| |
Scales Tip Toward 'Fish Oil' PUFA Intake for Heart Failure Prevention
Predicting Risk for Incident Heart Failure With Omega-3 Fatty Acids
From MESA - 1
|
| |
| |
Download the PDF here
Download the PDF here
2. Predicting Risk for Incident Heart Failure With Omega-3 Fatty Acids
From MESA
3. editorial - Omega-3 Fatty Acids and Heart Failure
Evidence and Still Open Questions
The recent trials and the observations of the MESA cohort suggests that with very high plasma levels of omega-3 fatty acids we can obtain a reduction of major CV events (6), a prevention of HF occurrence (4), and a favorable effect on the left ventricular remodeling processes (5). High plasma levels of omega-3 fatty acids can probably be achieved just with the use of purified pharmacological preparations.
Considering specifically HF, the study from Block et al. (4) suggests that high circulating levels of omega-3 fatty acids can prevent HF occurrence.
from original study:
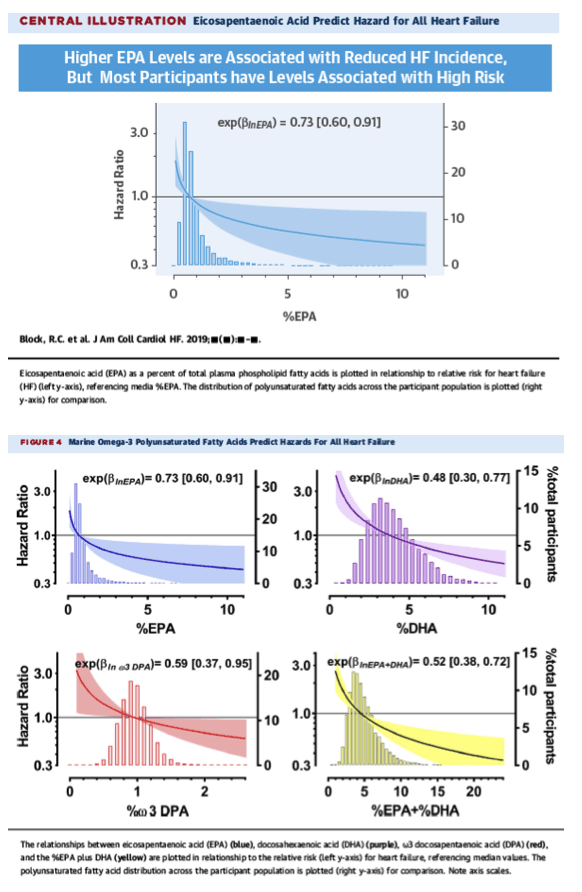
"Here we show in the MESA study that high plasma phospholipid EPA is associated with reduced risk for all HF, including both HFrEF and HFpEF Central Illustration, confirming our primary hypothesis. In addition, we found that high plasma DHA, ω3 DPA, and EPA plus DHA are similarly associated, indicating that unlike mice, humans may benefit from marine ω3 PUFAs generally. The findings were true in univariate analysis; after adjusting for age, sex, race, and study center; after accounting for replacement effects of other FAs; and after further selective adjustment for covariate factors known to predict HF. Finally, the protective associations remained evident after sensitivity analyses in which only HFpEF or only HFrEF participants were included......Recently, the VITAL (Vitamin D and Omega-3 Trial) study reported that low-dose ω3 PUFAs failed to prevent composite cardiovascular death, nonfatal MI, and stroke over 5-year follow-up; a secondary outcome suggested a reduced risk for MI (40). VITAL was preceded by a meta-analysis suggesting that low-dose ω3 PUFAs do not prevent CHD (24). Neither study directly reported HF outcomes. These results stand in contrast to results from REDUCE-IT (Reduction of Cardiovascular Events With Icosapent Ethyl–Intervention Trial), which demonstrated that high-dose icosapent ethyl (an EPA precursor) at 4 g/day produced a 25% reduction in the risk for composite cardiovascular death, nonfatal MI, nonfatal stroke, coronary revascularization, and unstable angina (27). These results are supported by the results of OMEGA-REMODEL, which indicate that high-dose ω3 PUFAs (4 g/day) prevent post-MI remodeling (26). Consistently, subjects achieving the highest red blood cell %EPA plus %DHA levels were associated with the greatest treatment benefits (26). These newer trials examining the effects of high-dose ω3-PUFA intake suggest a concentration-dependent cardioprotective effect as previously suggested (13). REDUCE-IT reported no difference in HF incidence (4.1% with placebo vs. 4.3% with EPA) (27), but the HF incidence was modest and could not exclude a large range of effects (+17% to −23% risk). MESA had a broader age range, a longer follow-up period, and broader diversity. Furthermore, REDUCE-IT included only subjects with hypertriglyceridemia on statin therapy, with known cardiovascular disease or equivalents (27)."
"Higher plasma EPA was significantly associated with reduced risk for HF, with both reduced and preserved EF. (Multi-Ethnic Study of Atherosclerosis [MESA]" study below
We have shown that dietary ω3 PUFAs at supraphysiological levels preserve left ventricular function and prevent interstitial fibrosis in a mouse model of pressure overload–induced HF (28). Follow-up with a diet designed to achieve ω3 PUFA levels closer to those achieved in patients treated with high-dose prescription omega-3 acid ethyl esters showed that only EPA was protective (13,25)......HF-free participants had higher baseline plasma phospholipid %EPA, %DHA, and %EPA plus %DHA. Age, body mass index, diabetes mellitus, glomerular filtration rate, hypertension, use of a loop diuretic agent, urine microalbuminuria, and use of an angiotensin-converting enzyme inhibitor were significantly different (p < 0.05) between participants with and those without HF.
In summary, %EPA was inversely related to HF risk, the relationship was robust and independent to adjustment, and this association was present among all participants, including those with HFrEF and those with HFpEF (see Online Table 5)."
------------------------------------------
Scales Tip Toward 'Fish Oil' PUFA Intake for Heart Failure Prevention
Marlene Busko
July 16, 2019
High plasma levels of the kinds of fatty acids found in fish oil were associated with a lower long-term risk for new heart failure (HF), whether with reduced or preserved ejection fraction (HFrEF or HFpEF), in a community-based cohort of more than 6000 people.
The greater the plasma levels of eicosapentaenoic acid (EPA), a prevalent n-3 polyunsaturated fatty acid (n-3 PUFA, also called omega-3 PUFA), the lower the risk for both forms of HF during a median follow-up of 13 years.
Similar independent observations were made for plasma levels of docosahexaenoic acid (DHA) and of EPA and DHA combined, suggesting that increased levels of n-3 PUFA in general may confer cardiovascular (CV) benefits, observe the study's authors, led by Robert C. Block, MD, MPH, University of Rochester School of Medicine and Dentistry, New York.
The findings from the Multi-Ethnic Study of Atherosclerosis (MESA), published July 10 in JACC: Heart Failure, add to a literature of abundant but diverse observations on the CV effects of elevated levels of n-3 PUFA, whether achieved by diet or supplements.
The MESA cohort consists of initially middle-aged adults and is noteworthy for being about evenly divided between women and men and including large proportions of African Americans and other non-white groups.
The analysis "may reopen the discussion on the role of omega-3 fatty acids in the context of prevention and treatment of heart failure," writes Aldo P. Maggioni, MD, Heart Care Foundation, Florence, Italy, in an accompanying editorial.
"This study clearly demonstrated a significant independent inverse correlation between circulating levels of omega-3 fatty acids, specifically eicosapentaenoic acid, and the occurrence of HF over a long median follow-up period of 13 years," he writes.
To reap such benefits, "shall we have to go to the fish market or to the pharmacy to elevate our circulating levels of omega-3 fatty acids and, in this way, to try to prevent (or treat) heart failure?" Maggioni asks.
He argues in favor of the pharmacy. Cardiovascular outcomes from OMEGA-REMODEL and REDUCE-IT, the current results, and studies in mice "suggest that just with very high plasma levels of omega-3 fatty acids we can obtain a reduction of major CV events, a prevention of HF occurrence, and a favorable effect on the left ventricular remodeling processes."
Moreover, "High plasma levels of omega-3 fatty acids can be probably achieved just with the use of purified pharmacological preparations."
"Most Americans, regardless of race or ethnicity," have insufficient levels of n-PUFAs and "would benefit from increased intake," senior author Gregory C. Shearer, PhD, Pennsylvania State University, University Park, said in emailed comments to theheart.org | Medscape Cardiology.
Fish-oil supplements would likely be more effective than eating more fish to achieve the n-3 PUFA levels that may be of benefit, Shearer and coauthor Timothy D O'Connell, PhD, University of Minnesota, Minneapolis, speculated in a joint interview.
As both the report and editorial note, n-3 PUFA supplementation at the fairly low dosage of 1 g/day, added to standard therapy, was associated with reduced all-cause mortality and HF hospitalization rates over about 4 years in the 2008 GISSI-HF trial.
In the 2015 OMEGA-REMODEL trial, patients who took a proprietary n-PUFA preparation (Lovaza, GlaxoSmithKline) at 4 g/day for 6 months after a myocardial infarction (MI) showed reductions in ventricular remodeling, fibrosis, and inflammatory markers.
More recently, in the REDUCE-IT trial, patients with high triglyceride levels and CV disease or diabetes plus one other CV risk factor showed a 25% reduction in the composite outcome of CV death, nonfatal MI, nonfatal stroke, coronary revascularization, or unstable angina over 5 years while taking icosapent ethyl (Vascepa, Amarin) at 4 g/day. The agent acts as a precursor of EPA.
The current analysis classified 65,632 high-risk MESA participants according to their plasma percent-EPA levels.
Levels were <1.0%, or "insufficient," compared to adjusted levels that prevented HF in animal models, in 73.1% of participants.
They were 1% to 2.5%, or "marginally sufficient," in 22.4% of participants; and 4.5% of the cohort had "sufficient" levels, at >2.5%.
Only 1.4% of Hispanic participants had sufficient percent-EPA levels; the proportions were 4.4% for African Americans, 4.9% for whites, and 9.8% for participants of Asian descent.
There were 292 HF events — 128 involving HFrEF, 110 in HFpEF, and 54 in people whose ejection fractions were unknown — during a median of 13 months. The mean percent-EPA level was 0.76% for participants who did not develop HF, vs 0.69% for participants who did develop HF (P = .005).
In an analysis adjusted for age, sex, race, body-mass index, smoking status, type 2 diabetes, blood pressure, lipids, lipid-lowering therapy, albuminuria, and types of PUFA, percent-EPA was inversely associated with risk for HF at a hazard ratio (HR) of 0.73 for each log-unit difference (P = .001).
"Clinical trials using 1 g/day omega-3 fatty acids," such as GISSI-HF, "seem to reduce HF risk by about 10%," said Shearer. But this dose would likely only increase the average percent-EPA from 0.7% to the marginally sufficient range, between 1% and 1.5%.
Any forthcoming trials should be designed "to achieve a goal of greater than 3.5% EPA, or greater than 12% EPA plus DHA," he said.
"Based on the results from MARINE and ANCHOR, we are optimistic that a larger [n-3 PUFA supplement] dose such as 4 g/day" would result in an average percent-EPA of 4% and about a 35% reduced risk for HF.
"If we want to move from hypotheses to more reliable evidences," Maggioni agrees, "it is probably the time to design again adequately sized randomized clinical trials testing high dosages of omega-3 fatty acids on top of current optimized pharmacological and non-pharmacological therapies."
Their goals, he writes, should be to determine whether the intervention has an effect on clinical outcomes in patients with overt HF and the ability to avert HF in patients with structural heart disease but who do not have signs or symptoms of HF.
"Considering the very favorable tolerability and safety profile of this therapeutic approach, any positive results of these trials could provide us with an additional strategy to improve the outcomes of patients with HF or at high risk to develop it," he writes.
Block has disclosed no relevant financial relationships. Shearer has received honoraria from Amarin Pharmaceuticals. Disclosures for the other authors are listed in the report. Maggioni has received honoraria for participation in committees of studies sponsored by Bayer, Novartis, and Fresenius outside the scope of the current study.
JACC: Heart Fail. Published online July 10, 2019. Abstract, Editorial
For more from theheart.org , follow us on Twitter and Facebook
-------------------------------------------
Editorial Comment
Omega-3 Fatty Acids and Heart Failure
Evidence and Still Open Questions
Aldo P. Maggioni
JACC: Heart Failure july 2019
Approximately 10 years ago, the first large-scale clinical trial testing omega-3 fatty acids versus placebo in nearly 7,000 patients with heart failure (HF) was publicly presented and published (1). The trial showed that adding 1 gram daily of omega-3 fatty acids to the best medical treatment of those days resulted in a 9% reduction of all-cause mortality and 8% reduction of the combined outcome measure of all-cause death or hospitalization for cardiovascular (CV) reasons over a median follow-up period of 3.9 years. The beneficial effect was modest but statistically significant and obtained in a context of a very reassuring safety and tolerability profile. The favorable message of the GISSI-HF (Gruppo Italiano per lo Studio della Sopravvivenza nell'Insufficienza cardiaca-Heart Failure) trial was mechanistically supported by some other smaller studies suggesting a positive effect of omega-3 fatty acids on left ventricular remodeling, endothelial function, and inflammatory markers (2–3). However, current international guidelines are not giving much consideration to these evidences, not considering at all omega-3 fatty acids (American Heart Association/American College of
Cardiology guidelines 2017) or suggesting a weak recommendation (Level 2b/B in the European Society of Cardiology guidelines 2016).
The paper by Block et al. (4) may reopen the discussion on the role of omega-3 fatty acids in the context of prevention and treatment of HF. This study clearly showed a significant independent inverse correlation between circulating levels of omega-3 fatty acids, specifically eicosapentaenoic acid (EPA), and the occurrence of HF over a long median follow-up period of 13 years. In addition, having analyzed the MESA (Multi-Ethnic Study of Atherosclerosis) cohort of patients, authors concluded that plasma levels of EPA can predict the occurrence of HF in all ethnicities. The same authors also offer a pathophysiologic plausibility to these results, showing in animal models with HF that these dietary supplements can preserve left ventricular function and reduce interstitial fibrosis.
A limit of this study is that, due to the relatively low number of cases with HF with preserved ejection fraction, it is not possible to have reliable information on the potential protective role of elevated plasma levels of omega-3 fatty acids on this specific HF condition. Even more important is the consideration that a small number of subjects presented with plasma levels of omega-3 fatty acids sufficiently high to be considered as protective of HF occurrence. This last observation opens the door to the question on the potential preventive role of different dosages of omega-3 fatty acids generally tested and used up to now at the low dosage of 1 gram per day (1). The use of higher dosages of omega-3 fatty acids has been supported by 2 recent studies: the OMEGA-REMODEL (Omega-3 Acid Ethyl Esters on Left Ventricular Remodeling After Acute Myocardial Infarction) trial (5) and the REDUCE-IT (Reduction of Cardiovascular Events with Icosapent Ethyl–Intervention Trial) (6).
In 358 post-infarction patients, the OMEGA-REMODEL trial showed that a 6-month treatment with 4 grams daily of omega-3 fatty acids on top of the current guideline-based standard of care was associated with a reduction of adverse left ventricular remodeling, myocardial fibrosis, and serum biomarkers of systemic inflammation.
The large-scale randomized clinical trial, REDUCE-IT, showed in 8,179 patients with elevated levels of triglycerides and with an established cardiovascular disease (70% in secondary prevention) or with diabetes mellitus and at least 1 additional risk factor (30% in primary prevention) that the treatment with 4 g/day of EPA was able to significantly reduce by 25% the primary combined endpoint of CV death, nonfatal myocardial infarction, nonfatal stroke, coronary revascularization, or unstable angina, over a median follow-up period of 4.9 years. High dosages of EPA also significantly reduced the typical endpoint of trials of CV prevention (CV death, nonfatal myocardial infarction, and nonfatal stroke) but had no effect on occurrence of HF, contradicting, in some way, the evidence provided by Block et al. (4). However, the rate of occurrence of HF events during the course of the follow-up was low, not allowing a reliable estimate of the EPA effects on this relevant secondary endpoint of the trial.
Which could be the role of omega-3 fatty acids in the field of CV medicine in 2019? With respect to primary CV prevention, several trials showed neutral effects with the use of a dosage of omega-3 fatty acids of 1 gram per day, therefore excluding a relevant role of this treatment (at this dosage) in high-risk patients, but without a prior atherothrombotic event. As far as secondary prevention is concerned, the high dosage of 4 grams per day of EPA was clearly shown to be effective in preventing major atherothrombotic events, including CV death, but not HF.
Considering specifically HF, the study from Block et al. (4) suggests that high circulating levels of omega-3 fatty acids can prevent HF occurrence. The question is: is it sufficient to give dietary advice of an increased fish consumption, or do we need to take purified pharmaceutical supplements such as those tested in trials? In other words, shall we have to go to the fish market or to the pharmacy to elevate our circulating levels of omega-3 fatty acids and, in this way, to try to prevent (or treat) HF?
The recent trials and the observations of the MESA cohort suggests that with very high plasma levels of omega-3 fatty acids we can obtain a reduction of major CV events (6), a prevention of HF occurrence (4), and a favorable effect on the left ventricular remodeling processes (5). High plasma levels of omega-3 fatty acids can probably be achieved just with the use of purified pharmacological preparations.
In any case, if we want to move from hypotheses to more reliable evidence, it is probably the time to redesign adequately sized randomized clinical trials testing high dosages of omega-3 fatty acids on top of current optimized pharmacological and non-pharmacological therapies with the aim: 1) to improve the clinical outcomes of patients with documented overt HF (HF Stage C); and 2) to prevent the occurrence of HF in patients with structural heart disease but without signs or symptoms of HF (HF Stage B). Considering the favorable tolerability and safety profile of this therapeutic approach, any positive results of these trials could provide us with an additional strategy to improve the outcomes of patients with HF or at high risk to develop it.
-----------------------
Predicting Risk for Incident Heart Failure With Omega-3 Fatty Acids
From MESA
Robert C. Block, Linxi Liu, David M. Herrington, Shue Huang, Michael Y. Tsai, Timothy D. O'Connell and Gregory C. Shearer
Discussion
Here we show in the MESA study that high plasma phospholipid EPA is associated with reduced risk for all HF, including both HFrEF and HFpEF Central Illustration, confirming our primary hypothesis. In addition, we found that high plasma DHA, ω3 DPA, and EPA plus DHA are similarly associated, indicating that unlike mice, humans may benefit from marine ω3 PUFAs generally. The findings were true in univariate analysis; after adjusting for age, sex, race, and study center; after accounting for replacement effects of other FAs; and after further selective adjustment for covariate factors known to predict HF. Finally, the protective associations remained evident after sensitivity analyses in which only HFpEF or only HFrEF participants were included.
Previous clinical trials have demonstrated that combined EPA and DHA administration improve HF outcomes. The CHS (Cardiovascular Health Study), a prospective cohort study from 1992 to 2006, also found an association between plasma phospholipid %EPA and a reduction in incident HF, 50% lower in the highest versus the lowest quartile (18). In our analysis, we estimated a more modest contrast, likely reflective of other care-related improvements since the CHS report.
In GISSI-HF (Gruppo Italiano per lo Studio della Sopravvivenza nell'Infarto Miocardico–Heart Failure), low-dose ω3 PUFAs (0.84 g/day) reduced hazards for total mortality (HR: 0.91; 95% CI: 0.83 to 0.99) and HF hospitalization when added to standard therapy (19). Red blood cell EPA plus DHA was measured in a subset of participants, and an increase from 4.8% to 6.7% was found (35), indicating that the participants did not achieve the proposed cardioprotective level of 8.0% (36,37). The final erythrocyte EPA was 1.2%, corresponding to 0.9% plasma phospholipid EPA (38), indicating that the dose did not achieve sufficient levels. In another smaller HF trial in patients with left ventricular insufficiency, combined EPA and DHA (1.7 g/day) improved systolic and diastolic dysfunction (20). We estimate that the dose would increase red blood cell %EPA plus %DHA from the Italian average of 4.7% (39) to 6.7% in 3 months, again still not reaching optimal levels. Furthermore, in OMEGA-REMODEL (Effect of High Dose Fish Oil Supplementation After Recent Heart Attack Using Magnetic Resonance Imaging), high-dose ω3 PUFA therapy (3.4 g/day) for 6 months post–myocardial infarction (MI) reduced infarct size, improved ventricular systolic function, and reduced noninfarct myocardial fibrosis (26). Treatment increased %EPA plus %DHA from 5.5 ± 1.8% to approximately 10%. A number of smaller studies also showed that ω3 PUFA therapy improves systolic and diastolic function (21,22). In each study, the beneficial effects of ω3 PUFAs were observed in patients with HFrEF. Therefore, evidence from 1 major HF trial (GISSI-HF), several smaller trials, and 1 trial examining post-MI remodeling all suggest that ω3 PUFAs prevent HF even when optimal tissue enrichment is not achieved.
We found that high plasma EPA is associated with reduced risk for HFpEF, a condition for which there are currently no U.S. Food and Drug Administration–approved therapies that reduce mortality or hospitalizations. Our prior studies in mice indicated a concentration-dependent effect for EPA to prevent pathological remodeling, preserving diastolic function and preventing interstitial fibrosis, in a pressure-overload model of HF that resembles HFpEF remodeling (13,25,28). We did not find strong evidence for the EPA cardioprotective threshold that exists in animals, but only 301 participants had %EPA levels with minimal sufficiency or greater (>2.5%), with only 6 HF events among them. To our knowledge this is the first clinical study to suggest a specific benefit for EPA or ω3 PUFAs in HFpEF.
Recently, the VITAL (Vitamin D and Omega-3 Trial) study reported that low-dose ω3 PUFAs failed to prevent composite cardiovascular death, nonfatal MI, and stroke over 5-year follow-up; a secondary outcome suggested a reduced risk for MI (40). VITAL was preceded by a meta-analysis suggesting that low-dose ω3 PUFAs do not prevent CHD (24). Neither study directly reported HF outcomes. These results stand in contrast to results from REDUCE-IT (Reduction of Cardiovascular Events With Icosapent Ethyl–Intervention Trial), which demonstrated that high-dose icosapent ethyl (an EPA precursor) at 4 g/day produced a 25% reduction in the risk for composite cardiovascular death, nonfatal MI, nonfatal stroke, coronary revascularization, and unstable angina (27). These results are supported by the results of OMEGA-REMODEL, which indicate that high-dose ω3 PUFAs (4 g/day) prevent post-MI remodeling (26). Consistently, subjects achieving the highest red blood cell %EPA plus %DHA levels were associated with the greatest treatment benefits (26). These newer trials examining the effects of high-dose ω3-PUFA intake suggest a concentration-dependent cardioprotective effect as previously suggested (13). REDUCE-IT reported no difference in HF incidence (4.1% with placebo vs. 4.3% with EPA) (27), but the HF incidence was modest and could not exclude a large range of effects (+17% to −23% risk). MESA had a broader age range, a longer follow-up period, and broader diversity. Furthermore, REDUCE-IT included only subjects with hypertriglyceridemia on statin therapy, with known cardiovascular disease or equivalents (27).
The overall HF rate observed in MESA was similar to those seen in other observational cohorts. HF occurred in 4.4% of PREVEND (Prevention of Renal and Vascular End-Stage Disease) participants during a median of 11.5 years (41), similar to 4.5% incidence in 13 years for MESA. The Physicians' Health Study I (1982 to 2008) reported a lifetime HF risk of 13.8% over a much longer 22.4 years (42).
Clinical implications
We propose 3 clinical implications of our findings. First, on the basis of prior work in animals and related findings in humans, it is reasonable to expect this finding to translate to ω3 PUFA intervention. In the interventional setting, each unit change in log %DHA would yield a greater risk reduction than each unit change in log %EPA, but this does not take into account that in the typical interventional setting, each gram per day of EPA proportionally increases %EPA more than each gram per day of DHA increases %DHA. Each gram per day of DHA increases the absolute erythrocyte %DHA more than EPA raises %EPA. However, when considered on the proportional or log scale, each gram per day of EPA is more effective than each gram per day of DHA (43). The latter expectation conforms better to our analysis, but more studies are required to determine which, if any, ω3 PUFA is superior. Second, patients with HF, regardless of ejection fraction status, would benefit from safe, effective therapies, with no adverse interactions with current medications, and ω3 PUFAs appear to meet these criteria (44). Third, the differences among ethnicities in ω3 PUFAs could explain a component of HF health disparities (45).
Study strengths
Strengths of this study include a large sample size, ethnic diversity, long duration of follow-up, modern medical therapy. and accounting for the competing effects of other FAs. No published studies exist in which a clinical trial of ω3 PUFAs in primary prevention of HF incidence has occurred (46), making this observation relevant.
Study limitations
Limitations include a population with few patients having HFpEF and few participants with protective levels of EPA, by our animal models. In addition, only baseline data were available, and we could not account for changes in ω3 PUFAs and other risk factors. We consider this study to strongly determine that a benefit of EPA exists but insufficient to determine whether a threshold for %EPA exists near 3%. In our discussion, we used red blood cell and plasma phospholipid abundance somewhat interchangeably. The analytic answers are valid, but the enrichment of red blood cells is 0.71-fold lower for EPA and 1.13-fold higher for DHA (38).
Conclusions
We show here that plasma phospholipid %EPA is inversely associated with all HF incidence, both HFpEF and HFrEF. In contrast to findings in animals, the inverse association was also found for other ω3 PUFAs and was strongest for combined %EPA plus %DHA.
Perspectives
COMPETENCY IN MEDICAL KNOWLEDGE: This study is the first to determine the ability of plasma phospholipid %EPA to predict HF outcomes in white, African American, Asian, and Hispanic populations. Given that plasma %EPA can be increased by the ingestion of seafood or fish oil capsules while being safe and relatively inexpensive, this preventive measure is limited in the response it produces but is quite feasible.
TRANSLATIONAL OUTLOOK 1:The study provides evidence for measuring plasma phospholipid ω3 PUFAs as an approach to estimating HF risk in adults.
TRANSLATIONAL OUTLOOK 2: A follow-up study should be considered that includes participants with higher levels of %EPA. Such a study would be better powered to detect a threshold for protective effect at high (%EPA >4.0%) levels.
TRANSLATIONAL OUTLOOK 3: An interventional study should be considered that includes a dose of EPA or EPA derivative capable of increasing %EPA from 0.70% to >4.0%.
Abstract
Objectives The aim of this study was to determine if plasma eicosapentaenoic acid (EPA) abundance (%EPA) is associated with reduced hazard for primary heart failure (HF) events in the MESA (Multi-Ethnic Study of Atherosclerosis) trial.
Background Clinical trials suggest that omega-3 polyunsaturated fatty acids (ω3 PUFAs) prevent sudden death in coronary heart disease and HF, but this is controversial. In mice, the authors demonstrated that the ω3 PUFA EPA prevents contractile dysfunction and fibrosis in an HF model, but whether this extends to humans is unclear.
Methods In the MESA cohort, the authors tested if plasma phospholipid EPA predicts primary HF incidence, including HF with reduced ejection fraction (EF) (EF <45%) and HF with preserved EF (EF ≥45%) using Cox proportional hazards modeling.
Results A total of 6,562 participants 45 to 84 years of age had EPA measured at baseline (1,794 black, 794 Chinese, 1,442 Hispanic, and 2,532 white; 52% women). Over a median follow-up period of 13.0 years, 292 HF events occurred: 128 HF with reduced EF, 110 HF with preserved EF, and 54 with unknown EF status. %EPA in HF-free participants was 0.76% (0.75% to 0.77%) but was lower in participants with HF at 0.69% (0.64% to 0.74%) (p = 0.005). Log %EPA was associated with lower HF incidence (hazard ratio: 0.73 [95% confidence interval: 0.60 to 0.91] per log-unit difference in %EPA; p = 0.001). Adjusting for age, sex, race, body mass index, smoking, diabetes mellitus, blood pressure, lipids and lipid-lowering drugs, albuminuria, and the lead fatty acid for each cluster did not change this relationship. Sensitivity analyses showed no dependence on HF type.
Conclusions Higher plasma EPA was significantly associated with reduced risk for HF, with both reduced and preserved EF. (Multi-Ethnic Study of Atherosclerosis [MESA]; NCT00005487)
ABBREVIAT IONS
AND ACRONYMS
AA = arachidonic acid
CHD = coronary heart disease
CI = confidence interval
DHA = docosahexaenoic acid
DPA = docosapentaenoic acid
EPA = eicosapentaenoic acid
FA = fatty acid
HF = heart failure
HFpEF = heart failure with
preserved ejection fraction
HFrEF = heart failure with
reduced ejection fraction
HR = hazard ratio
MI = myocardial infarction
u3 PUFA = omega-3
polyunsaturated fatty acid
Heart failure (HF) is a leading cause of hospitalization in the United States (1). Its incidence increases with age and is higher in men than women (2,3). Currently, 26 million patients globally have HF, with >1 million annual hospitalizations in the United States and Europe, accounting for 1% to 2% of total health expenditures (4,5).
Clinically, HF manifests in 2 modes defined by ventricular function: HF with reduced ejection fraction (HFrEF) (ejection fraction ≤45%) and HF with preserved ejection fraction (HFpEF) (ejection fraction >45%) (6). Half of all current diagnoses are HFpEF (7), and the incidence rate of HFpEF has surpassed that of HFrEF (8). Survival for HFpEF is marginally higher than for HFrEF but is only 35% at 5 years (6,9). Generally, patients with HFpEF are older, female, and more likely to have hypertension, renal disease, atrial fibrillation, and/or pulmonary disease (10). Unfortunately, standard pharmacological therapies for HFrEF show no efficacy in HFpEF (6).
In humans, the omega-3 polyunsaturated fatty acids (ω3 PUFAs) eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are important regulators of cardiovascular health (11–13). Several clinical trials have demonstrated that ω3 PUFAs confer a survival benefit in coronary heart disease (CHD) by preventing sudden death (14–18), and clinical trials have indicated ω3 PUFAs might improve outcomes in patients with HF (19–23). Despite these potential benefits, the use of ω3 PUFAs in patients with CHD and HF remains controversial. A recent meta-analysis involving more than 77,000 participants reported no evidence supporting supplemental ω3 PUFAs in patients with CHD (24) but did not evaluate either HF as an endpoint or studies using doses sufficient to achieve protective concentrations in animals (25) or humans (26,27).
We have shown that dietary ω3 PUFAs at supraphysiological levels preserve left ventricular function and prevent interstitial fibrosis in a mouse model of pressure overload–induced HF (28). Follow-up with a diet designed to achieve ω3 PUFA levels closer to those achieved in patients treated with high-dose prescription omega-3 acid ethyl esters showed that only EPA was protective (13,25).
MESA (Multi-Ethnic Study of Atherosclerosis) is a longitudinal cohort study African American, Hispanic, Asian, and white adults in the United States. Because of its population characteristics, baseline plasma phospholipid fatty acid (FA) measurements, and HF outcomes, we used this cohort to determine whether higher levels of EPA predict reduced risk for HF. Our goal was to test the following hypotheses: 1) in humans, plasma %EPA is inversely associated with all HF incidence; 2) high plasma %EPA is inversely associated with incidence of HFpEF; and 3) the inverse association of high plasma %EPA with HF incidence is unique among ω3 PUFAs.
Results
Participant baseline demographics
The inclusion and exclusion of study participants for our analysis are outlined in Figure 1. Of the 6,814 total MESA participants, 6,562 (96%) were included. During a median follow-up period of 4,774 days (13.0 years), 292 participants had positive HF evaluations, including 128 with HFrEF, 110 with HFpEF, and 54 with unknown ejection fraction status. Baseline descriptive characteristics for each group are reported in Table 1. Details of post hoc tests are in Online Table 1, and details for differences among ethnic groups are in Online Table 2. HF-free participants had higher baseline plasma phospholipid %EPA, %DHA, and %EPA plus %DHA. Age, body mass index, diabetes mellitus, glomerular filtration rate, hypertension, use of a loop diuretic agent, urine microalbuminuria, and use of an angiotensin-converting enzyme inhibitor were significantly different (p < 0.05) between participants with and those without HF.
Participant plasma phospholipid EPA levels
We first evaluated the distribution of %EPA (Figure 2A, Online Table 3). Median %EPA was 0.70% for all MESA participants. Participant %EPA status was defined as insufficient (<1.0%), marginal (≥1.0% and ≤2.5%), or sufficient (>2.5%) to prevent HF on the basis of prior definition of EPA levels that prevent HF in animal models (13); 73.1% of participants had insufficient plasma EPA, 2.4% had marginal levels, and 4.5% had sufficient levels (Figure 2A). Hispanic participants had the lowest %EPA levels, with only 1.4% having sufficient EPA (Figure 2B), followed by black (4.4%), white (4.9%), and finally Chinese (9.8%) participants. Variance among Chinese participants was greater than it was among the other 3 races, which were each nearly identical. We evaluated the distribution of other PUFAs (Online Figure 1). Plasma %EPA was highly skewed, proportion ω3 DPA and %DHA were moderately skewed, and proportion arachidonic acid (AA) was least skewed; all fit better to a lognormal distribution than a normal distribution (Online Table 3).
FA clustering
Twenty-five FAs were measured in all participants. Strong collinearities existed among %FA, making it difficult to distinguish between direct and replacement effects on FA levels. Plasma %EPA and %DHA were strongly correlated because they co-occur in food products; both typically replace other PUFAs such as AA, hence increased %EPA can also report the replacement of AA. Eight FA clusters were identified (Online Table 4), explaining 63% of the total variability. The marine ω3 PUFAs clustered together with EPA as the most representative. The remaining PUFAs clustered together with AA as the lead PUFA. Lead FAs in each cluster were used as adjusters in developing hazard models, which allowed us to preserve interpretability and avoid collinearity.
Proportional hazard models for EPA and other ω3 PUFAs
We tested for associations of EPA with HF risk using a 4-step approach: 1) testing a univariate association; 2) adjusting for age, sex, race, and study center; 3) adjusting for other lead FAs; and 4) adjusting for other known risk factors. This approach allowed us to evaluate the independence and robustness of associations. At each step we performed sensitivity analyses in which we included only participants with HFrEF or HFpEF in order to evaluate the associations by HF type. In step 1, high plasma phospholipid %EPA at visit 1 was associated with reduced HF risk, regardless of HF type. Figure 3A shows the HR (95% confidence interval [CI]) from proportional hazard models for log %EPA with each successive adjustment block. Sensitivity analyses demonstrated that the association was not dependent on HF type. For step 2, we adjusted for age, sex, race, and study center. %EPA remained significantly associated with risk, without any change in the strength of association, indicating that age, sex, race, and study center did not mediate the association. Each successive model included these adjusters. In step 3, FA cluster leads were entered, and backward stepwise selection eliminated those unrelated to risk. Only proportion behenic acid was selected, but it did not change the association of %EPA. In step 4, all other HF-related adjusters were entered into the model and subjected to backward selection: body mass index, heart rate, fasting glucose, use of hypertensive medication, smoking status, pulse pressure, and urinary albuminuria status were selected; systolic blood pressure, non–high-density lipoprotein cholesterol, log triglycerides, and diabetes mellitus status were not selected. The final adjusted HR was a 0.73-fold reduction (95% CI: 0.60 to 0.91; p = 0.004) in risk per unit increase in log %EPA, which was not dependent on HF type. In summary, %EPA was inversely related to HF risk, the relationship was robust and independent to adjustment, and this association was present among all participants, including those with HFrEF and those with HFpEF (see Online Table 5).
To evaluate associations with other ω3 PUFAs, we used the same 4-step approach as for %EPA, with sensitivity analyses. As univariate predictors, %DHA and %EPA plus %DHA were both associated with reduced risk for HF; proportion ω3 DPA was not. The HRs (95% CIs) and sensitivity analyses for each are shown in Figures 3B to 3D (see also Online Table 5). After step 2, proportion ω3 DPA became significant. Adjustment in step 3 again showed that only the cluster represented by proportion behenic acid was significantly associated, but again, the ω3-PUFA HRs were unchanged and independent. Final adjustment not did not substantially alter the HRs, indicating that other proportion ω3 PUFAs are also significant and independent predictors of HF risk. The final adjusted HR for %DHA was 0.51 (95% CI: 0.38 to 0.70; p < 0.0001); for proportion ω3 DPA, it was 0.59 (95% CI: 0.37 to 0.95; p = 0.03); and for %EPA plus %DHA, it was 0.54 (95% CI: 0.39 to 0.73). Across all ω3 PUFAs, sensitivity analyses showed no substantial differences when only participants with HFrEF or only those with HFpEF were included. Because HRs derived from log-transformed predictors are difficult to interpret, Figure 4⇓ plots hazards for all HF relative to median across the entire observed PUFA ranges.
|
|
| |
| |
|
|
|