 |
 |
 |
| |
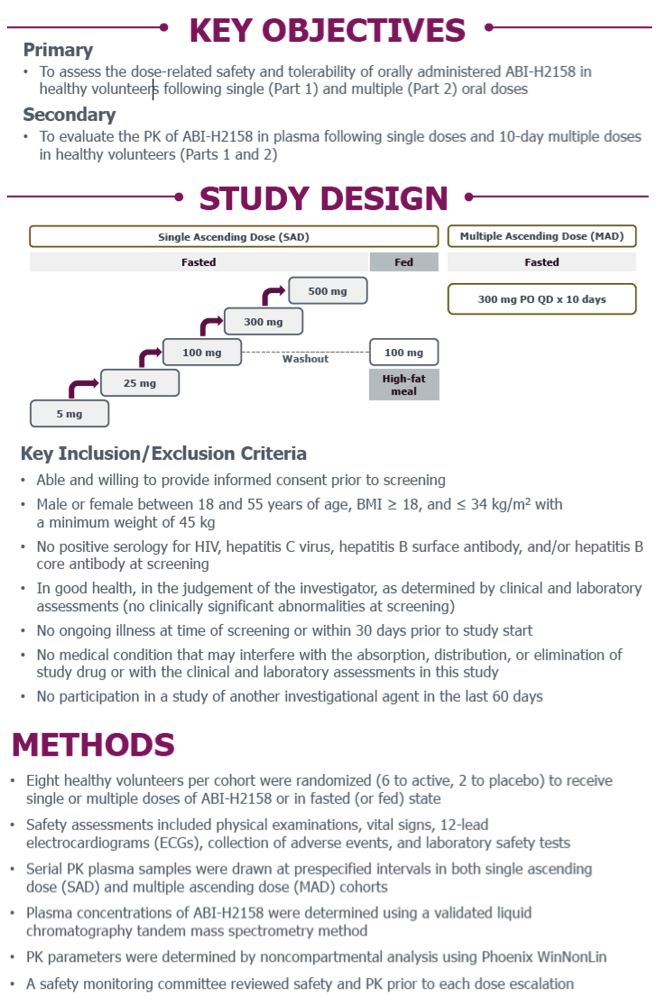
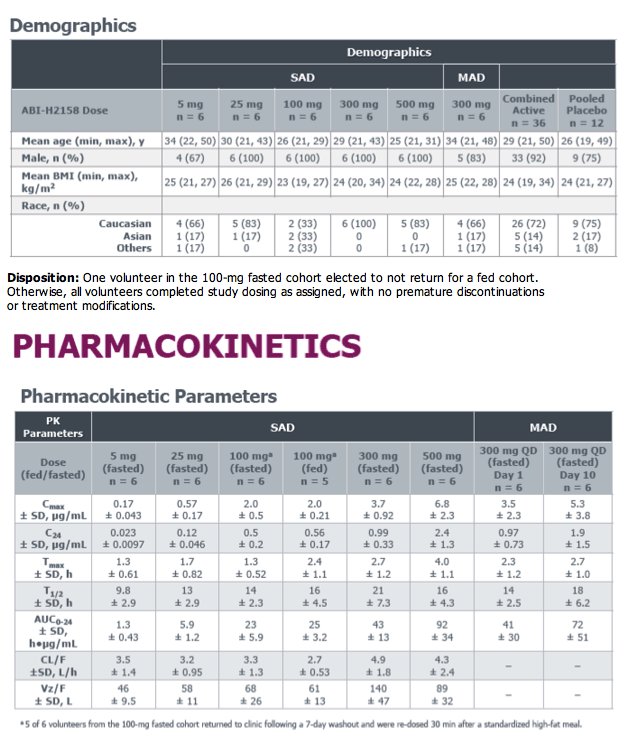
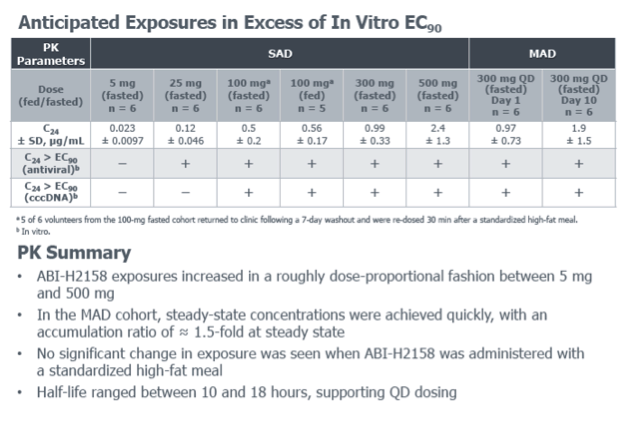
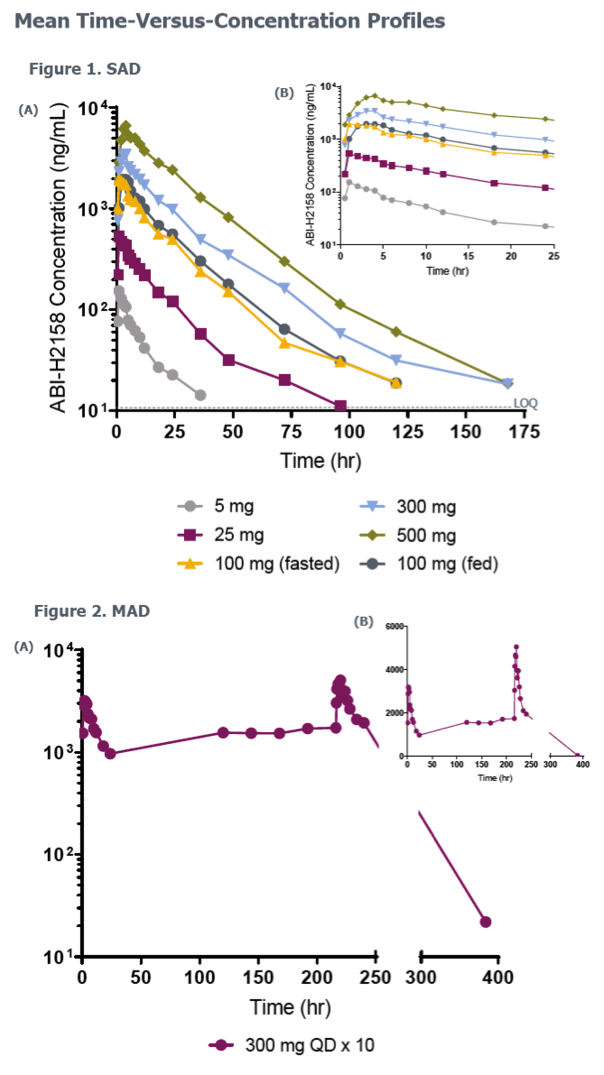
Phase 1a Study of the Safety, Tolerability and Pharmacokinetics of ABI-H2158, a Novel Second-Generation HBV Core Inhibitor, In Healthy Volunteers
|
| |
| |
Reported by Jules Levin
EASL 2019 April 10-14 Vienna
E.J. Gane,1C. Schwabe,1M. Evanchik,2E. Ruby,2R. Colonno,2K. Alves,2S. Liaw,2and U. Lopatin2
1Clinical Trial Unit, Auckland Clinical Studies, Auckland, New Zealand; 2Assembly Biosciences, Inc., San Francisco, California, USA
Download the PDF here

|
| |
|
 |
 |
|
|