| |
Fanconi Syndrome and Tenofovir Alafenamide: A Case Report
|
| |
| |
Download the PDF here
Letters |29 January 2019
Nathan C. Bahr, MD
Sri G. Yarlagadda, MD
University of Kansas
Kansas City, Kansas
Annals of Internal Medicine Jan 29 2019
Background: In the United States, tenofovir disoproxil fumarate (TDF) has been almost completely replaced by tenofovir alafenamide (TAF) for treatment of HIV infection and chronic hepatitis B virus infection. This change occurred in part because TAF causes less injury to the kidney than TDF (1).
Objective: To alert clinicians to the possibility that TAF can cause Fanconi syndrome.
Case Report: A 54-year-old man with HIV infection came to our institution to establish an ongoing source of care. In 1994, he had been diagnosed elsewhere with HIV. His HIV had been well controlled with antiretroviral regimens containing TDF for the 10 years before he came to our institution. His serum creatinine level 3 months before the transfer of care was 52.16 µmol/L (0.59 mg/dL), and his antiretroviral regimen was TDF–emtricitabine, darunavir–ritonavir, and raltegravir, based on his resistance profile. To avoid chronic toxicities from TDF, his previous provider had arranged for a transition to TAF 1 month later when his TDF ran out.
When we first saw the patient 2 months after the medication change, his initial creatinine level was 491.50 µmol/L (5.56 mg/dL) (Table). We admitted him to the hospital and discontinued all antiretroviral medications because withdrawing TAF alone would have put him at risk for additional HIV resistance. Other medications before admission included omeprazole, L-thyroxine, and rosuvastatin, which were continued during hospitalization. The results of many of his laboratory studies are shown in the Table. A renal ultrasound was unremarkable. No additional nephrotoxic agents were identified.
The combination of hypokalemia, non–anion gap acidosis, glycosuria, and borderline low phosphate levels in the setting of acute kidney injury was consistent with Fanconi syndrome. We discharged the patient 4 days later with improving levels of creatinine. Two weeks after his initial presentation, we started an antiretroviral regimen that was based on previous resistance patterns and did not contain tenofovir, and he rapidly achieved viral control. His creatinine levels remained within normal limits (Table).
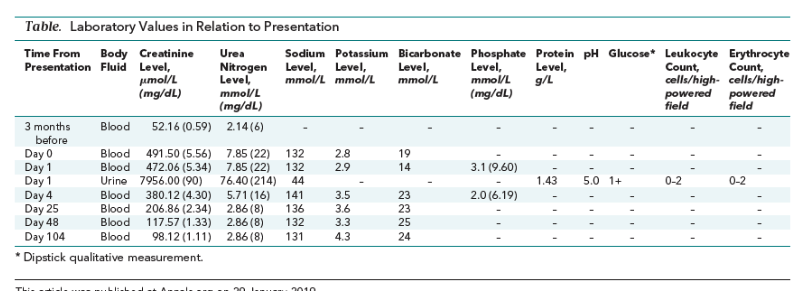
Discussion: Fanconi syndrome is a well-known complication of TDF therapy. Three other case reports have described renal injury related to TAF. One patient had an intentional overdose without tubular injury (2); another had evidence of chronic tubular injury induced by tenofovir in addition to acute kidney injury from other causes (3); and the third had evidence of tubular injury with TAF but only after starting a treatment regimen for hepatitis C that contained ledipasvir, which can increase tenofovir levels (4). To our knowledge, the current case is the first report of Fanconi syndrome due to TAF alone. Using guidelines for reporting adverse drug reactions, we classify this case as “non–dose-related (bizarre)” (5). Adverse drug effects in this category have the potential for high mortality and are uncommon, unpredictable, and unrelated to the drug's pharmacologic action. The mechanism by which TAF might be related to Fanconi syndrome is presumed to be similar to that of TDF, although this is not proven. Using these guidelines, we assess the causality of TAF as “certain” (5). This report therefore reflects an increased possibility that persons starting TAF therapy have risk for kidney injury, although that risk is lower with TAF than TDF. As a result, we recommend that clinicians consider monitoring renal function after beginning TAF therapy while we wait for additional studies to confirm this report, measure how frequently this reaction occurs, and determine whether it is confined to predictable subgroups.
|
|
| |
| |
|
|
|