| |
Declining hepatitis C virus-related liver disease burden in the
direct-acting antiviral therapy era in New South Wales, Australia
|
| |
| |
Download the PDF here
The Australian Government has provided unrestricted access to subsidized DAA therapy for adults with chronic HCV infection since March 2016. The program incorporates a broad prescriber population, including general practitioners, and has no restrictions based on liver disease stage, or drug and alcohol use. In Australia, around 54,110 patients were treated over the period March 2016 to December 2017, equivalent to 24% of the estimated population with chronic HCV infection (B. Hajarizadeh, personal communication, February 2019). Importantly, in relation to potential prevention of advanced liver disease complications, an estimated 70% of Australian people with HCV-related cirrhosis had received DAA therapy.11 DAA uptake in New South Wales, where around one-third of the Australian population reside, is consistent with the overall Australian uptake; approximately 19,200 people were treated in 2016-2017, equivalent to 24% of the estimated population with chronic HCV infection (B. Hajarizadeh, personal communication, February 2019).
from Jules: the USA rands lat in the world in on track for HCV elimination, only about 40$ mill is provided to CDC by Congress:
http://www.natap.org/2019/EASL/EASL_24.htm
http://www.natap.org/2019/AASLDEASL/AASLDEASL_05.htm
Discussion
Our study supports a major population-level impact of DAA therapy on HCV-related liver disease morbidity and mortality, and all-cause mortality. Over the decade prior to the introduction of DAA therapy in Australia, the numbers of people with HCV in New South Wales hospitalised for decompensated cirrhosis, HCC, or who died following these end-stage liver disease complications or all-cause mortality increased by 2- to 3-fold. Between 2015 and 2017, declines of 21% and 17% in decompensated cirrhosis diagnoses and liver-related deaths, respectively, and an 8% and 3% increase in HCC diagnoses and all-cause mortality (vs. an expected 34% and 19% increase without DAA therapy introduction, respectively) clearly demonstrate the impact of DAA therapy on advanced liver disease morbidity and mortality, and all-cause mortality. The decline in liver-related deaths is considerably more marked when compared to the expected continued upward trend from the interferon-based therapy era. The lower relative declines in liver-related morbidity and mortality among people with a history of alcohol-use disorder, and the increasing proportion of people with end-stage liver disease with a history of alcohol-use disorder, highlights the need to enhance efforts to reduce the impact of this major liver disease comorbidity.
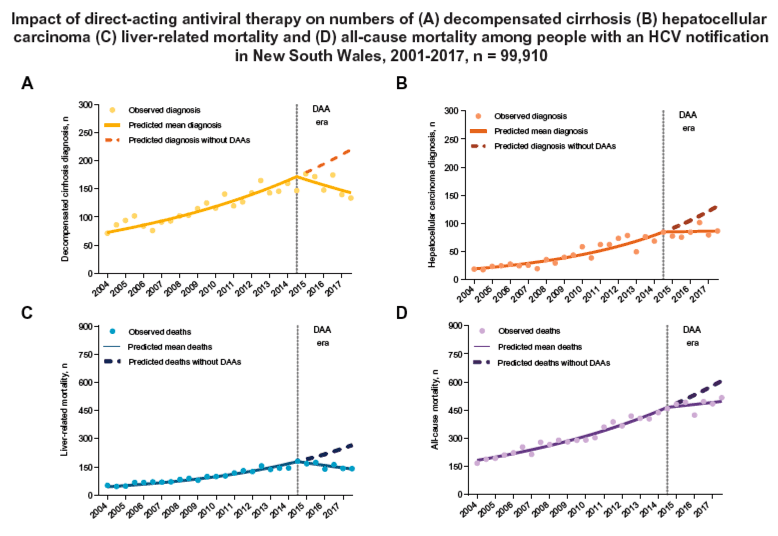
The Government-funded DAA program has transformed HCV clinical management in Australia, with HCV treatment increasing from 1-2% per year in 2004-2015 to 24% over the 2016-2017 period, and cure rates rising to above 95%.11 Major changes in HCV models of care have also been implemented, with a large proportion of people with chronic HCV now treated in primary care.11 Although there have been no liver disease-based restrictions on DAA therapy access, there clearly has been some prioritisation, by patients, clinicians, or both, for treatment of those with more advanced liver disease, with an estimated 70% of people with HCV-related cirrhosis having received treatment by the end of 2017.11 The Australian Government-funded DAA program commenced in March 2016. There was, however, DAA access for people with cirrhosis from late 2014 through 2015, via pharmaceutical company access programs, DAA clinical trials, and generic importation, providing treatment to 4,340 people in Australia, including 1,200 people in New South Wales.16 Overall, an estimated 7,045 people with HCV-related cirrhosis in New South Wales received DAA therapy between 2015 through 2017 (B. Hajarizadeh, personal communication, February 2019). The estimated 434 liver-related deaths prevented in New South Wales in 2015-2017 could be largely attributed to treatment of this advanced liver disease population.
Jnl. of Hepatology Aug 2019 - Maryam Alavi1,⇑, Matthew G. Law1, Heather Valerio1, Jason Grebely1, Janaki Amin2,
Behzad Hajarizadeh1, Christine Selvey3, Jacob George4, Gregory J. Dore1
1The Kirby Institute, UNSW Sydney, Sydney, NSW, Australia; 2Department of Health Systems and Populations, Macquarie University,
Sydney, NSW, Australia; 3Communicable Diseases Branch, Health Protection NSW, Sydney, NSW, Australia; 4Storr Liver Centre, Westmead Millennium Institute, University of Sydney and Westmead Hospital, Westmead, NSW, Australia
Highlights
• Prior to the introduction of DAA therapy, the disease burden of HCV was rising in New South Wales.
• DAA scale-up has had a major population-level impact on HCV morbidity and mortality.
• The World Health Organization has set a 65% HCV mortality reduction target by 2030.
• To achieve this target, enhanced efforts are required to continue DAA scale-up.
• In the DAA era, the impact of heavy alcohol use on liver disease should be monitored.
Background & Aims
Population-level evidence for the impact of direct-acting antiviral (DAA) therapy on hepatitis C virus (HCV)-related disease burden is lacking. We aimed to evaluate trends in HCV-related decompensated cirrhosis and hepatocellular carcinoma (HCC) hospitalisation, and liver-related and all-cause mortality in the pre-DAA (2001-2014) and DAA therapy (2015-2017) eras in New South Wales, Australia.
Methods
HCV notifications (1993-2016) were linked to hospital admissions (2001-2017) and mortality (1995-2017). Segmented Poisson regressions and Poisson regression were used to assess the impact of DAA era and factors associated with liver-related mortality, respectively.
Results
Among 99,910 people with an HCV notification, 3.8% had a decompensated cirrhosis diagnosis and 1.8% had an HCC diagnosis, while 3.3% and 10.5% died of liver-related and all-cause mortality, respectively. In the pre-DAA era, the number of decompensated cirrhosis and HCC diagnoses, and liver-related and all-cause mortality consistently increased (incidence rate ratios 1.04 [95% CI 1.04-1.05], 1.08 [95% CI 1.07-1.08], 1.07 [95% CI 1.06-1.07], and 1.05 [95% CI 1.04-1.05], respectively) over each 6-monthly band. In the DAA era, decompensated cirrhosis diagnosis and liver-related mortality numbers declined (incidence rate ratios 0.97 [95% CI 0.95-0.99] and 0.96 [95% CI 0.94-0.98], respectively), and HCC diagnosis and all-cause mortality numbers plateaued (incidence rate ratio 1.00 [95% CI 0.97-1.03] and 1.01 [95% CI 1.00-1.02], respectively) over each 6-monthly band. In the DAA era, alcohol-use disorder (AUD) was common in patients diagnosed with decompensated cirrhosis and HCC (65% and 46% had a history of AUD, respectively). AUD was independently associated with liver-related mortality (incidence rate ratio 3.35; 95% CI 3.14-3.58).
Conclusions
In the DAA era, there has been a sharp decline in liver disease morbidity and mortality in New South Wales, Australia. AUD remains a major contributor to HCV-related liver disease burden, highlighting the need to address comorbidities.
Lay summary
Rising hepatitis C-related morbidity and mortality is a major public health issue. However, development of highly effective medicines against hepatitis C (called direct-acting antivirals or DAAs) means hepatitis C could be eliminated as a public health threat by 2030. This study shows a sharp decline in liver disease morbidity and mortality since the introduction of DAAs in New South Wales, Australia. Despite this, heavy alcohol use remains an important risk factor for liver disease among people with hepatitis C. To ensure that the benefits of new antiviral treatments are not compromised, management of major comorbidities, including heavy alcohol use must improve among people with hepatitis C.
| |
| |
| |
|
|
|