 |
 |
 |
| |
IAS 2019: ViiV Healthcare showcasing
innovation in HIV science - PRESS RELEASE
|
| |
| |
Download the PDF here
NOTE TO EDITORS: ViiV Healthcare will hold a press conference at IAS 2019 on Tuesday 23 July, 9:00 - 9:45 am CDT to preview abstracts to be presented during the conference. To register, please visit: http://bit.ly/IAS-ViiV
Data to be presented span the company's diverse portfolio, challenging the current treatment paradigm and investigating new options to meet the evolving needs of people living with HIV
THIS is EXCERPTED FROM PRESS RELEASE, to SEE FULL PRESS RELEASE see this LINK or pdf attached:
https://www.gsk.com/en-gb/media/press-releases/ias-2019-viiv-healthcare-showcasing-innovation-in-hiv-science/
London, 15 July 2019 - ViiV Healthcare, the global specialist HIV company majority-owned by GSK, with Pfizer Inc. and Shionogi Limited as shareholders, announced today that 20 abstracts from its portfolio of late-stage pipeline and authorised HIV treatments will be presented at the 10th International AIDS Society Conference on HIV Science (IAS 2019) in Mexico City, 21-24 July, in Mexico City, Mexico.
Since its inception 10 years ago, as the only pharmaceutical company solely focused on HIV, ViiV Healthcare continues its industry-leading commitment by delivering scientific advances that address the needs of the HIV community. The data presented at IAS 2019 further build upon the company's innovative approach to research and development by investigating treatments that have the potential to reduce the number of medicines people living with HIV take during their lifetime and provide a range of options that meet their diverse and evolving needs.
Highlights at IAS 2019 include the presentation of safety and efficacy data for the 2-drug regimen (2DR) of dolutegravir plus lamivudine in treatment-naïve and treatment-experienced patients; longer-term clinical trial data of investigational fostemsavir in heavily treatment-experienced patients; and pooled clinical trial data and patient-reported outcomes from the investigational long-acting, injectable 2DR of cabotegravir plus rilpivirine.
John C. Pottage, Jr., M.D., Chief Scientific Officer and Medical Officer, ViiV Healthcare, said: "The depth and focus of our data at IAS 2019 is reflective of ViiV Healthcare's patient-centred approach to innovation. We have made significant advances in the development of 2-drug treatment regimens and we are looking forward to presenting additional data regarding this new treatment paradigm. We are also looking forward to presenting long-term data for a new first in class therapy for people with few or no treatment options. With our pipeline of innovative medicines, we are aiming to make a difference in the lives of people living with HIV."
Key ViiV Healthcare abstracts to be presented at IAS 2019
• Week 96 data for the 2DR of dolutegravir plus lamivudine in treatment-naïve patients (GEMINI 1 & 2): The GEMINI 1 & 2 phase III studies compared a 2DR of dolutegravir plus lamivudine to a 3-drug regimen of dolutegravir plus the fixed-dose tablet tenofovir/emtricitabine (TDF/FTC). Week 48 results, which included the primary endpoint, were presented at the 2018 International AIDS Conference.[i] The data presented at IAS 2019 will evaluate the efficacy of dolutegravir plus lamivudine compared to the 3-drug regimen at the Week 96 secondary endpoint.[ii] Dovato has been authorised in the EU[iii] and US[iv] as a once-daily, single-tablet regimen, with further regulatory marketing applications submitted worldwide.
• Week 48 data for the 2DR of dolutegravir plus lamivudine in treatment-experienced patients (TANGO): TANGO is a phase III, randomised, open-label, active-controlled, multicentre, parallel-group study comparing dolutegravir plus lamivudine once daily against continuation of a ≥3-drug tenofovir alafenamide (TAF)-based regimen over 48-weeks in HIV-1 infected, antiretroviral treatment-experienced, virally suppressed subjects.[v] Headline results for the primary endpoint, efficacy at Week 48, were recently announced. The detailed results from the TANGO Week 48 data will be presented at IAS 2019.
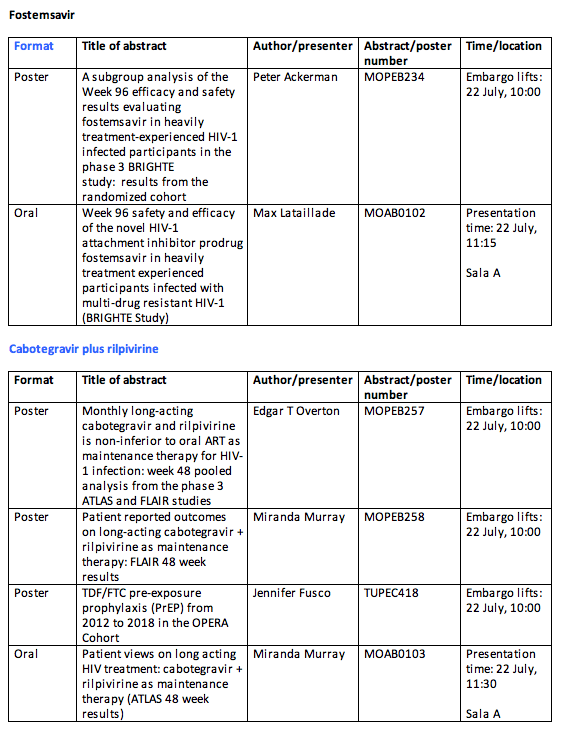
• Week 96 data for fostemsavir in heavily treatment-experienced patients (BRIGHTE):
The BRIGHTE study is a two-cohort (randomised and non-randomised), phase III clinical trial evaluating the safety and efficacy of the HIV-1 attachment inhibitor fostemsavir in heavily treatment-experienced adults with HIV-1 infection. Results from the primary endpoint, mean change in log10 HIV-1 RNA between day 1 and day 8 for the randomised cohort, and at Week 48 have been previously announced.[vi] The Week 96 data presented at IAS will offer further insight into the efficacy of fostemsavir over a longer period.[vii]
• Clinical trial data and patient-reported outcomes on the long-acting injectable regimen of ViiV Healthcare's cabotegravir plus Janssen's rilpivirine (ATLAS and FLAIR):
Week 48 pooled analysis of the phase III ATLAS and FLAIR studies, as well as patient-reported views and outcomes from both studies, will be presented at IAS 2019.[viii],[ix],[x] In ATLAS and FLAIR, patients were asked to report their treatment satisfaction (HIV-Treatment Satisfaction Questionnaire), acceptability of treatment (general acceptance domain of ACCEPT) and health status (SF-12), as well as tolerability and acceptability of the cabotegravir and rilpivirine injections (Perception of Injections (PIN)). The findings presented at IAS expand on patient preference data presented at the 2019 Conference on Retroviruses and Opportunistic Infections.[xi],[xii] Development of this regimen is a collaboration with the Janssen Pharmaceutical Companies of Johnson & Johnson.
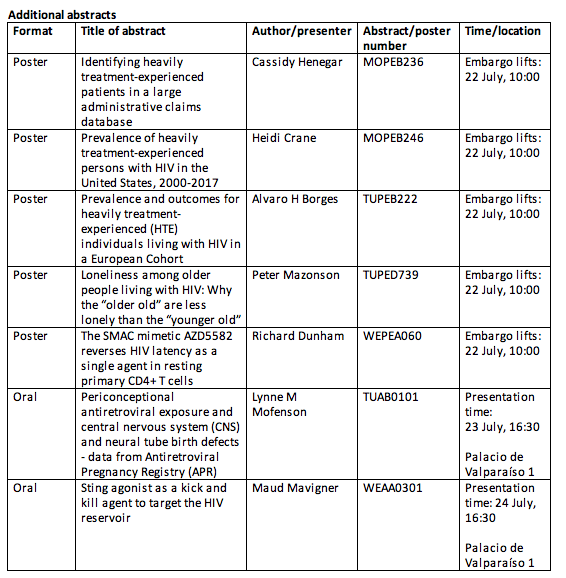
The full list of data that will be presented by ViiV Healthcare at IAS 2019 is listed below:
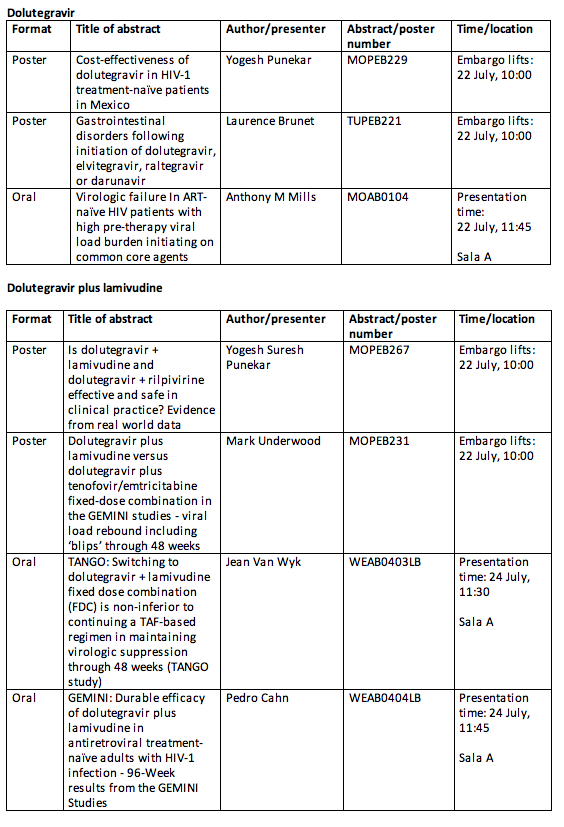
About dolutegravir plus lamivudine
Dolutegravir is an integrase inhibitor (INI) for use in combination with other antiretroviral agents for the treatment of HIV.[xiii] INIs block HIV replication by preventing the viral DNA from integrating into the genetic material of human immune cells (T-cells). This step is essential in the HIV replication cycle and is also responsible for establishing chronic infection. Dolutegravir is authorised in over 100 countries across North America, Europe, Asia, Australia, Africa and Latin America.
Lamivudine, commonly known as 3TC, is a nucleoside analogue used in combination with other antiretroviral agents for the treatment of HIV infection. Lamivudine is available in branded (Epivir®, 300mg) and generic forms.[xiv]
Dolutegravir plus lamivudine (Dovato) is a once-daily, single-pill, 2-drug regimen that combines the INI dolutegravir (Tivicay, 50 mg)13,[xv]with the nucleoside analogue reverse transcriptase inhibitor (NRTI) lamivudine (Epivir, 300 mg).4 It is authorised in the EU for the treatment of HIV-1 infection in adults and adolescents above 12 years of age weighing at least 40 kg, with no known or suspected resistance to the integrase inhibitor class, or lamivudine,3 and in the US for the treatment of HIV-1 infection in adults with no antiretroviral treatment history and with no known resistance to either dolutegravir or lamivudine.4
Trademarks are owned by or licensed to the ViiV Healthcare group of companies.
Important Safety Information
Important Safety Information for Dovato (50mg dolutegravir/300mg lamivudine) tablets in the EU
The following Important Safety Information is based on the Summary of Product Characteristics for Dovato. Please consult the full Summary of Product Characteristics for all the safety information.
Dovato (50mg dolutegravir/300mg lamivudine)
Dovato is indicated for the treatment of Human Immunodeficiency Virus type 1 (HIV-1) infection in adults and adolescents above 12 years of age weighing at least 40 kg, with no known or suspected resistance to the integrase inhibitor class, or lamivudine.
The recommended dose of Dovato in adults and adolescents is one 50 mg/300 mg tablet once daily.
Method of administration
Oral use. Dovato can be taken with or without food.
Contraindications
Hypersensitivity to the active substances or to any of the excipients listed in section 6.1.
Dose adjustments
A separate preparation of dolutegravir is available where a dose adjustment is indicated due to drug-drug interactions (e.g. rifampicin, carbamazepine, oxcarbazepine, phenytoin, phenobarbital, St. John's wort, etravirine (without boosted protease inhibitors), efavirenz, nevirapine, or tipranavir/ritonavir. In these cases the physician should refer to the individual product information for dolutegravir.
Missed doses
If the patient misses a dose of Dovato, the patient should take Dovato as soon as possible, providing the next dose is not due within 4 hours. If the next dose is due within 4 hours, the patient should not take the missed dose and simply resume the usual dosing schedule.
Please refer to the full European Summary of Product Characteristics for dolutegravir/lamivudine
for full prescribing information, including contraindications, special warnings and precautions for use. For the US, please refer to the US Prescribing Information.
About fostemsavir
Fostemsavir is an investigational prodrug of temsavir, an HIV-1 attachment inhibitor class, and is not authorised by regulatory authorities anywhere in the world. Fostemsavir is being developed by ViiV Healthcare for the treatment of HIV-1-infected heavily treatment-experienced patients in combination with other antiretroviral agents.
About cabotegravir
Cabotegravir is an investigational integrase inhibitor (INI) and is not authorised by regulatory authorities anywhere in the world. Cabotegravir is being developed by ViiV Healthcare for the treatment and prevention of HIV. It is being evaluated as a long-acting formulation for intramuscular injection and also as a once-daily oral tablet for use as a lead-in, to establish the tolerability of cabotegravir prior to long-acting injection.
About rilpivirine
EDURANT® (rilpivirine) is a once daily non-nucleoside reverse transcriptase inhibitor (NNRTI) used for the treatment of human immunodeficiency virus (HIV-1) infection in combination with other antiretroviral agents in antiretroviral treatment-naïve adult patients with a viral load ≤ 100,000 HIV RNA copies/mL. Long-acting injectable rilpivirine is not authorised by regulatory authorities anywhere in the world.
Rilpivirine was developed by Janssen Sciences Ireland UC, one of the Janssen
Pharmaceutical Companies of Johnson & Johnson. Rilpivirine is authorised in the U.S. and E.U. as EDURANT® as a 25mg tablet taken once-a-day and is always taken with a meal.
The most common side effects of EDURANT include: depression, headache, trouble sleeping (insomnia) and rash.
Important Safety Information (ISI) for EDURANT® (Rilpivirine)
Note: this is taken from the US label and local variations apply. Please refer to applicable local labelling.
About EDURANT® (Rilpivirine)
• EDURANT® (rilpivirine) is a prescription medicine that is used with other antiretroviral medicines to treat Human Immunodeficiency Virus-1 (HIV-1) in people 12
years of age and older and who weigh at least 77 lbs (35 kg):
• Have never taken HIV medicines before, and
• Have an amount of HIV in their blood (called "viral load") that is no more than 100,000 copies/mL
• EDURANT® is not recommended for patients less than 12 years of age or who weigh less than 77 lbs (35 kg)
IMPORTANT SAFETY INFORMATION
Who should not take EDURANT®?
Do not take EDURANT® if you also take:
• anti-seizure medicines:
o carbamazepine
o oxcarbazepine
o phenobarbital
o phenytoin
• anti-tuberculosis (anti-TB) medicines:
o rifampin
o rifapentine
• proton pump inhibitor (PPI) medicine for certain stomach or intestinal problems:
o esomeprazole
o lansoprazole
o omeprazole
o pantoprazole sodium
o rabeprazole
• more than 1 dose of the steroid medicine dexamethasone or dexamethasone sodium phosphate
• St. John's wort (Hypericum perforatum)
You are encouraged to report side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088. You may also report side effects to Janssen Products, LP, at 1-800-JANSSEN (1-800-526-7736).
Please see accompanying full Product Information for more details.
Full US prescribing information including is available at:
http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/EDURANT-pi.pdf
For the EU Summary of Product Characteristics, please visit:
http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/002264/WC500118874.pdf
About ViiV Healthcare
ViiV Healthcare is a global specialist HIV company established in November 2009 by GlaxoSmithKline (LSE: GSK) and Pfizer (NYSE: PFE) dedicated to delivering advances in treatment and care for people living with HIV and for people who are at risk of becoming infected with HIV. Shionogi joined in October 2012. The company's aims to take a deeper and broader interest in HIV/AIDS than any company has done before and take a new approach to deliver effective and innovative medicines for HIV treatment and prevention, as well as support communities affected by HIV.
For more information on the company, its management, portfolio, pipeline, and commitment, please visit www.viivhealthcare.com.
About GSK
GSK is a science-led global healthcare company with a special purpose: to help people do more, feel better, live longer. For further information please visit www.gsk.com.
Cautionary statement regarding forward-looking statements
GSK cautions investors that any forward-looking statements or projections made by GSK, including those made in this announcement, are subject to risks and uncertainties that may cause actual results to differ materially from those projected. Such factors include, but are not limited to, those described under Item 3.D 'Principal risks and uncertainties' in the company's Annual Report on Form 20-F for 2018.
ViiV Healthcare media enquiries: Melinda Stubbee +1 919 491 0831
Patricia O'Connor +44 (0) 20 8047 5982
Audrey Abernathy +1 919 605 4521
GSK Global media enquiries: Simon Steel +44 (0) 20 8047 5502
Kristen Neese +1 804 217 8147
Analyst/Investor enquiries: Sarah Elton-Farr
+44 (0) 20 8047 5194
Danielle Smith +44 (0) 20 8047 0932
James Dodwell +44 (0) 20 8047 2406
Jeff McLaughlin +1 215 751 7002
References
[i] Cahn J, Sierra Madero J, Arribas J, et al. Non-inferior efficacy of dolutegravir (DTG) plus lamivudine (3TC) versus DTG plus tenofovir/emtricitabine (TDF/FTC) fixed-dose combination in antiretroviral treatment-naïve adults with HIV-1 infection - 48-week results from the GEMINI studies. Presented at the 22nd International AIDS Conference (AIDS 2018). Amsterdam, The Netherlands, 23rd-27th July 2018.
[ii] Cahn P, Sierra Madero J, Arribas J, et al. Durable efficacy of dolutegravir (DTG) plus lamivudine (3TC) in antiretroviral treatment-naïve adults with HIV-1 infection - 96-week results from the GEMINI studies. Planned presentation at the 10th International AIDS Conference on HIV Science (IAS 2019), 21-24th July 2019, Mexico City, Mexico.
[iii] Dovato EU Summary of Product Characteristics. July 2019. Available at: https://www.medicines.org.uk/emc/product/10446/smpc Accessed July 2019.
[iv] Dovato (dolutegravir plus lamivudine) Prescribing Information. U.S. Approval 8 April 2019.
[v] Clinical trials.gov Switch Study to Evaluate Dolutegravir Plus Lamivudine in Virologically Suppressed Human Immunodeficiency Virus Type 1 Positive Adults (TANGO). Available at: https://clinicaltrials.gov/ct2/show/NCT03446573?term=TANGO+dolutegravir&rank=1. Accessed July 2019
[vi] Aberg J, Molina J-M, Kozal M et al. Week 48 safety and efficacy of the HIV-1 attachment inhibitor prodrug fostemsavir in heavily treatment-experienced participants (BRIGHTE study). Abstract number 0334A. Presented at HIV Glasgow 2018.
[vii] Lataillade M, Lalezari J, Aberg J, et al. Week 96 safety and efficacy of the novel HIV-1 attachment inhibitor prodrug fostemsavir in heavily treatment experienced participants infected with multi-drug resistant HIV-1 (BRIGHTE Study). Planned presentation at the 10th International AIDS Conference on HIV Science (IAS 2019), 21-24th July 2019, Mexico City, Mexico.
[viii] Murray M, Antela A, Mills A, et al. Patient views on long acting HIV treatment: cabotegravir + rilpivirine as maintenance therapy (ATLAS 48 Week results). Planned presentation at the 10th International AIDS Conference on HIV Science (IAS 2019), 21-24th July 2019, Mexico City, Mexico.
[ix] Murray M, Bernal E, Chounta V, et al. Patient reported outcomes on long-acting cabotegravir + rilpivirine as maintenance therapy: FLAIR 48 Week results. Planned presentation at 10th International AIDS Conference on HIV Science (IAS 2019), 21-24th July 2019, Mexico City, Mexico.
[x] Overton ET, Orkin C, Swindell S, et al. Monthly long-acting cabotegravir and rilpivirine is non-inferior to oral ART as maintenance therapy for HIV-1 infection: Week 48 pooled analysis from the phase 3 ATLAS and FLAIR studies. Planned presentation at the 10th International AIDS Conference on HIV Science (IAS 2019), 21-24th July 2019, Mexico City, Mexico..
[xi] Swindells S, Andrade-Villnueva J-F, Richmond GJ, et al. Long-acting cabotegravir + rilpivirine as maintenance therapy: ATLAS Week 48 results. Abstract number 139. Presented at the Conference for Retroviruses and Opportunistic Infections (CROI 2019), Seattle, Washington, 4th-7th March 2019.
[xii] Orkin C, Arastéh K, Hernández-Mora MG, et al. Long-acting cabotegravir + rilpivirine for HIV maintenance: FLAIR Week 48 results. Abstract number 140. Presented at the Conference for Retroviruses and Opportunistic Infections (CROI 2019), Seattle, Washington, 4th-7th March 2019.
[xiii] Tivicay (dolutegravir) European Summary of Product Characteristics. Available at: https://www.ema.europa.eu/en/documents/product-information/tivicay-epar-product-information_en.pdf Last accessed July 2019.
[xiv] European Medicines Agency. Epivir (lamivudine) European Summary of Product Characteristics. Available at: https://www.ema.europa.eu/en/documents/product-information/epivir-epar-product-information_en.pdf
Last accessed July 2019.
[xv] Tivicay (dolutegravir) U.S. Prescribing Information. Available at https://www.gsksource.com/pharma/content/dam/GlaxoSmithKline/US/en/Prescribing_Information/Tivicay/pdf/TIVICAY-PI-PIL.PDF. Last accessed July 2019.
|
| |
|
 |
 |
|
|