 |
 |
 |
| |
Adherence to Long-Acting Injectable Cabotegravir + Rilpivirine through 48 Weeks of Maintenance Therapy in the Phase 3 ATLAS and FLAIR Studies
|
| |
| |
IDWeek Oct 2-6 2019 Wash DC
Reported by Jules Levin


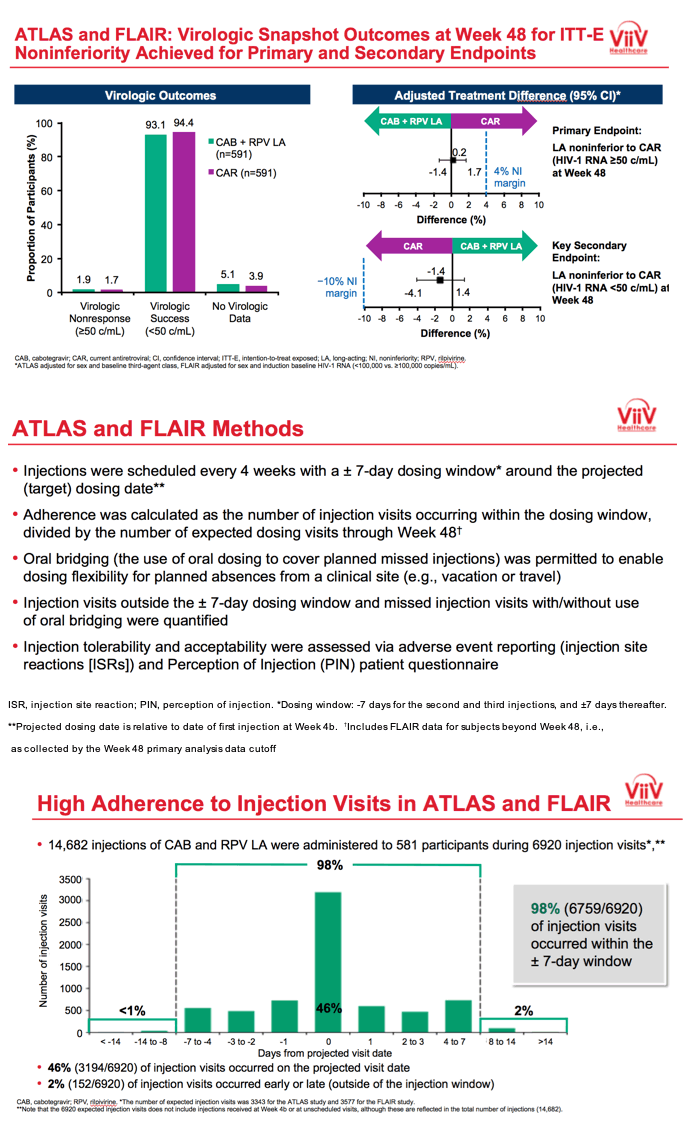
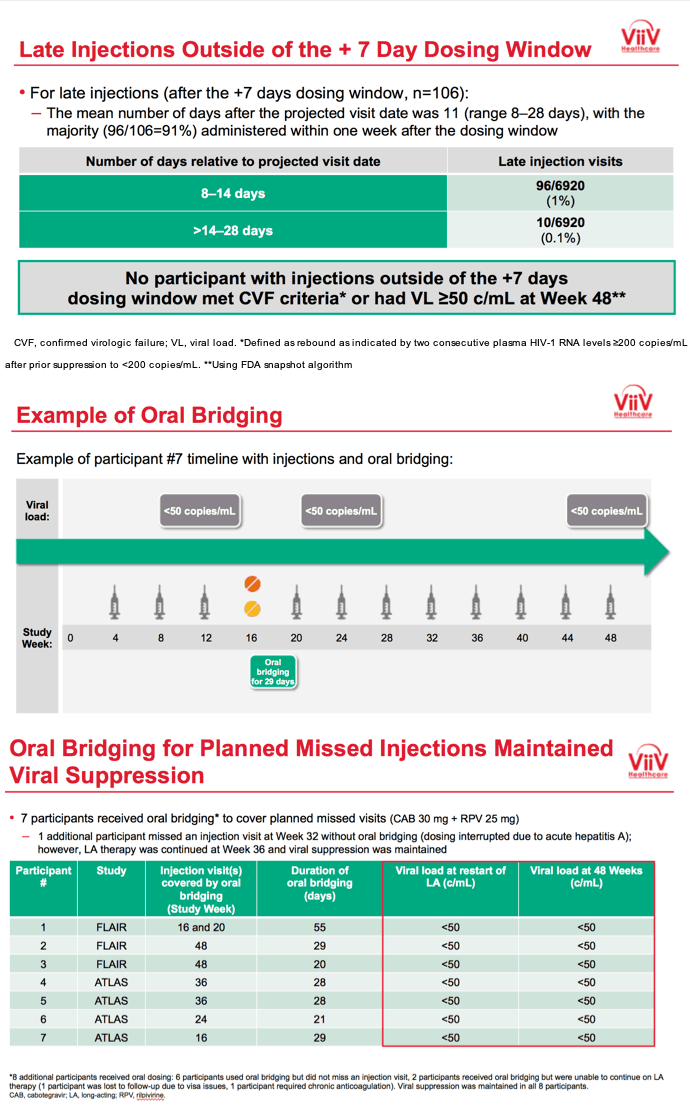
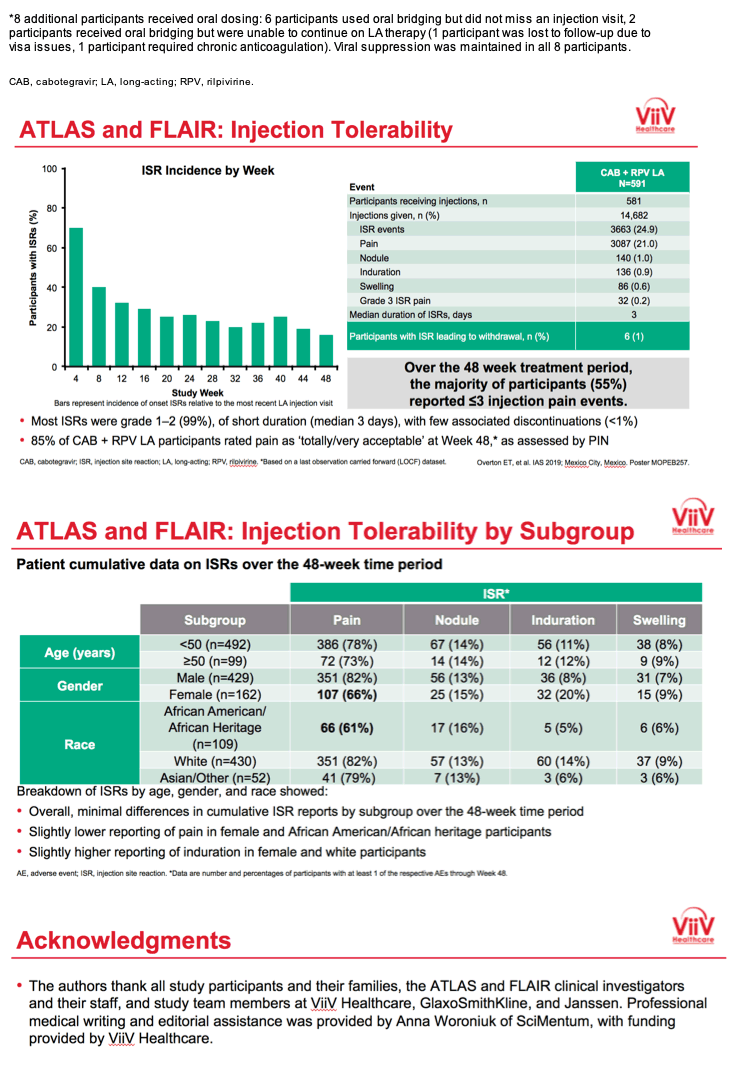
3TC, lamivudine; ABC, abacavir; ART, antiretroviral therapy; CAB, cabotegravir; CAR, current antiretroviral; DTG, dolutegravir; IM, intramuscular; INSTI, integrase strand transfer inhibitor; HBsAg, hepatitis B surface antigen; HLA, human leukocyte antigen; LA, long-acting; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor; RAM, resistance-associated mutation; RPV, rilpivirine; VL, viral load. *Uninterrupted ART 6 months and VL <50 c/mL at Screening, 2x VL <50 c/mL ≤12 months; Triumeq excluded from study; Optional switch to CAB LA + RPV LA at Week 52 for those on CAR; Participants received an initial loading dose of CAB LA (600 mg) and RPV LA (900 mg) at Week 4b. From Week 8 onwards, participants received CAB LA (400 mg) + RPV LA (600 mg) injections every 4 weeks; NNRTI RAMs but not K103N were exclusionary; Participants who withdraw/complete CAB LA + RPV LA enter 52-week long-term follow-up. **DTG plus 2 alternative non-ABC NRTIs was permitted if participant was intolerant or HLA-B*5701-positive (n=30 as last regimen during induction: n=2 discontinued during induction, n=14 randomized to CAB LA + RPV LA, n=14 randomized to DTG/ABC/3TC arm and continued on DTG plus 2 alternative non-ABC NRTIs in Maintenance Phase).
|
| |
|
 |
 |
|
|