (HCC).5 Using both a large administrative data set (24 156 patients with CHB and 984 with HCC) and a tertiary hospital-based cohort (2701 patients with CHB and 154 with HCC), the authors found that the risk of HCC was consistently about 35% lower in those treated with TDF than in those treated with ETV. This association persisted even when stratified by cirrhosis status and after very thorough adjustment for available confounders. These striking findings raise the question of whether the current CHB guidelines should be updated to reflect this observed superior anticancer benefit of TDF over ETV.
Multiple studies have shown that treatment with either ETV or TDF reduces the incidence of HCC in patients with CHB when compared with untreated patients.6,7 Prior studies examining the use of ETV vs TDF, though limited in numbers, did not find a difference between these 2 first-line agents.8,9 Based on the observed adjusted hazard ratios in the study by Choi and colleagues,5 these other cohort studies would have needed more than 200 cases of HCC to have adequate power to detect a 35% reduction in HCC risk. Therefore, since the largest known study had only 56 incident cases of HCC,8 these earlier studies may have been underpowered to detect a difference in HCC rates by antiviral drug.
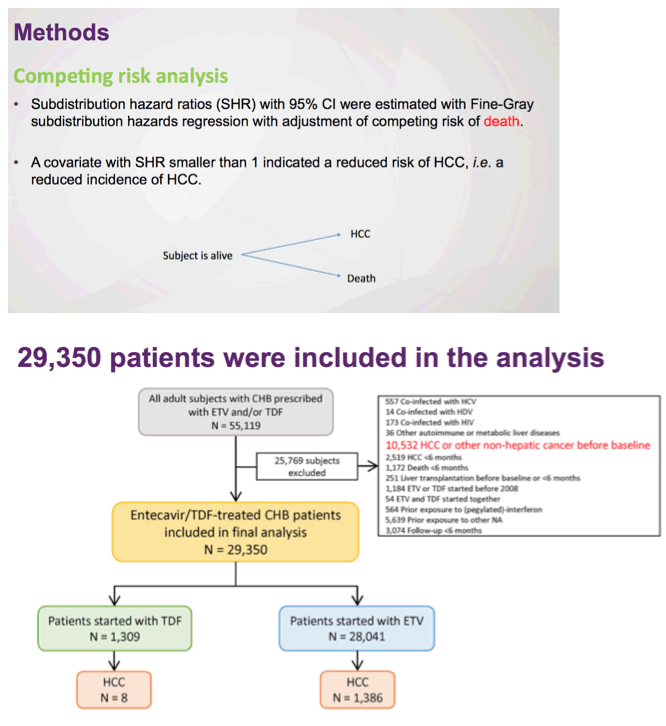
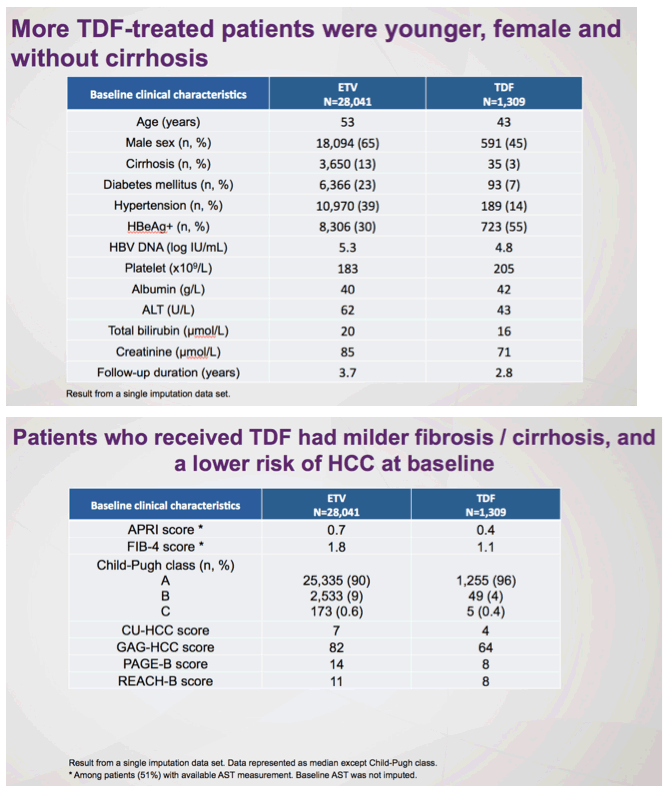
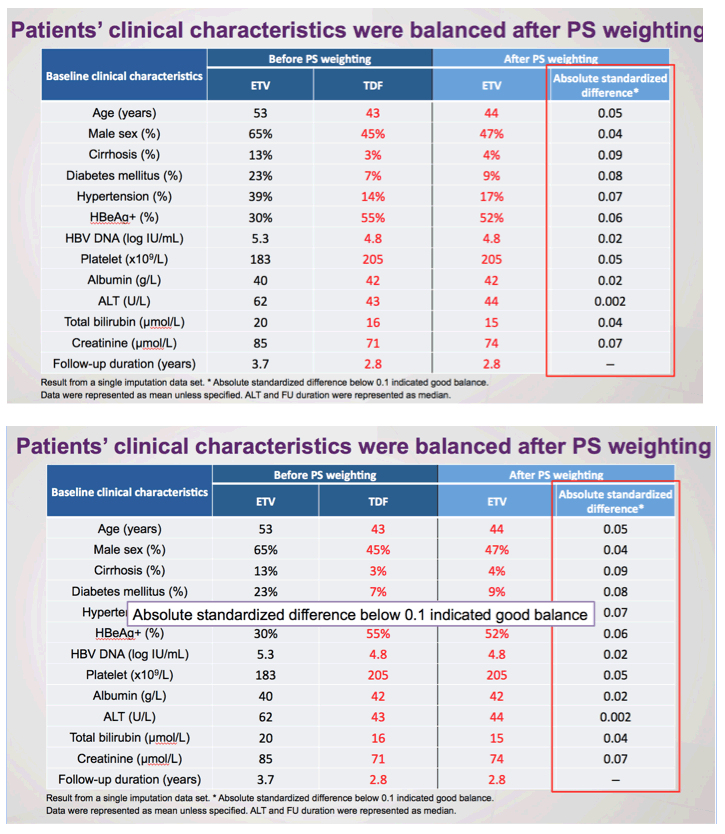
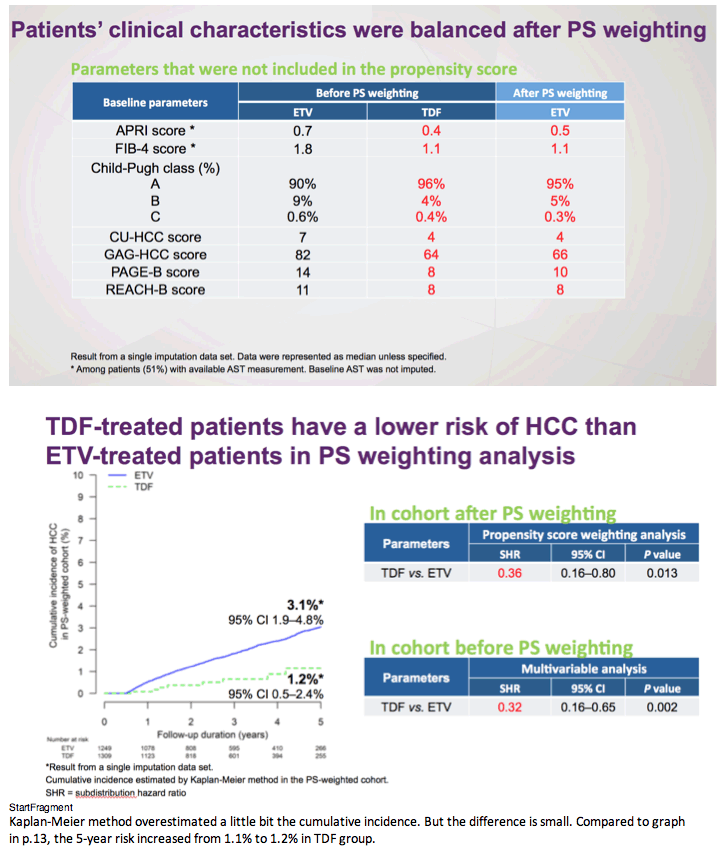
There remains a concern that the observed difference in HCC rates is due to residual confounding. This is a limitation of all observational studies, but Choi and colleagues5 used a robust analytic approach that included multiple subgroup analyses, the use of propensity scores and competing risks, as well as validation in a hospital-based cohort having data on disease severity and virologic responses. Remarkably, the association of ETV vs TDF and HCC risk was minimally changed by any of these adjustments. However, adherence to antiviral therapy and surveillance programs was not captured and may have influenced rates of HCC. Indeed, adherence of less than 90% to antiviral therapy in patients with CHB has been associated with a higher risk of HCC.10 Moreover, especially in countries where prior lamivudine exposure is prevalent, patients treated with ETV may be at increased risk of virologic breakthrough with time, and this, in turn, affects risk for HCC. Indeed, in the study by Choi and colleagues, there was a substantially higher rate of changing therapy in patients treated with ETV (12%) vs TDF (0.2%). Might reduced adherence and increasing rates of ETV resistance represent unmeasured confounders that contribute to the divergence in rates of HCC evident 2 years after initiation of therapy? Ideally, a randomized study design would help overcome these challenges and provide stronger evidence of the association between type of antiviral agent and HCC outcomes. As such a study seems unlikely, at least in the near term, we would advocate for additional observational cohorts with data on liver disease severity, virologic response, and adherence to surveillance to evaluate this important question.
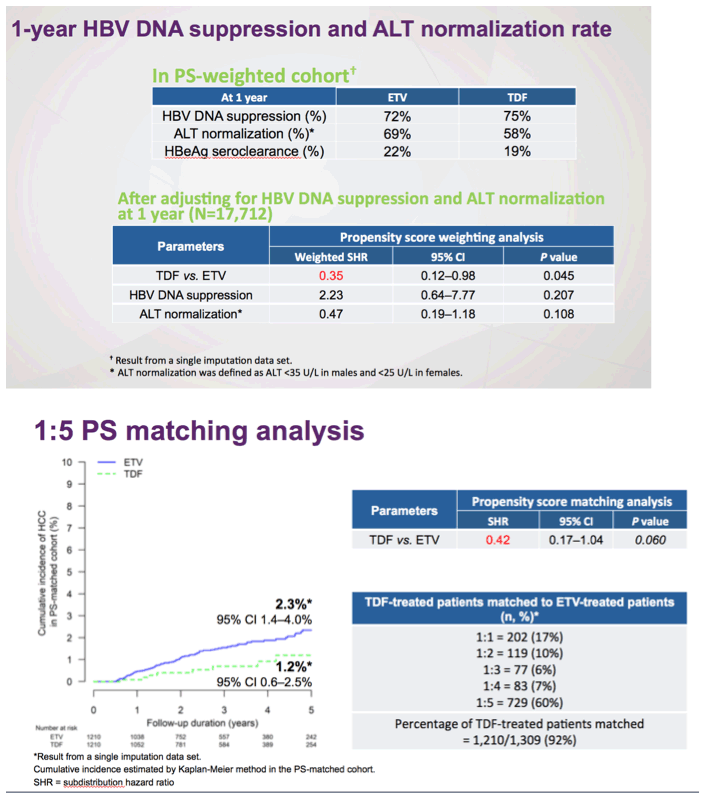
In considering HBV therapy as prevention of HCC, a critical question remains. How important is the achievement and maintenance of undetectable HBV DNA during treatment? Current HBV guidelines do not recommend changing or adding another antiviral agent in patients with low-level intermittent or persistent viremia during treatment with first-line agents, as long as criteria for virologic breakthrough are not met. In terms of HCC risk, natural history studies show that HCC risk is highest among those with HBV DNA levels of 200 000 IU/mL or higher but not different from uninfected controls if HBV DNA levels are lower than 2000 IU/mL.11 More recent studies of patients undergoing long-term antiviral therapy provide conflicting results on the importance of low-level viremia and HCC risk. In a Korean study of patients with CHB and cirrhosis on ETV therapy for up to 5 years, rates of HCC were 2.2-fold higher in patients with persistent or intermittent HBV DNA viral loads less than 2000 IU/mL compared with those with persistently undetectable HBV DNA (<12 IU/mL), though this did not hold true for those without cirrhosis.12 In a study of patients with decompensated cirrhosis, maintained virologic response (<20 IU/mL) vs suboptimal responses (<2000 but >20 IU/mL) influenced transplant-free survival but not HCC risk over 10 years.13 These studies highlight the importance of cirrhosis as a primary driver of HCC risk, independent of effective therapy.12,13 However, for those without cirrhosis, which represent the majority of patients with CHB, the level of viremia may be relevant to treatment decisions. While the study by Choi et al5 includes information on virologic response in the first year with the validation cohort, the absence of longitudinal virologic response over the duration of follow-up is a shortcoming of the larger observational cohort, and could be the major explanatory variable for the differences in HCC outcomes in the ETV vs TDF groups. Collectively, recent studies suggest that viral suppression is important, and thus selecting an antiviral agent that achieves and maintains suppression is highly desirable, but the findings do not dictate that one drug should be preferred over the other.
Finally, if TDF is superior to ETV in reducing HCC risk, the big question is why. Choi and colleagues5 offer 2 potential explanations, neither of which is well supported by human data. They suggest that ETV may be procarcinogenic and/or that nucleotide analogues (vs nucleoside analogues) may induce higher antitumor cytokines. That viral clearance may induce an altered intrahepatic milieu that influences risk of hepatocarcinogenesis is possible, though the rapidity of viral and antigen decline may be more important than the specific drug type (nucleoside vs nucleotide) used. The hypothesis that ETV may be procarcinogenic is based on preclinical studies in mice and rats treated with high-dose ETV; there are no human correlative data to support this. Indeed, if this were the case, one would have expected to see increasing rates of HCC with increasing duration of drug exposure; a feature not seen in this cohort or others.5,6,14 The study by Choi et al with just over 4 years of follow-up data does not provide a yearly HCC incidence, but visual inspection of the survival curves in ETV-treated patients suggests that the rate of incident HCC is not increasing. Another Korean cohort of almost 2000 patients treated with ETV showed that the annual HCC incidence in the first 5 years of treatment was similar to the rate after 5 years of ETV exposure (2.29% vs 1.66%, P = .22).14 Additionally, in a cohort of over 1900 white patients with CHB undergoing long-term treatment with either ETV or TDF with a median follow-up of 6 years, HCC incidence was similar beyond 5 years in those without cirrhosis, while there was a significant decrease in HCC incidence after 5 years of therapy in those with cirrhosis at baseline.6 Thus, there is no clinical support for a procarcinogenic tendency with ETV, and the biologic underpinnings of the observed difference in HCC rates between ETV and TDF remain obscure.
Three safe and effective oral antivirals are endorsed by experts as first-line therapy for CHB: ETV, TDF, and tenofovir alafenamide fumarate.1,2 These antiviral agents share the qualities of high efficacy in reducing HBV DNA levels and excellent tolerability and safety. In deciding on the best option for a given patient with CHB, the clinician should consider the treatment history, comorbidities, including renal disease or human immunodeficiency virus, and cost and accessibility. Prevention of liver-related complications requires adherence to the treatment regimen and a therapy achieving viral load suppression. The work of Choi and colleagues5 is the first to our knowledge to suggest that TDF offers advantages over ETV in terms of HCC prevention, but given the inherent limitations of observational data, this study should not lead to a widespread paradigm shift in selecting TDF over ETV. Rather, it should spur more investigators to address the issue of whether one of these HBV drugs is not like the other.
--------------------------------------------




|
| |
|
 |
 |
|
|