| |
Suboptimal SVR rates in African patients with atypical
Genotype 1 subtypes: implications for global elimination of Hepatitis C
|
| |
| |
Download the PDF here
This was presented at EASL 2019, here is link to slides and just published
• The evidence indicates that the future desired expansion of HCV treatment in Africa may risk unacceptable rates of failure if first generation NS5A inhibitors are utilised without appropriate epidemiological and viral sequence data. Global equity of access to curative treatment is required to avoid jeopardising the hepatitis C elimination agenda.
• In this analysis we report the prevalence of HCV genotypes in a consecutive cohort of 91 African patients from 18 countries, and documents the outcome of DAA therapy in patients with subtypes of genotype 1 and 4 that are prevalent in Africa but unusual in the West. Our study has three main findings. Firstly, unusual and previously undescribed subtypes were common. Secondly, there was a high degree of viral diversity and thirdly there was a lower SVR rate in those with unusual African subtypes, particularly in those who received a first generation NS5A inhibitor.
• Five patients who failed initial treatment have thus far been commenced on retreatment: Three of these patients have completed 16 weeks of glecaprevir and pibrentasvir, two of whom have achieved an SVR. One individual with cirrhosis and G1* HCV virus failed to achieve an SVR with 16 weeks of glecaprevir and pibrentasvir. Two other patients were retreated with sofosbuvir, -velpatasvir and voxilaprevir and achieved an SVR.
NATAP/EASL Unacceptably low SVR rates in African patients with unusual HCV sub-genotypes: Implications for global elimination - (04/18/19)
Article in Press
Suboptimal SVR rates in African patients with atypical Genotype 1 subtypes: implications for global elimination of Hepatitis C
Jnl of Hepatology Aug 7 2019
Kate Childs, Christopher Davis, Mary Cannon, Sarah Montague, Ana Filipe, Lily Tong, Peter Simmonds, Donald Smith, Emma C. Thomson, Geoff Dusheiko, Kosh Agarwal
• An SVR was observed in only 21 of the 28 (75%) patients infected with unusual African genotype 1.
• Five patients who failed initial treatment have thus far been commenced on retreatment: Three of these patients have completed 16 weeks of glecaprevir and pibrentasvir, two of whom have achieved an SVR. One individual with cirrhosis and G1* HCV virus failed to achieve an SVR with 16 weeks of glecaprevir and pibrentasvir. Two other patients were retreated with sofosbuvir, -velpatasvir and voxilaprevir and achieved an SVR.
• There were numerous NS5A polymorphisms present at baseline in patients with unusual G1 subtypes, particularly at positions 24, 30 and 31. Resistance associated polymorphisms (RASs) listed in the EASL 2018 HCV guidelines as conferring reduced susceptibility to NS5A inhibitors or being associated with reduced treatment response were seen at baseline in 18/22 (82%) patients. All the treatment failures had either M28 polymorphisms (L and S) (2 failures) or L31M (4 failures). Five SVR patients had M28 (L and V), Three patients (2 with 1* and one with 1g) who achieved SVR had Y93 (F/H/N) at baseline. These data are expanded in table 4. The individual patient with 1* who has failed treatment with both sofosbuvir/ledipasvir and then glecaprevir/pibrentasvir had Q62E, L31M at baseline and Q62D, L31M following SOF/LDV. Following glecaprevir/pibrentasvir treatment Q30H, H58S and Y93H were also accumulated.
Highlights
• Unusual genotypes (G1 non 1a/1b or G4 non 4a/4d) were common in African patients.
• 11 previously unclassified HCV subtypes were represented including novel G1p.
• Patients with unusual G1 subtypes had a lower SVR rate than any other genotype.
• Failures were driven by patients treated with a first generation NS5A inhibitor.
• The majority of unusual G1 subtypes had baseline NS5A resistance mutations.
Lay Summary
Direct acting antiviral medications are able to cure Hepatitis C in the majority of patients. The most common genotype of Hepatitis C in Europe and the United States is genotype 1a or 1b and most clinical trials focused on these genotypes. We report that in a group of African patients, most of them had unusual (non-1a/1b) genotype 1 subtypes, and that the cure rate in these unusual genotypes was lower than in genotypes 1a and 1b.
Discussion
In this analysis we report the prevalence of HCV genotypes in a consecutive cohort of 91 African patients from 18 countries, and documents the outcome of DAA therapy in patients with subtypes of genotype 1 and 4 that are prevalent in Africa but unusual in the West. Our study has three main findings. Firstly, unusual and previously undescribed subtypes were common. Secondly, there was a high degree of viral diversity and thirdly there was a lower SVR rate in those with unusual African subtypes, particularly in those who received a first generation NS5A inhibitor.
In this unselected cohort of immigrant African patients, forty seven of 91 (52%) were infected by unusual African subtypes, (i.e. G1 non 1a/1b/ unassigned G1 subtype), or with G4 other than 4a or 4d. The distribution of genotypes was different from the non-African majority of our HCV patient cohort where G1a and 3a were the most common. The finding that unusual subtypes were common amongst African patients mirrors the results from the Los Alamos HCV database which contains 288 sequences of genotype 1c to 1m; 47.4% of these isolates originated in Africa [12]. The fact the majority of catalogued non 1a/b G1 subtypes originated in Africa and that the majority of our cohort were infected with unusual subtypes suggests that these subtypes are more common in Africa than Europe. Since fifteen percent of global cases of HCV are based in the WHO African region, [1] these variants of HCV could be numerically significant. Population based studies are required to establish the true prevalence of these “unusual” subtypes in Africa.
Our second finding was the high diversity of HCV sequences. We were able to sequence 15 previously undescribed subtypes of G1 HCV. One of these sequences was found to be a strain infecting 3 different patients, allowing formal classification as the novel genotype 1p. This is concordant with recent data from a cohort from Uganda and the Democratic Republic of Congo, where highly diverse genotype 4 and 7 strains, including unconfirmed novel strains were recently identified [2]. We can speculate that the diversity of HCV observed in this cohort and other studies, if confirmed by larger phylogenetic analyses, perhaps points to an early evolutionary origin of human hepatitis C genotype 1 and 4 in Africa [13].
We observed a high number of resistance- associated polymorphisms in pre-treatment samples from our cohort. NS5A RASs listed in the 2018 EASL guidelines as conferring reduced susceptibility to NS5A inhibitors, for example at positions 24, 28, 30 and 31 were present in 82% of patients with baseline sequence data [14]. This contrasts with published findings from the pooled analysis of phase 2 and 3 studies of sofosbuvir and ledipasvir; of 2108 patients only 16% patients had detectable baseline RASs [15]. That we saw such high frequencies of polymorphisms further demonstrates the diversity of the virus in the African region.
Our third finding was that SVR rates were markedly lower in this population. Although we have studied a relatively small number of patients, we show that the overall SVR rate for African patients at an experienced centre (89%), is lower than expected in a group of predominantly non-cirrhotic patients. Importantly the SVR rate fell to 21/28 (75%), in those infected with non 1a/b G1 subtypes. The low SVR rate was driven by 6 failures in patients treated with sofosbuvir and ledipasvir, 1 failure with a protease inhibitor-based regimen and was associated with multiple baseline polymorphisms.
There is limited data available on treatment outcome in these uncommonly encountered genotypes,. Zeuzem et al have reported 25 patients with genotype 1 (1c/e/g/h/l) who all achieved SVR with an NS5a inhibitor-based regimen [8]. These results were considered reassuring. It should be noted, however, that 18 patients received either sofosbuvir and velpatasvir, (7 with voxilaprevir) or 24 weeks of sofosbuvir and ledipasvir. Therefore the majority of patients received the most potent treatment currently available, or else lengthened courses of treatment
The French National Reference Centre recently reported that genotype 4r is associated with a lower response rate to DAAs. Five percent of all treatment failures were in patients infected with G4r, although this subtype is rare in the French population overall. Treatment failure was associated with multiple NS5A RASs which were present in all those analysed at baseline, similarly to our cohort [16]. Gupta et al recently published a large scale single arm trial in Rwanda showing that genotype 4r, present in 16% of participants, was associated with high rates of unsuccessful treatment with sofosbuvir and ledipasvir, despite good adherence [17]. This has been linked to the presence of an amino acid motif, MPRMP, at positions 28-32 of the NS5A gene that confers high-level resistance to DAAs in vitro [18].
Population based studies to determine the frequency of unusual subtypes and the efficacy of DAA regimens against them, are urgently needed to formulate guidelines for treating HCV in Africa [19]. The corollary of these data, and the fact that in resource poor regions of Africa genotyping or full genome sequencing will not be available, is that second generation NS5A inhibitors should be preferred as first line agents in the region. Use of second line NS5A inhibitors seems to overcome resistance [20]. However further evaluation of the efficacy of newer regimens including sofosbuvir-velpatasvir or glecaprevir-pibrentasvir against the genotype 1 and 4 subtypes characterised in our analysis, is necessary. It has been suggested that a triple DAA regimen, including a protease inhibitor is required as first line treatment [16, 21].
We have shown that in a metropolitan United Kingdom cohort of African patient with HCV, most patients were infected with unusual and often novel African subtypes which were associated with reduced SVR rates. The reality of global migration means that these data are also relevant for clinicians in high income countries who should exercise caution in selecting regimens for African patients with unusual or un-subtypeable genotypes.
The evidence indicates that the future desired expansion of HCV treatment in Africa may risk unacceptable rates of failure if first generation NS5A inhibitors are utilised without appropriate epidemiological and viral sequence data. Global equity of access to curative treatment is required to avoid jeopardising the hepatitis C elimination agenda.
Five patients who failed initial treatment have thus far been commenced on retreatment: Three of these patients have completed 16 weeks of glecaprevir and pibrentasvir, two of whom have achieved an SVR. One individual with cirrhosis and G1* HCV virus failed to achieve an SVR with 16 weeks of glecaprevir and pibrentasvir. Two other patients were retreated with sofosbuvir, -velpatasvir and voxilaprevir and achieved an SVR.
There are eight known HCV genotypes which have been classified based on the analysis of HCV genetic sequences[4]. Except for genotypes 5 and 8, each genotype is further divided into a number of subtypes. A genome-wide nucleotide sequence difference of 31–33 % is considered sufficient to differentiate a genotype and a difference of 12-15% is sufficient to distinguish a sub-genotype (or subtype), although these boundaries are not strict and phylogeny is also considered.
Clinical trial and real-life data routinely report sustained virological response (SVR) rates in excess of 95% for genotype 1a, (G1a) 1b, 3 and 4. There are fewer data available on less prevalent genotypes such as 5 or 6, nor are unusual subtypes well represented. In an analysis of over 1700 patients with genotype 1 HCV, who had been enrolled in clinical trials of NS5A inhibitors, less than 1% had subtypes of genotype 1 other than 1a or 1b [5]. The available evidence suggests that other genotype 1 subtypes are common in Africa but less frequent in the industrialised countries where clinical trials have been centered [16]. In this paper we refer to these as “unusual genotypes”. We choose this nomenclature for clarity as these genotypes are unusual in Europe, but not unusual in Africa, as we will discuss. However, there is a paucity of data as to whether the treatment response in patients with these less well characterised subtypes is comparable to more common subtypes. This had led to calls for more treatment outcome data in well characterised cohorts of patients[7, 8].
Our institution serves a population of high ethnic diversity with a high prevalence of chronic hepatitis C infection. In this analysis we report the distribution of HCV genotypes and subtypes according to country of birth and treatment outcomes in an immigrant population cohort of patients born in Africa.
Results
Subgenotypes prevalent in African patients
The total cohort of patients with HCV seen at our centre between 2010 and 2018 was 2211 patients, of whom we identified 91 (4.1%) patients who were born in Africa, the majority from sub-Saharan Africa (table 1). The ethnicity or country of birth of the whole HCV cohort are shown in figure 1. Amongst the African patients 20/91 (22%) patients were infected with genotype 1a or 1b, 35 (39%) patients had unusual African genotype 1 subtypes, including sub-genotypes 1e, 1g, 1h, 1l, and 23 patients with unassigned genotype 1. Five patients (5.6%) were infected with genotype 2, three (3.3%) were infected with genotype 3, fourteen (15.6%) had genotype 4, and twelve (13.1%) were infected with unusual subtypes of genotype type 4 including 4c, 4e, 4f, 4k, 4r; two patients had genotype 5 and 6 infection. The frequency of HCV genotypes of the whole cohort compared to the African group are shown in figure 2. Of the non-African patients, who were predominantly British born, but also included Asian, Caribbean and other European backgrounds, 38.7% were infected with genotype 1a, 15.2% with G1b, 30.4% with G3a and only 3.3% with unassigned G1.
In the African cohort, HCV genotypes according to country of birth are shown in table 1 and figure 3. Of the 23 unassigned genotype 1 samples that underwent NGS, 18 complete open reading frame (ORF) sequences from 15 novel unassigned subtypes were identified. Formal HCV subgenotype assignment and classification requires at least three epidemiologically unrelated isolates [4]. We met this recognised criteria with samples from three individuals from Nigeria, shown on the p-distance tree as Patient (P)15, P17, and P38. Two of the patients were a married couple, however phylogenetic analysis indicated that they were infected with different variants of the same subtype. This novel subtype has been assigned as 1p. It should be noted that 1 one of our patients who failed treatment with sofosbuvir and ledipasvir, P17, was infected with the newly identified genotype 1p.
Of our remaining unassigned 14 subtypes, three had pairs of unrelated sequences and the remainder only one. We refer to these novel unclassified subtypes as G1*. These are shown on the phylogenetic tree in figure 4.
Treatment response
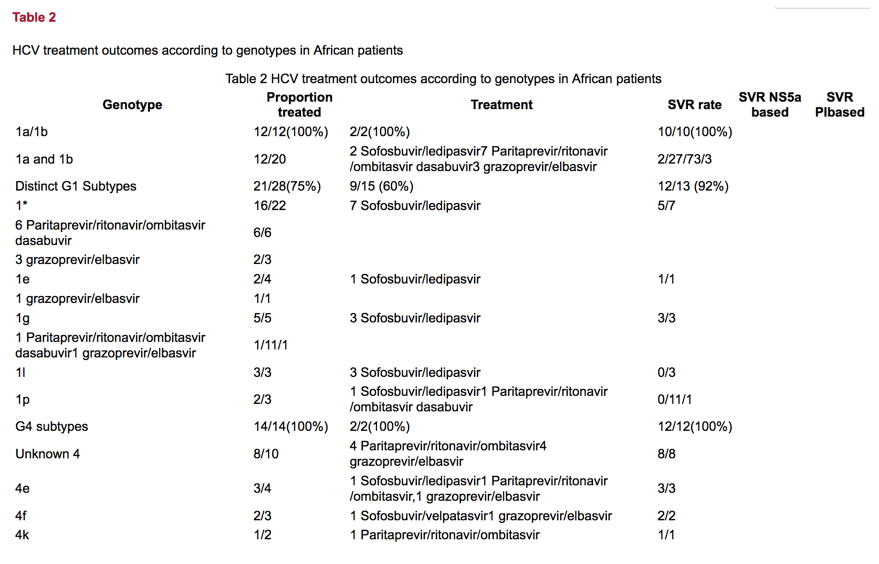
To date, 63 patients have completed DAA treatment and follow up. Fifty six of these have achieved an SVR, 7 failed treatment giving an overall SVR rate of 89%. As expected response rates in genotypes 1a or 1b were uniformly high. Similarly, all patients infected with genotype 2, 3, 4 and 5 achieved an SVR. In those patients with unusual G1 and G4 African subtypes, treatment response according to genotype and choice of treatment regimen are shown in table 2. An SVR was observed in only 21 of the 28 (75%) patients infected with unusual African genotype 1.

Five patients who failed initial treatment have thus far been commenced on retreatment: Three of these patients have completed 16 weeks of glecaprevir and pibrentasvir, two of whom have achieved an SVR. One individual with cirrhosis and G1* HCV virus failed to achieve an SVR with 16 weeks of glecaprevir and pibrentasvir. Two other patients were retreated with sofosbuvir, -velpatasvir and voxilaprevir and achieved an SVR.
Factors associated with lack of SVR
A univariate categorical analysis was carried out to investigate which factors were associated with response. The presence of an unusual genotype 1 African subtype or the use of an NS5A inhibitor based regimen were both significantly associated with a lack of SVR in univariate analysis, no other factors were. The results are shown in table 3.
In multivariate analysis with SVR as the outcome variable, unusual genotype 1 remained highly significant with a p-value of 0.016 (Chi-Squared: 5.78, df=1), treatment regimen (NS5A inhibitor based vs protease inhibitor) was close to significant with a p-value of 0.054 (Chi-Squared: 3.69, df=1).
HCV sequence results
Baseline sequence data was available for 22 patients, 14 who achieved SVR, 6 treatment
failures, 2 who have not yet been treated. There were numerous NS5A polymorphisms present at baseline in patients with unusual G1 subtypes, particularly at positions 24, 30 and 31. Resistance associated polymorphisms (RASs) listed in the EASL 2018 HCV guidelines as conferring reduced susceptibility to NS5A inhibitors or being associated with reduced treatment response were seen at baseline in 18/22 (82%) patients. All the treatment failures had either M28 polymorphisms (L and S) (2 failures) or L31M (4 failures). Five SVR patients had M28 (L and V), Three patients (2 with 1* and one with 1g) who achieved SVR had Y93 (F/H/N) at baseline. These data are expanded in table 4. The individual patient with 1* who has failed treatment with both sofosbuvir/ledipasvir and then glecaprevir/pibrentasvir had Q62E, L31M at baseline and Q62D, L31M following SOF/LDV. Following glecaprevir/pibrentasvir treatment Q30H, H58S and Y93H were also accumulated.

| |
| |
| |
|
|
|