 |
 |
 |
| |
ISLATRAVIR (ISL, MK-8591) at doses of 0.25 to 2.25 mg QD
in combination with doravirine maintains viral suppression through 48 weeks in adults with HIV-1 infection
|
| |
| |
Reported by Jules Levin
10th IAS Conference on HIV Science (IAS 2019), July 21-24, 2019, Mexico City


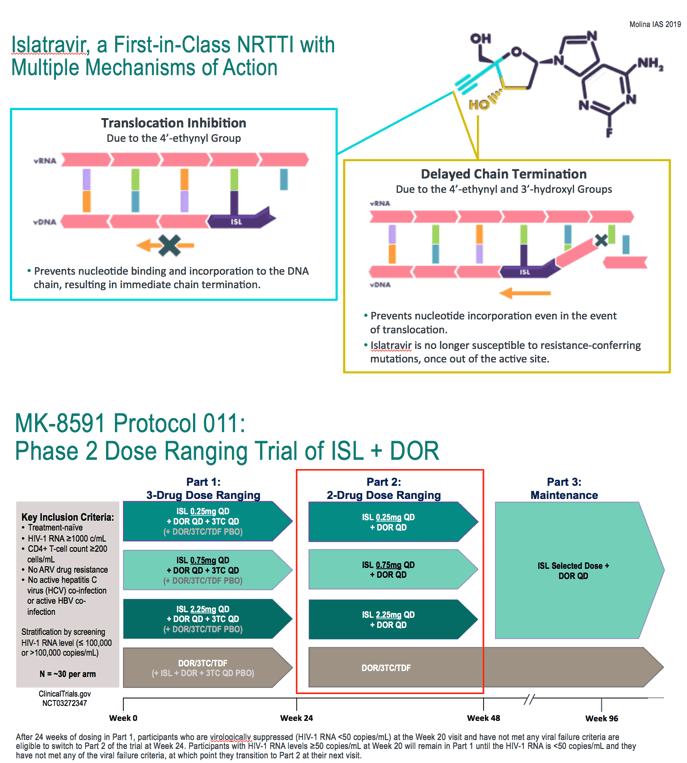
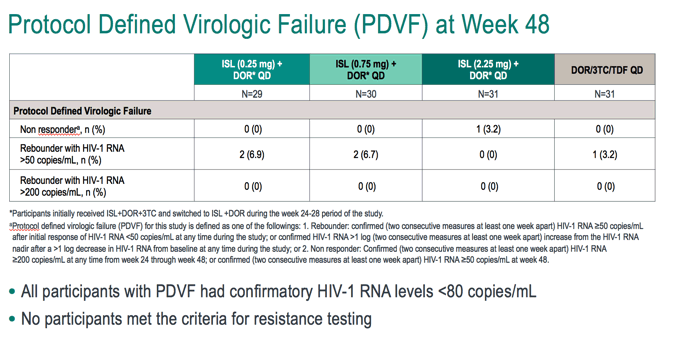
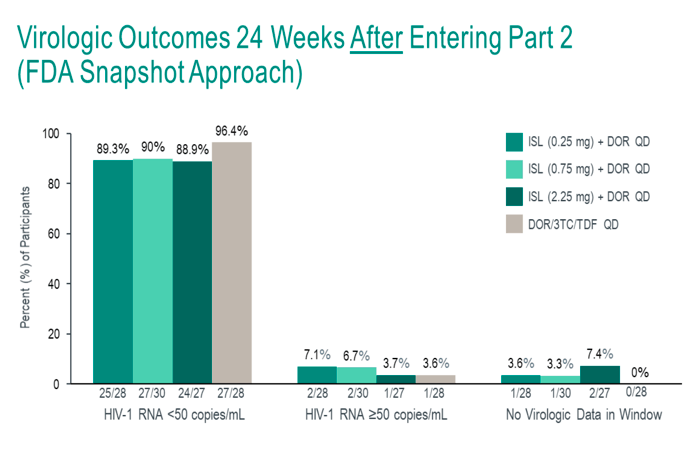
After 24 weeks of dosing in Part 1, participants who are virologically suppressed (HIV-1 RNA <50 copies/mL) at the Week 20 visit and have not met any viral failure criteria are eligible to switch to Part 2 of the trial at Week 24. Participants with HIV-1 RNA levels ≥50 copies/mL at Week 20 will remain in Part 1 until the HIV-1 RNA is <50 copies/mL and they have not met any of the viral failure criteria, at which point they transition to Part 2 at their next visit.

|
| |
|
 |
 |
|
|