 |
 |
 |
| |
High Rates of Virologic Suppression Achieved in HIV-1-infected Adults Rapidly Starting Antiretroviral Therapy (ART) With the Single-tablet Regimen (STR) of Darunavir/Cobicistat/Emtricitabine/Tenofovir Alafenamide (D/C/F/TAF) 800/150/200/10 mg Regardless of Baseline Disease Characteristics: Week 48 Subgroup Analyses From the Phase 3 DIAMOND Trial
|
| |
| |
Reported by Jules Levin
IDWeek 2019™, October 2-6, 2019, Washington, DC
Gregory D. Huhn,1 Moti Ramgopal,2 Gordon Crofoot,3 Joseph Gathe, Jr,4 Sareh Seyedkazemi,5 Patricia Cosler,5 Richard Bruce Simonson,5 Donghan Luo,6 Keith Dunn5
1The Ruth M. Rothstein CORE Center, Chicago, IL; 2Midway Immunology and Research Center, Fort Pierce, FL; 3Crofoot Research Center, Houston, TX; 4Therapeutic Concepts, Houston, TX;
5Janssen Scientific Affairs, LLC, Titusville, NJ; 6Janssen Research & Development, LLC, Titusville, NJ.

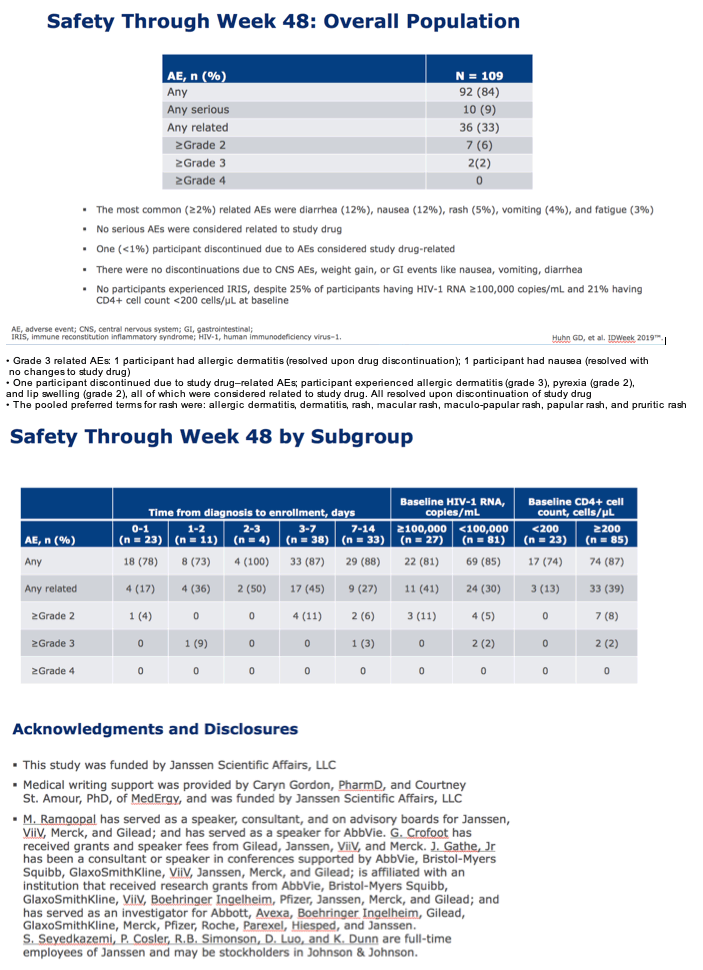
|
| |
|
 |
 |
|
|