| |
HIV Prevention Trials Network (HPTN) Announces Initiation of HPTN 084: First Large-Scale Study in Women of a Long-Acting Injectable to Prevent HIV
|
| |
| |
Nov 30, 2017
HPTN 084
A Phase 3 Double Blind Safety and Efficacy Study of Long-Acting Injectable Cabotegravir Compared to Daily Oral TDF/FTC for Pre-Exposure Prophylaxis in HIV-Uninfected Women.
https://www.hptn.org/research/studies/hptn084
DURHAM, N.C. - The HIV Prevention Trials Network (HPTN) today announced the initiation of HPTN 084, a Phase 3 double-blind safety and efficacy study of long-acting injectable cabotegravir (CAB) compared to a combination of daily oral tenofovir disoproxil fumarate 300 mg plus emtricitabine 200 mg (TDF/FTC). The study is the first large-scale clinical trial of a long-acting injectable medication for HIV prevention in sexually active women. If found to be safe and effective for HIV pre-exposure prophylaxis (PrEP), injectable CAB may be an easier, more desirable, discreet alternative to daily oral TDF/FTC for some women. Several countries have approved TDF/FTC for PrEP including Australia, Canada, France, Kenya, Peru, South Africa, and the U.S.
"The development of safe alternative options for PrEP could increase HIV prevention choices for women," said Sinead Delany-Moretlwe, MBBCh, Ph.D., DTM&H, HPTN 084 protocol chair and research director at RHI, University of the Witwatersrand in Johannesburg, South Africa. "Long-acting injectable agents like cabotegravir may be more acceptable to some women for HIV prevention by providing an alternative with different adherence requirements."
HPTN 084 will enroll approximately 3,200 HIV-uninfected, sexually active women in Botswana, Kenya, Malawi, Swaziland, South Africa, Uganda, and Zimbabwe. Study participants will be assigned by chance to either CAB or TDF/FTC in a double-blind manner, meaning neither the participant nor research staff know what the participant is receiving. Study participants will be transitioned to locally available HIV prevention services when their participation in the study ends.
"In sub-Saharan Africa, more than 60 percent of all people living with HIV are women," said Wafaa El-Sadr M.D., M.P.H., HPTN co-principal investigator and professor of epidemiology and medicine at Columbia University. "Young African women share a disproportionate burden and are especially vulnerable to this epidemic."
A related study, HPTN 083, is testing the safety and efficacy of injectable CAB for HIV prevention in 4,500 men who have sex with men and transgender women who have sex with men in seven countries in the Americas, Asia and Africa.
"If cabotegravir is shown to protect against HIV acquisition, the long-acting injectable agent could become another major tool in our HIV prevention toolbox," said Myron Cohen, M.D., HPTN co-principal investigator and director of the Institute for Global Health and Infectious Diseases at the University of North Carolina at Chapel Hill.
HPTN 084 is jointly funded through a unique partnership of the U.S. National Institute of Allergy and Infectious Diseases, part of the U.S. National Institutes of Health; the Bill &
Melinda Gates Foundation; and ViiV Healthcare. Study drugs are provided by ViiV Healthcare and Gilead Sciences, Inc.
For more information about HPTN 084, visit hptn.org, or ClinicalTrials.gov using study identifier NCT03164564.
What is HPTN 084? https://www.hptn.org/research/studies/hptn084
HPTN 084 is a study being done to to evaluate the safety and efficacy of the injectable agent, cabotegravir (CAB LA) compared to daily oral tenofovir disoproxil fumarate/emtricitabine (TDF/FTC), for pre-exposure prophylaxis (PrEP) in HIV-uninfected women.
Who is participating in the study?

HPTN 084 will enroll approximately 3,200 women 18 to 45 years old in sub-Saharan Africa who are at risk for acquiring HIV.
Why is HPTN 084 important?
PrEP agents are needed that do not depend on daily or near-daily pill-taking. The development of alternative agents for PrEP, and/or more adherence-friendly schedules for currently available agents, could increase prevention choices and increase acceptability. Long-acting injectable agents have the potential to prevent HIV acquisition without relying on adherence to a daily oral regimen.
What happens during the study?
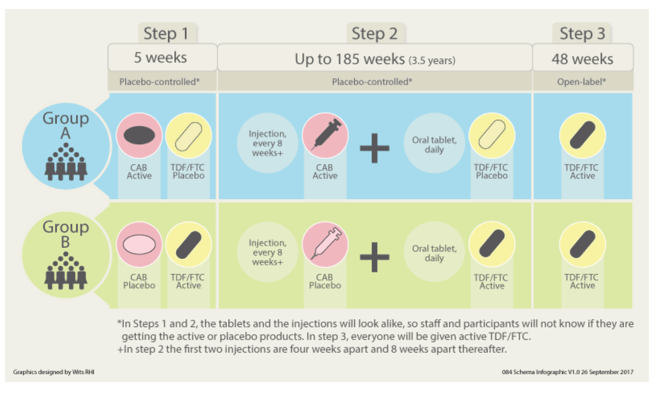
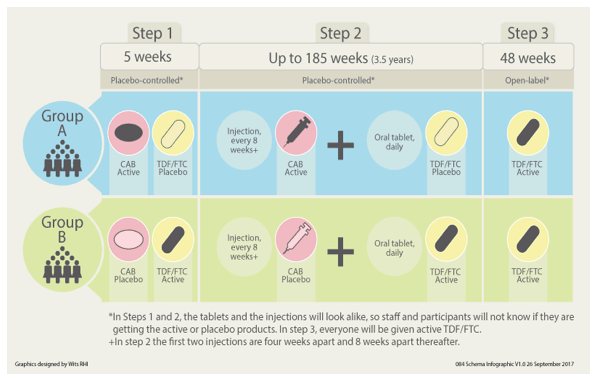
Once randomized to one of two arms, participants move through the steps below and are followed for up to 4 and a half years (active drugs are shown in bold text):
Step 1:
Arm A - Daily oral CAB (30 mg tablets) and oral TDF/FTC placebo for five weeks plus an HIV prevention package including behavioral risk reduction and adherence counseling, provision of condoms.
Arm B - Daily oral TDF/FTC (300 mg/200 mg fixed-dose combination tablets) and oral CAB placebo for five weeks plus an HIV prevention package including behavioral risk reduction and adherence counseling, provision of condoms.
A participant that becomes HIV-infected during Step 1 of the study will permanently discontinue study product and will be terminated from the study, and referred for HIV-related care.
Step 2:
Arm A - CAB LA (600 mg as a single intramuscular [IM] injection at two time points 4 weeks apart and every 8 weeks thereafter) and daily oral TDF/FTC placebo plus an HIV prevention package including behavioral risk reduction and adherence counseling, provision of condoms.
Arm B - Daily oral TDF/FTC (300/200 mg fixed-dose combination tablets) and IM placebo at two time points 4 weeks apart and every 8 weeks thereafter (matching vehicle, identical volume as active injectable product in Arm A) plus an HIV prevention package including behavioral risk reduction and adherence counseling, provision of condoms.
This step will continue until the required number of endpoints (111) is reached, estimated to be 81 weeks after enrolling the last participant.
A participant that becomes HIV-infected during Step 2 of the study will permanently discontinue study product, be placed on immediate suppressive ART, and be followed for 52 weeks after their last injection, after which their participation in the study will end and they will be transitioned to continued HIV-related care.
Step 3:
Arms A and B - Open-label daily TDF/FTC (in order to cover the pharmacokinetic [PK] tail for Arm A participants) will be provided no later than eight weeks after the last injection visit, for up to 48 weeks plus an HIV prevention package including behavioral risk reduction and adherence counseling, and provision of condoms. Participants will then transition to locally available HIV prevention services, including services for PrEP, if available.
A participant with confirmed HIV infection during Step 3 will be followed at least for the duration of Step 3.
Study Documents
HPTN 084 Version 1.0
• HPTN 084 Protocol V1.0 - 2 March 2017
• HPTN 084 LoA #1 - 15 August 2017
• HPTN 084 LoA #2 - 24 January 2018
• HPTN 084 LoA #3 - 31 May 2018
• HPTN 084 CM #1 - 11 May 2017
• HPTN 084 CM #2 - 26 Sept. 2017
• HPTN 084 FAQ
Other
• Tips and Lessons Learned
Press
• HIV Prevention Trials Network (HPTN) Announces Initiation of HPTN 084: First Large-Scale Study in Women of a Long-Acting Injectable to Prevent HIV
About HPTN
The HIV Prevention Trials Network (HPTN) is a worldwide collaborative clinical trials network that brings together investigators, ethicists, community and other partners to develop and test the safety and efficacy of interventions designed to prevent the acquisition and transmission of HIV. HPTN studies evaluate new HIV prevention interventions and strategies in populations and geographical regions that bear a disproportionate burden of infection. The HPTN research agenda is focused primarily on the use of integrated strategies: use of antiretroviral drugs (antiretroviral therapy and pre-exposure prophylaxis); interventions for substance abuse, particularly injection drug use; behavioral risk reduction interventions and structural interventions. NIH funds HPTN. For more information, visit www.hptn.org.
Media Contact
Kevin Bokoch
kbokoch@fhi360.org
+1.440.376.1901
|
|
| |
| |
|
|
|