 |
 |
 |
| |
A Phase 1, Double-Blind, Randomised, Placebo-Controlled, First-in-Human Study of the Safety, Tolerability, Pharmacokinetics and
Pharmacodynamics of oral JNJ-64794964, a Toll-like Receptor-7 Agonist, in Healthy Adults
|
| |
| |
Reported by Jules Levin
EASL The International Liver Congress, Vienna, Austria, April 10-14, 2019
EASL: Antiviral activity of JNJ-4964 (AL-034/TQ-A3334),a selective Toll-Like Receptor 7 agonist, after oral administration for 12 weeks in AAV/HBV mice - (04/19/19)
EASL: Short term RNA interference (RNAi) therapy in chronic hepatitis B (CHB) using JNJ-3989 brings majority of patients to HBsAg <100 IU/ml threshold - (04/19/19)
Ed Gane1, Mina Pastagia2, An De Creus3, Christian Schwabe4, Joris Vandenbossche3, Leen Slaets3, Bart Fevery3, Ilham Smyej3, Liviawati S Wu2, Rui Li5, Samia Siddiqui5, Abbie Oey2, Clark Musto2, Bruce Bryan2, Pieter Van Remoortere5
1New Zealand Liver Transplant Unit, University of Auckland, Auckland, New Zealand; 2Janssen BioPharma Inc., South San Francisco, CA, USA; 3Janssen Research & Development, Janssen Pharmaceutica NV, Beerse, Belgium; 4Auckland Clinical Studies, Auckland, New Zealand; 5Janssen Research & Development LLC, Titusville, NJ, USA


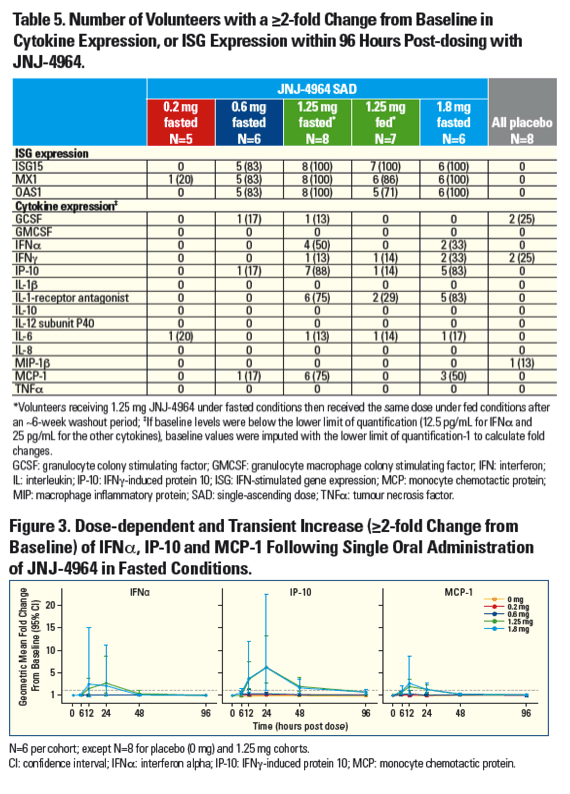
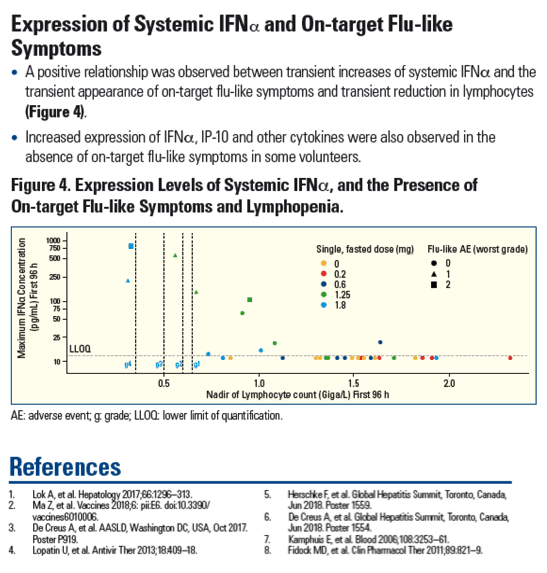
|
| |
|
 |
 |
|
|