| |
Association Between HIV Infection and the Risk of Heart Failure With Reduced Ejection Fraction and Preserved Ejection Fraction in the Antiretroviral Therapy Era - Results From the Veterans Aging Cohort Study
|
| |
| |
Download the PDF here
Download the PDF here
EDITORIAL: both young and old appear to be at risk and HFrEF is the major HF manifestation among those younger than 40 years old. These individuals, now into their fifth and sixth decades, represent a vulnerable group for which HFrEF has received scant attention........From a public health standpoint, more attention should be paid to heighten awareness of the comorbidity of HIV and HFrEF in the aging HIV-positive population and to design approaches to incorporate HF screening, risk estimation, and treatment. Considering that by 2020 more than 50% of all PLWH will be older than 50 years old,6 there is a critical need to design and implement effective HF prevention strategies in this population. Second, immune system function is an important determinant of HF in HIV. This study supports prior epidemiological research9 that shows that restoring immune system function, as measured by CD4+ cell count, decreases HF risk. Thus, while we await empirical evidence, it is reasonable to initiate therapies early in the disease course to mitigate HF and total cardiovascular disease risk as some have suggested. The elephant in the room, which this study was not designed to address, is the mechanism by which HIV infection and its treatment impact HF risk. The HF risk identified in this study appears to be independent of hypertension and myocardial infarction, which are the 2 leading determinants of HF risk in the general population. The extent to which this risk is driven by direct effects on the myocardium vs more systemic effects (such as inflammation or vascular injury) are uncertain. Can preclinical abnormalities be detected in PLWH using imaging (eg, myocardial strain imaging and diastolic dysfunction) or circulating biomarkers that predict incident HF? Which ART regimens are most “cardiac friendly”? Is the HFrEF seen in the group younger than 40 years old reversible with appropriate HF and HIV therapy? Last, treatment patterns for HF among PLWH remain poor.13 Effective approaches to improve HF medication use patterns for this group have also not been demonstrated. On these issues, there is great need and opportunity for further knowledge generation.
..........Of the 2636 HF events, 941 were in PLWH and HFrEF was the single most common type (40%). Human immunodeficiency virus infection conferred a 41% increased risk (95% CI, 29%-54%) for total HF, with a stronger risk for developing HFrEF (HR, 1.61; 95% CI, 1.40-1.86) than HFpEF (HR, 1.21; 95% CI, 1.03-1.41). Risk for HF was greater among PLWH despite having a lower burden of traditional HF risk factors, including hypertension, diabetes, high low-density lipoprotein cholesterol level, and obesity. In subgroup analyses adjusted for numerous clinical variables, the HF risk was driven mainly by HFrEF for white, black, and young (<40 years of age) veterans. Consistently low CD4+ cell counts significantly increased the risk for HFrEF (HR, 1.87; 95% CI, 1.36-2.57) and HFpEF (HR, 1.87; 95% CI, 1.28-2.73), but CD4+ cell count greater than 500 cells/mm3 at baseline also portended greater HF risk (HR, 1.25; 95% CI, 1.08-1.43). Consistently detectable viremia increased the risk for HFrEF (HR, 1.63; 95% CI, 1.28-2.08), but not HFpEF. In a robust set of sensitivity analyses, the risk for HF attributed to HIV persisted after restricting the data set to those without hypertension, alcohol or cocaine abuse, and never smokers and adjusted for incident myocardial infarction.
-----------------------------
May 2017
Association Between HIV Infection and the Risk of Heart Failure With Reduced Ejection Fraction and Preserved Ejection Fraction in the Antiretroviral Therapy Era - Results From the Veterans Aging Cohort Study
Matthew S. Freiberg, MD, MSc; Chung-Chou H. Chang, PhD; Melissa Skanderson, MSW; Olga V. Patterson, PhD; Scott L. DuVall, PhD;
Cynthia A. Brandt,MD, MPH; Kaku A. So-Armah, PhD; Ramachandran S. Vasan, MD; Kris Ann Oursler,MD, ScM; John Gottdiener, MD;
Stephen Gottlieb, MD; David Leaf,MD, MPH; Maria Rodriguez-Barradas, MD; Russell P. Tracy, PhD; Cynthia L. Gibert, MD, MS;
David Rimland, MD; Roger J. Bedimo,MD, MS; Sheldon T. Brown, MD; Matthew Bidwell Goetz, MD; AlbertaWarner, MD; Kristina Crothers, MD;
Hilary A. Tindle,MD, MPH; Charles Alcorn, MA; Justin M. Bachmann, MD, MPH; Amy C. Justice,MD, PhD; Adeel A. Butt, MD,MS
Given these facts, health care professionals should focus on guideline-recommended HIV treatment and HF risk factor prevention (including diabetes, hypertension, renal disease, smoking, alcohol abuse and dependence, and obesity), as well as screening for HIV in individuals with new-onset HF where appropriate.25 Developing tools designed to risk stratify HIV-infected individuals for HF will also be required.
While the exact mechanisms underlying the association between HIV and types of HF remain unclear, the fact that time-updated low CD4 cell count was associated with HFrEF and HFpEF suggests that duration of HIV infection and, by extension, chronic inflammation, T-cell activation, and loss of adaptive immunity likely all have important roles. Individuals who are infected with HIV with low CD4 cell counts have increased levels of immune activation and inflammation,34 which are associated with an increased HF risk.35 Most important, our data also suggest that even HIV-infected individuals with high CD4 cell counts are likely still at risk for HF compared with uninfected individuals, in part because HIV-infected individuals with high CD4 cell counts who are rapidly diagnosed, treated, and virally suppressed do not return to their pre-HIV levels of inflammation.37 Moreover, this residual inflammation is associated with an increased risk of future non-AIDS diseases.37 In contrast, time-updated elevated HIV-1 RNA viral load was only significantly associated with HFrEF. These findings are consistent with reports before the ART era in which unsuppressed HIV viremia, perhaps through direct infection of cardiac myocytes38,39 or cardiac autoantibodies,40 results in a cardiomyopathy consistent with HFrEF.41 The role of ART in the development of HF is less clear. Cardiac mitochondrial toxic effects in the highly active ART era is well documented.42 In the present study, baseline ART use was associated with an increased HFpEF risk, whereas our time-updated data suggested that successful ART as measured by lower HIV-1 RNA viral load and higher CD4 cell count reduces the risk of HFrEF and HFpEF. As prior studies have shown, ART can simultaneously lower AMI risk through viral suppression43 and increase AMI risk likely through medication adverse effects.44 Therefore, determining if newer ART medications have a role in the development of HF should be explored because many HIV-infected individuals will be receiving ART medications for decades.
In the VACS, HIV-infected veterans had an increased risk of HFpEF, borderline HFpEF, and HFrEF. Among HIV-infected veterans, time-updated HIV-1 RNA viral load of at least 500 copies/mL compared with less than 500 copies/mL was associated with an increased risk of HFrEF, whereas time-updated CD4 cell count less than 200 cells/mm3 compared with at least 500 cells/mm3 was associated with an increased risk of total HF, HFpEF, borderline HFpEF, and HFrEF......we show herein that the risk of HFrEF extends beyond AMI, is present across multiple decades of age groups, and occurs among individuals of black or white race/ethnicity, those without decades-long exposure to HF risk factors, and those with high HIV-1 RNA viral load and low CD4 cell count over time.
Key Points
Question Does HIV infection increase the risk of heart failure with reduced ejection fraction, heart failure with preserved ejection fraction, or both, and do these risks vary by age, race/ethnicity, HIV–specific biomarkers, and receipt of antiretroviral therapy?
Findings In this cohort study of 98 015 veterans, individuals with HIV infection had a 61% increased risk of heart failure with reduced ejection fraction (ejection fraction <40%), a 21% increased risk of heart failure with preserved ejection fraction (ejection fraction ≥50%), and a 37% increased risk of borderline heart failure with preserved ejection fraction (ejection fraction 40%-49%) compared with uninfected veterans. These risks are significant, even after adjusting for possible confounders, and the association between HIV infection and types of heart failure varies by age, race/ethnicity, HIV–specific biomarkers, and receipt of antiretroviral therapy.
Meaning A strategy that encompasses HIV infection treatment, heart failure risk factor prevention and management, and the development of heart failure risk stratification tools would be beneficial for this high-risk population.
Abstract
Importance With improved survival, heart failure (HF) has become a major complication for individuals with human immunodeficiency virus (HIV) infection. It is unclear if this risk extends to different types of HF in the antiretroviral therapy (ART) era. Determining whether HIV infection is associated with HF with reduced ejection fraction (HFrEF), HF with preserved ejection fraction (HFpEF), or both is critical because HF types differ with respect to underlying mechanism, treatment, and prognosis.
Objectives To investigate whether HIV infection increases the risk of future HFrEF and HFpEF and to assess if this risk varies by sociodemographic and HIV-specific factors.
Design, Setting, and Participants This study evaluated 98 015 participants without baseline cardiovascular disease from the Veterans Aging Cohort Study, an observational cohort of HIV-infected veterans and uninfected veterans matched by age, sex, race/ethnicity, and clinical site, enrolled on or after April 1, 2003, and followed up through September 30, 2012. The dates of the analysis were October 2015 to November 2016.
Exposure Human immunodeficiency virus infection.
Main Outcomes and Measures Outcomes included HFpEF (EF≥50%), borderline HFpEF (EF 40%-49%), HFrEF (EF<40%), and HF of unknown type (EF missing).
Results Among 98 015 participants, the mean (SD) age at enrollment in the study was 48.3 (9.8) years, 97.0% were male, and 32.2% had HIV infection. During a median follow-up of 7.1 years, there were 2636 total HF events (34.6% were HFpEF, 15.5% were borderline HFpEF, 37.1% were HFrEF, and 12.8% were HF of unknown type). Compared with uninfected veterans, HIV-infected veterans had an increased risk of HFpEF (hazard ratio [HR], 1.21; 95% CI, 1.03-1.41), borderline HFpEF (HR, 1.37; 95% CI, 1.09-1.72), and HFrEF (HR, 1.61; 95% CI, 1.40-1.86). The risk of HFrEF was pronounced in veterans younger than 40 years at baseline (HR, 3.59; 95% CI, 1.95-6.58). Among HIV-infected veterans, time-updated HIV-1 RNA viral load of at least 500 copies/mL compared with less than 500 copies/mL was associated with an increased risk of HFrEF, and time-updated CD4 cell count less than 200 cells/mm3 compared with at least 500 cells/mm3 was associated with an increased risk of HFrEF and HFpEF.
Conclusions and Relevance Individuals who are infected with HIV have an increased risk of HFpEF, borderline HFpEF, and HFrEF compared with uninfected individuals. The increased risk of HFrEF can manifest decades earlier than would be expected in a typical uninfected population. Future research should focus on prevention, risk stratification, and identification of the mechanisms for HFrEF and HFpEF in the HIV-infected population.
During a median follow-up of 7.1 years, there were 2636 total HF events. Of these events, 35.7% occurred in HIV-infected veterans (34.6% were HFpEF, 15.5% were borderline HFpEF, 37.1% were HFrEF, and 12.8% were HF of unknown type). Compared with uninfected veterans, HIV-infected veterans had higher rates of total HF and HFrEF but not HFpEF and borderline HFpEF (Table 2). Similar results were observed when rates were stratified by HIV infection status and age group categories except among those 70 years or older.
Compared with uninfected veterans, HIV-infected veterans had a significantly increased risk of total HF, HFpEF, borderline HFpEF, and HFrEF after adjusting for possible confounders (Table 3). In sensitivity analyses that included non-VA HF events and VA EF data, the association between HIV and total HF, HFpEF, borderline HFpEF, and HFrEF remained essentially unchanged (eTable in the Supplement). Similarly, the association between HIV and HF held when we restricted the sample to those without hypertension (HR, 1.32; 95% CI, 1.08-1.61), individuals without alcohol or cocaine abuse or dependence (HR, 1.43; 95% CI, 1.25-1.65), and never smokers (HR, 1.33; 95% CI, 1.05-1.70). The association between HIV infection and HFrEF persisted after further adjustment for incident AMI during the follow-up period (HR, 1.58; 95% CI, 1.37-1.82).
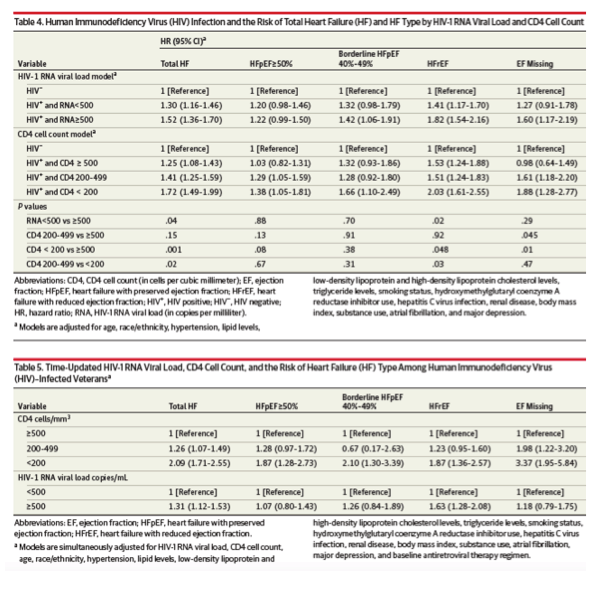
Among the younger veterans (<40 years at baseline) and individuals of white or black race/ethnicity, HIV infection was significantly associated with an increase in total HF and HFrEF but not HFpEF or borderline HFpEF (Table 3). When we compared uninfected veterans with HIV-infected veterans stratified by HIV-specific biomarkers, the risk of HFrEF persisted even among HIV-infected veterans with a baseline HIV-1 RNA viral load less than 500 copies/mL compared with uninfected veterans (HR, 1.41; 95% CI, 1.17-1.70) (Table 4).
When we restricted the sample to only HIV-infected veterans and adjusted for covariates, including baseline HIV-1 RNA viral load and CD4 cell count, baseline NRTI plus PI (HR, 1.80; 95% CI, 1.19-2.71), NRTI plus NNRTI (HR, 1.48; 95% CI, 1.01-2.15), and other (HR, 3.46; 95% CI, 1.79-6.72) compared with no ART were associated with an increased risk of HFpEF but not HFrEF. In time-updated analyses, CD4 cell count less than 200 cells/mm3 was associated with an increased risk of total HF, HFpEF, borderline HFpEF, and HFrEF (Table 5), whereas time-updated HIV-1 RNA viral load of at least 500 copies/mL was only associated with HFrEF.
|
|
| |
| |
|
|
|