 |
 |
 |
| |
Rosuvastatin Does Not Slow Atherosclerosis in HIV+ With Moderate Risk
|
| |
| |
10th IAS Conference on HIV Science (IAS 2019), July 21-24, 2019, Mexico City
Mark Mascolini
Rosuvastatin for 96 weeks cut total and low-density lipoprotein (LDL) cholesterol in people with HIV and moderate cardiovascular risk, but it did not slow atherosclerotic progression estimated by carotid artery intima-media thickness (cIMT) [1]. Because rosuvastatin did boost the risk of side effects, the researchers concluded "the benefits of statin therapy in [people with HIV] at low-moderate risk may not justify the potential harms."
Whether people with HIV should start primary preventive therapy for cardiovascular disease at a different threshold than people without HIV remains uncertain because risk scores to predict myocardial infarction (MI) lack sensitivity and specificity in people with HIV. Researchers in Australia and Switzerland planned this study to see whether 96 weeks of rosuvastatin slow cIMT-determined atherosclerotic progression in people with moderate cardiovascular risk who do not meet standard criteria for statin therapy.
This double-blind, placebo-controlled trial randomized participants 1-to-1 to 20 mg of rosuvastatin daily or matching placebo for 96 weeks. People taking a boosted protease inhibitor or cobicistat took 10 mg of the statin daily. Participants had to be older than 30 and have moderate coronary heart disease risk (Framingham Risk Score 10% to 15%). They needed a viral load below 200 copies on a stable antiretroviral regimen for more than 3 months. The researchers excluded people who met Australian clinical or lipid guidelines for lipid-lowering therapy. Participants had cIMT measured at screening for the trial and at weeks 48 and 96.
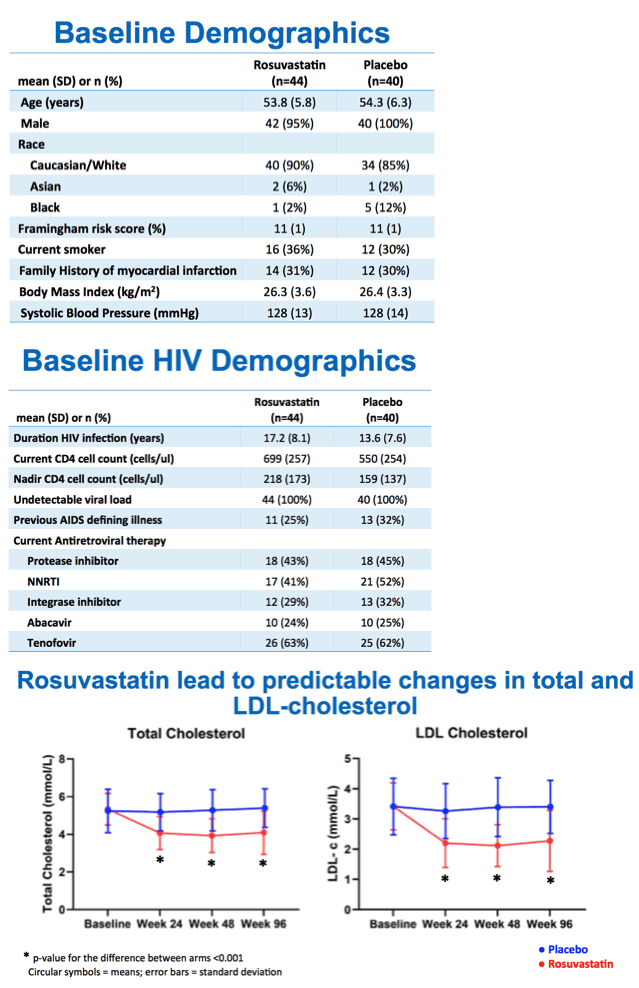
Of 84 people randomized, 76 completed week 48 and 73 week 96. Age averaged 54 years in both the 44 people randomized to rosuvastatin and the 40 randomized to placebo. All but 2 participants were men and almost 90% white. Framingham Risk Score averaged 11% in the rosuvastatin and placebo groups. About one third in both groups smoked, and about one third had a family history of MI.
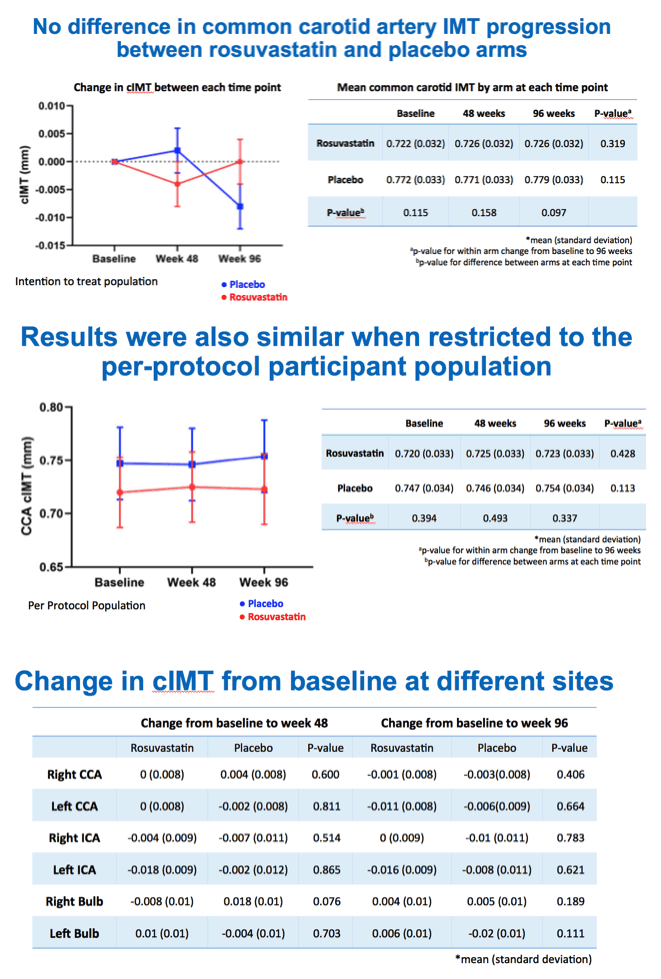
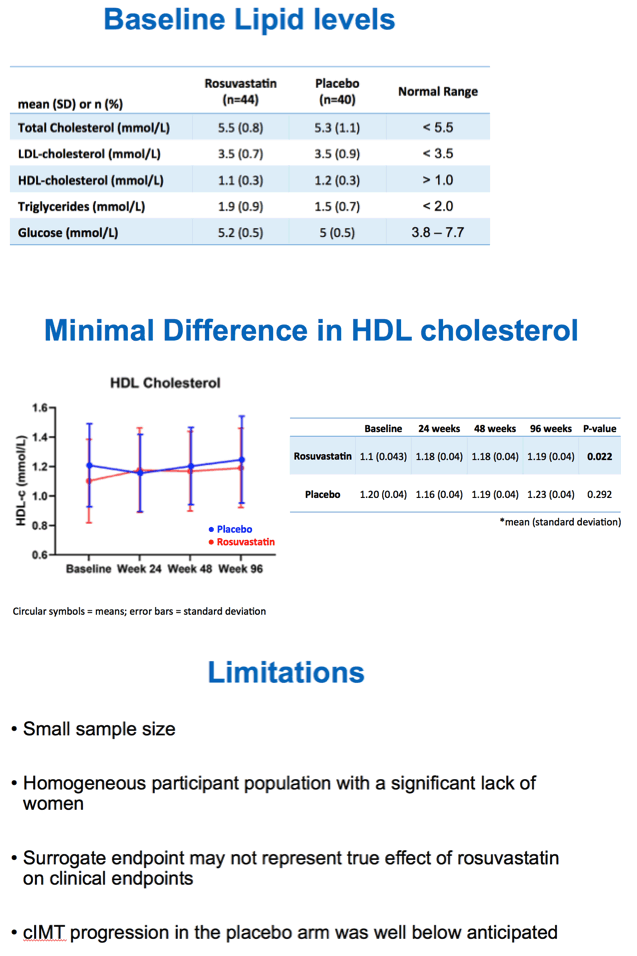
Total cholesterol and LDL cholesterol both fell significantly with rosuvastatin compared with placebo by week 24, and participants maintained that difference through weeks 48 and 96 (P < 0.001 for all). But in the intention-to-treat population, cIMT did not change significantly over 96 weeks with rosuvastatin (0.722 to 0.726 mm, P = 0.319) or placebo (0.772 to 0.779 mm, P = 0.115). Those results did not vary much when researchers restricted the analysis to people who remained on assigned treatment through 96 weeks. Nor did cIMT results differ substantially after adjustment for age, gender, and initial Framingham score.
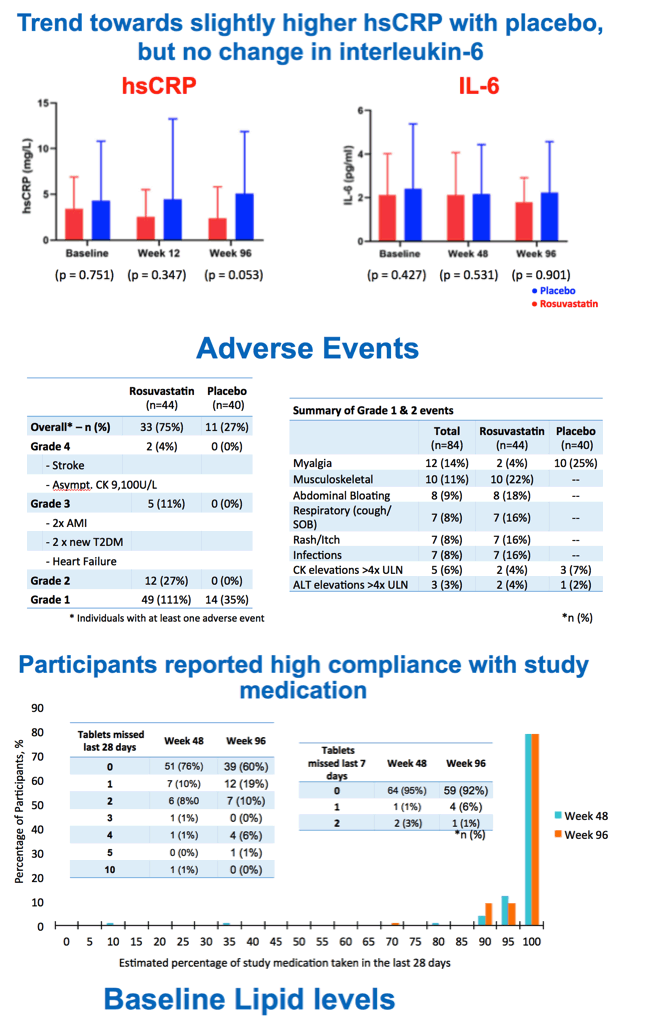
The researchers recorded a trend toward a slightly higher level of high-sensitivity C-reactive protein (an inflammation marker) with placebo than with rosuvastatin at week 96 (P = 0.53), but interleukin-6 levels did not vary much between the two groups through 96 weeks.
Compared with the placebo group, higher proportions of people assigned to rosuvastatin had grade 2 adverse events (27% versus 0%), grade 3 events (11% versus 0%), and grade 4 events (4% versus 0%). Grade 3 and 4 events included some of the hard cardiovascular endpoints statins aim to prevent--stroke, acute MI, heart failure, and type 2 diabetes. Adherence to assigned treatment was high.
Given rosuvastatin's failure to slow atherosclerotic progression in HIV-positive people at moderate cardiovascular risk, the researchers underlined the "need to await the currently running REPRIEVE trial [2] before different thresholds for the institution of primary preventative therapy in people with HIV can be recommended."
Reference
1. Trevillyan J, Dart A, Cavassini M, et al. Impact of rosuvastatin on atherosclerotic progression in people with HIV at moderate cardiovascular risk; A multinational, randomized, double blind, placebo-controlled trial. 10th IAS Conference on HIV Science (IAS 2019), July 21-24, 2019, Mexico City. Abstract MOAB0201.
2. REPRIEVE. Randomized Trial to Prevent Vascular Events in HIV. https://www.reprievetrial.org/overview/






|
| |
|
 |
 |
|
|