 |
 |
 |
| |
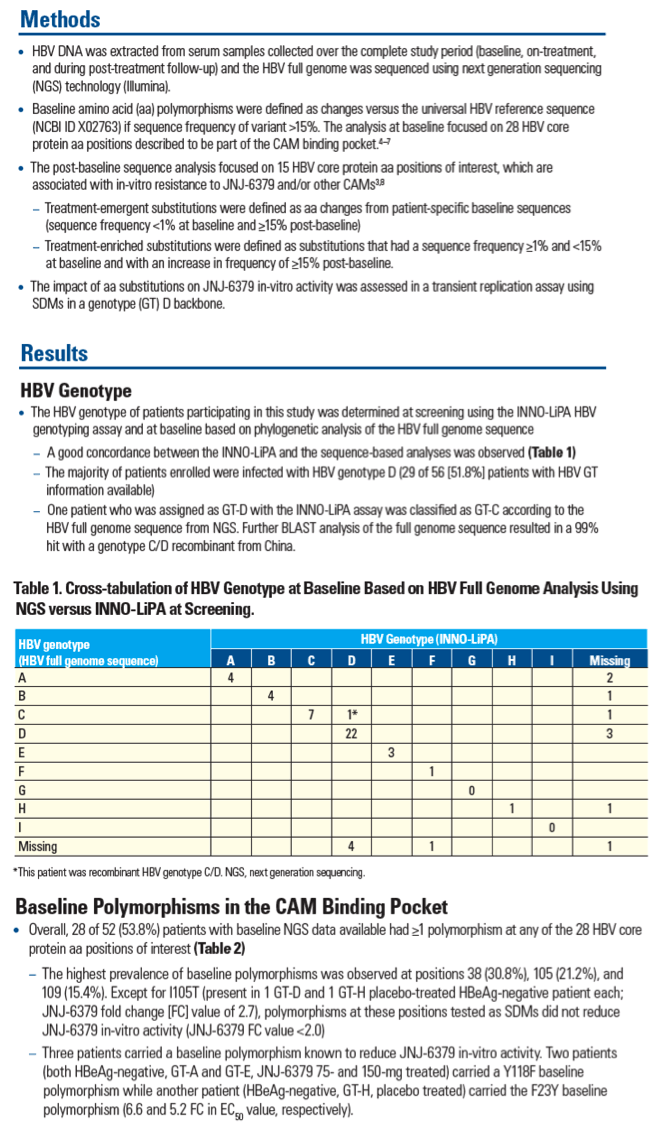
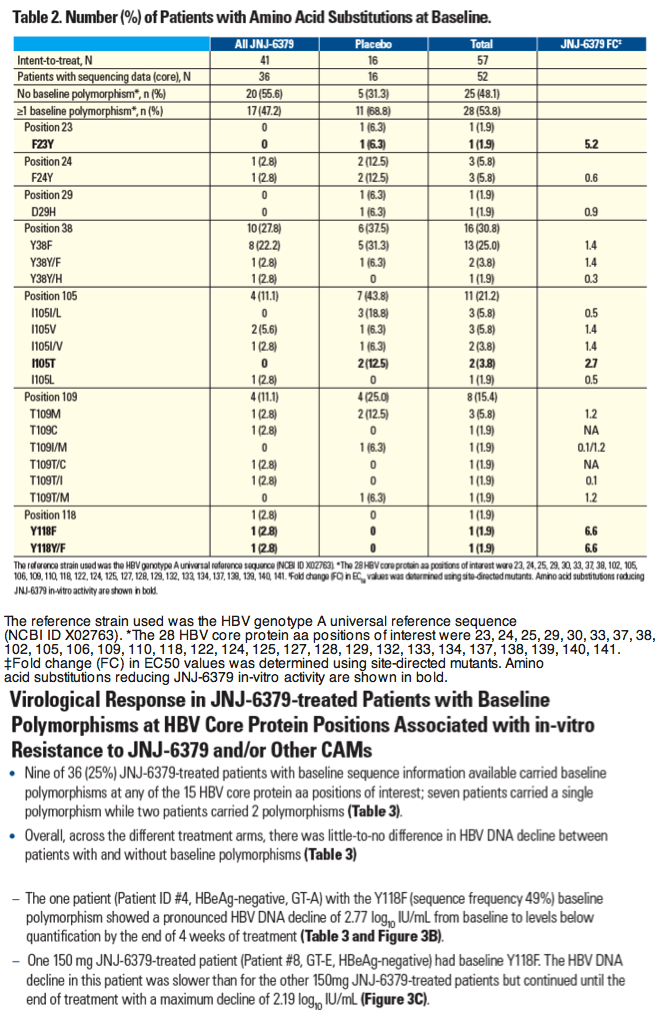
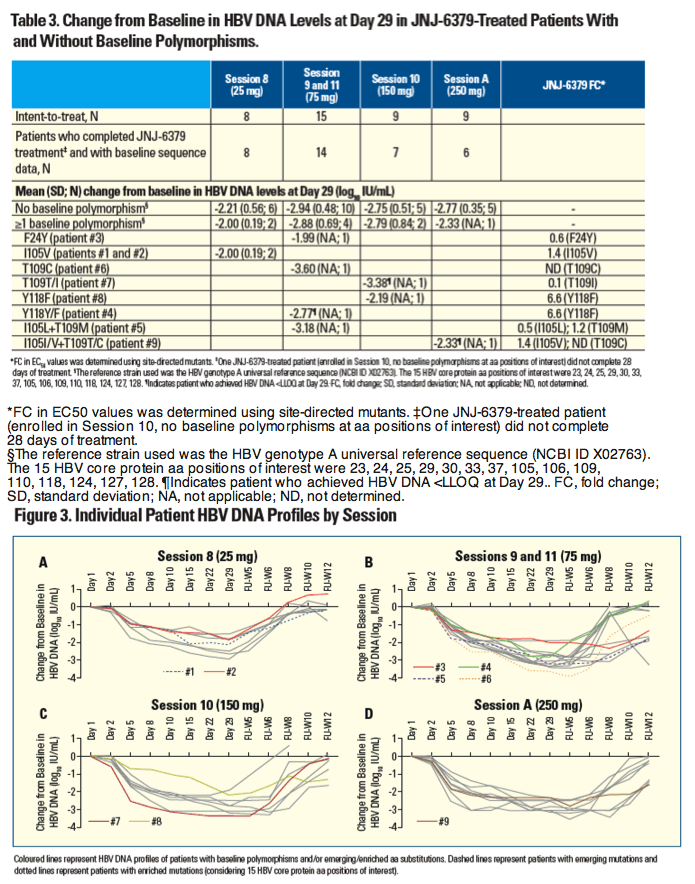
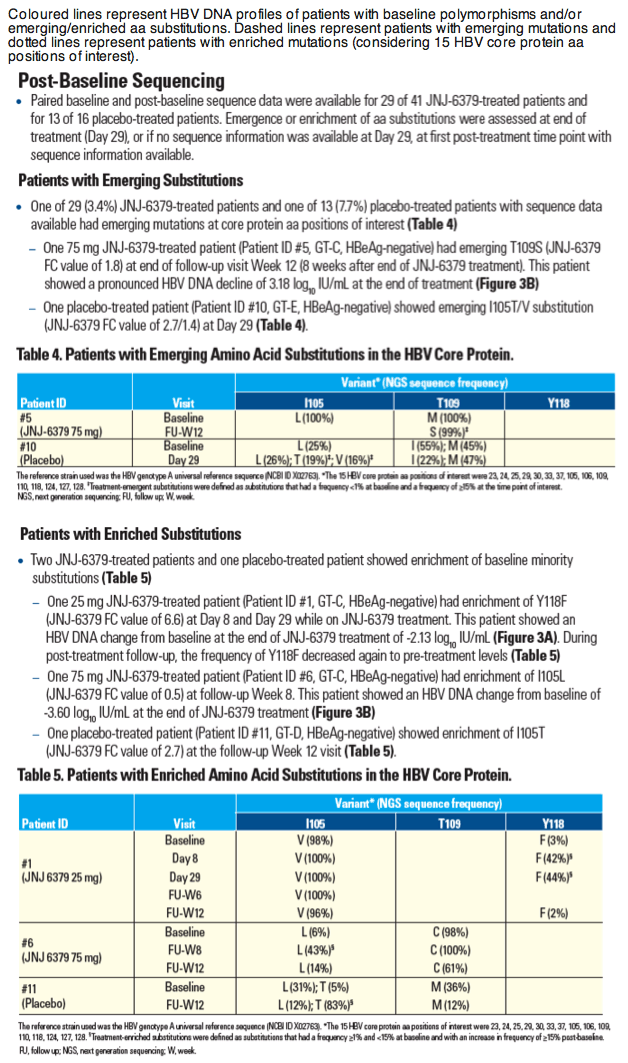
Sequence Analysis of Baseline and On-treatment Samples from HBV-infected Chronic Hepatitis B Patients Treated for 28 Days with JNJ-56136379 Monotherapy
|
| |
| |
Reported By Jules Levin
EASL The International Liver Congress, Vienna, Austria, April 10-14, 2019
Thierry Verbinnen1, Moana Hodari1, Willem Talloen1, Jan Martin Berke1, David Blue2, Jeysen Yogaratnam2, Umesh Shukla3, Sandra De Meyer1, Oliver Lenz1
1Janssen Research & Development, Beerse, Belgium; 2Janssen Biopharma Inc., South San Francisco, CA, USA; 3Janssen Pharmaceuticals Research & Development, Titusville, USA

|
| |
|
 |
 |
|
|