| |
Long-Acting Once A Month Injection of Cabotegravi/rilpivirine in PLWH with History of Sub-Optimal Adherence Enrolling
|
| |
| |
- study sites and contact information in ink below to clinicaltrials.gov - about 40 study sites & contacts listed below also. Link to sites & contacts:
https://clinicaltrials.gov/ct2/show/study/NCT03635788?term=actg+5359&cond=Hiv&rank=1&show_locs=Y#locn
This study will compare the efficacy, safety, and durability of two different strategies to treat participants with a history of sub-optimal adherence and control of their HIV infection: long-acting (LA) antiretroviral therapy (ART) with rilpivirine (RPV) LA and cabotegravir (CAB) LA versus all-oral standard of care (SOC).
LATITUDE will enroll approximately 350 volunteers with documented treatment lapses within the past 18 months. All study participants will begin a daily oral ART regimen and individualized adherence and retention support.
LATITUDE will enroll approximately 350 volunteers with documented treatment lapses within the past 18 months. All study participants will begin a daily oral ART regimen and individualized adherence and retention support. Volunteers who become virally suppressed by week 24 will then be randomized to either continue standard-of-care therapy for one year or begin an oral regimen consisting of RPV and CAB for 4 weeks, followed by long-acting injectable formulations of those drugs every 4 weeks for 48 weeks. After this 52-week period, participants originally randomized to the standard-of-care arm may cross over to long-acting therapy, and participants originally randomized to the long-acting therapy arm may remain on that regimen for an additional year.
---------------------------
A Phase III Study to Evaluate Long-Acting Antiretroviral Therapy in Non-adherent HIV-Infected Individuals
A Multicenter Trial of the AIDS Clinical Trials Group (ACTG)
Sponsored by:
The National Institute of Allergy and Infectious Diseases
In Collaboration with:
The National Institute of Mental Health
The National Institute of Drug Abuse
Industry Support Provided by:
ViiV
Janssen
Brief Summary:
The purpose of this study is to compare the efficacy, safety, and durability of two different strategies to treat participants with a history of sub-optimal adherence and control of their HIV infection: long-acting (LA) antiretroviral therapy (ART) and all-oral standard of care (SOC).
LATITUDE will enroll approximately 350 volunteers with documented treatment lapses within the past 18 months. All study participants will begin a daily oral ART regimen and individualized adherence and retention support. Volunteers who become virally suppressed by week 24 will then be randomized to either continue standard-of-care therapy for one year or begin an oral regimen consisting of RPV and CAB for 4 weeks, followed by long-acting injectable formulations of those drugs every 4 weeks for 48 weeks. After this 52-week period, participants originally randomized to the standard-of-care arm may cross over to long-acting therapy, and participants originally randomized to the long-acting therapy arm may remain on that regimen for an additional year.
--------------------------
The LATITUDE Study: Long-Acting Therapy to Improve Treatment SUccess in Daily LifE
https://clinicaltrials.gov/ct2/show/study/NCT03635788?term=actg+5359&cond=Hiv&rank=1&show_locs=Y#locn
NIH Announcement:
https://www.niaid.nih.gov/news-events/nih-trial-evaluates-long-acting-hiv-medication-unable-adhere-strict-daily-regimens
Detailed Description:
This study will compare the efficacy, safety, and durability of two different strategies to treat participants with a history of sub-optimal adherence and control of their HIV infection: long-acting (LA) antiretroviral therapy (ART) with rilpivirine (RPV) LA and cabotegravir (CAB) LA versus all-oral standard of care (SOC).
The study includes four steps. In Step 1, participants will receive a SOC oral induction regimen consisting of an ART regimen that involves at least 3 drugs for 24 weeks. Participants who achieve milestones will receive conditional economic incentives at Weeks 2, 4, 8, 12, 16, and 20.
In Step 2, eligible participants will be randomized to receive either oral RPV + oral CAB for 4 weeks followed by RPV-LA + CAB-LA every 4 weeks for 48 weeks or to continue on SOC for 52 weeks.
At the completion of Step 2, eligible participants randomized to SOC will have the option to register to Step 3 and receive LA ART, which includes oral RPV + oral CAB for 4 weeks followed by RPV-LA + CAB-LA every 4 weeks for 48 weeks. Participants already receiving RPV-LA + CAB-LA in Step 2 will continue on this regimen in Step 3 for 52 weeks.
Eligible participants will enter Step 4 and be followed for 52 weeks on locally sourced oral ART.
Participants will be followed for up to a total of 180 weeks. Study visits, which will occur throughout the study, may include physical examinations; blood, urine, and hair collection; liver function tests; questionnaires; and an electrocardiogram (ECG).
Adherence Monitoring and Support
This study seeks to enroll participants with a history of non-adherence to medications, a group not traditionally targeted for recruitment in ACTG trials. Since CEI alone might not be sufficient to encourage adherence, addressing the challenges to adherence in this study population will be imperative to ensure study retention and allow for complete interpretation of study results.
In addition to the CEI used during the induction period, sites will be required to develop a multi-component adherence support protocol utilizing the framework of the Guidelines for Improving Entry Into and Retention in Care and ARV Adherence for Persons With HIV: Evidence-Based Recommendations From an International Association of Physicians in AIDS Care Panel, with a focus on the recommendations to promote adherence to visits and improve adherence to ART (see MOPS) [36]. In addition, multiple methods of participant recruitment and retention will be utilized, including appointment reminder via phone call, email, SMS and social media in a HIPAA-compliant fashion. We expect these recommendations will enhance current study adherence and retention efforts by ACTG sites.
Conditional Economic Incentives (CEI)
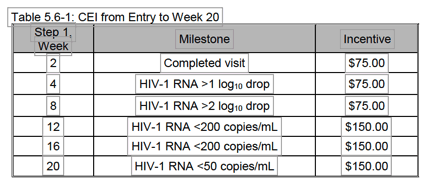
Adherence benchmarks using study visit and HIV-1 RNA decline will be evaluated during Step 1 the induction phase (first 20 weeks) and CEI will be provided to participants if the predetermined milestones are met during this period (Table 5.6-1). As described above, the rationale behind limiting the cash incentives to the induction phase is to utilize them as a time-limited intervention to support adherence to oral therapy in individuals with a history of sub-optimal adherence to oral medications and to improve the chance that viral suppression will be achieved before transitioning to LA ART.
At Step 1, week 2, a $75 incentive will be provided for completion of a return study visit. At Step 1, weeks 4 and 8, individuals who have a decrease of >1 log10 and >2 log10 in HIV-1 RNA compared to HIV-1 RNA at entry, respectively, will be incentivized $75 for each milestone met. If unable to assess log drop at Step 1, week 4 and 8, a benchmark on HIV-1 RNA decline to <200 copies/mL will be used. At Step 1, week 12, HIV-1 RNA <200 copies/mL will be used as the milestone (if achieved) and will be incentivized $150, with the opportunity for a repeat incentive at Step 1, week 16, and at Step 1, week 20, if the individual remains virologically suppressed at <50 copies/mL.
If a participant skips visit(s), he or she can be eligible for an incentive not received in a previous missed visit; e.g., if participant misses Step 1, week 8 visit, he or she can be eligible for the $75 incentive of dropping more than 2 log10 copies in HIV RNA at Step 1, weeks 9, 10, or 11 visits.
Participants who do not achieve the benchmarks at any point will not receive the CEI.
All incentives will be additional to site’s usual compensation for participation in the study (i.e., attending study visits and completing assessments). Since there will be a short lapse between study visits and virologic assay results, incentives may be provided using a debit card system, which will allow for funds to be added remotely once the data becomes available, although in person incentive distribution will also be permitted. Study participants will be notified immediately via phone call, text SMS, email, or other preferred communication method after their benchmark is achieved.
No other steps will be incentivized financially beyond standard site-specific compensation for study visit attendance.
|
|
| |
| |
|
|
|