 |
 |
 |
| |
Initial DTG+3TC Noninferior to DTG+TDF/FTC
|
| |
| |
IDWeek, October 2-6, 2019, Washington, DC
Mark Mascolini
At week 96 of the GEMINI trials, dolutegravir plus lamivudine (DTG+3TC) proved virologically noninferior to DTG plus tenofovir/emtricitabine (TDF/FTC) [1]. People starting DTG+3TC with a CD4 count below 200 had a worse 96-week virologic response than those starting DTG+TDF/3TC.
GEMINI 1 and GEMINI 2 were identical international double-blind trials comparing DTG+3TC and DTG+TDF/FTC in previously untreated adults. In the 48-week combined analysis, DTG+3TC proved virologically noninferior to the 3-drug combination [2]. DTG+3TC maintained noninferiority through 96 weeks. The new analysis focused on 96-week response rates in subgroups enrolled in the two trials.
This pooled analysis involved 716 people randomized to DTG+3TC and 717 randomized to DTG+TDF/FTC. Median ages in the two groups were 32 and 33, 16% and 14% were women, and 68% and 70% were white. About 20% of participants began treatment with a viral load above 100,000 copies and about 10% with a CD4 count below 200.
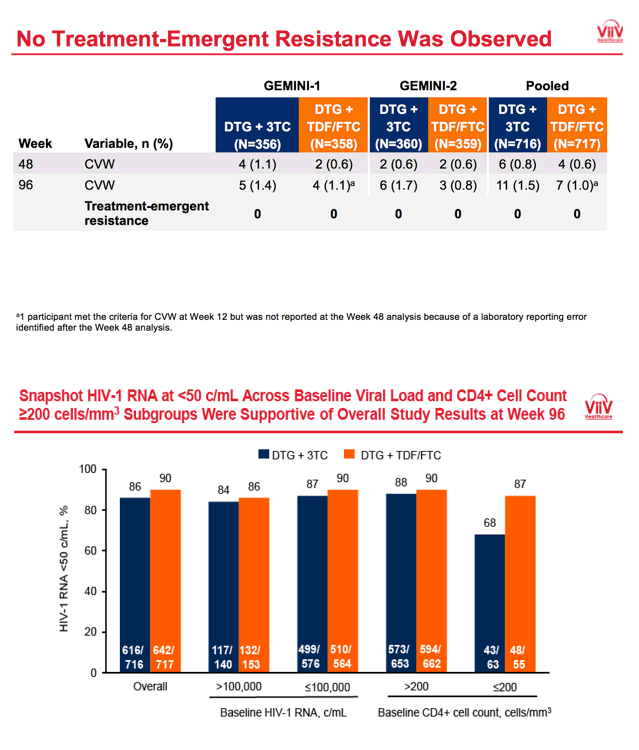
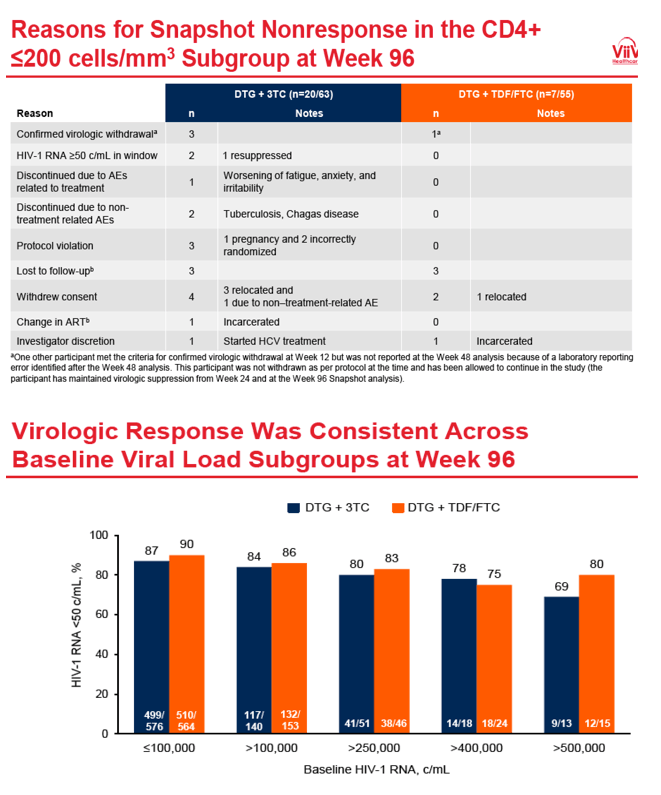
A week-96 FDA snapshot analysis determined that 86.0% randomized to DTG+3TC and 89.5% randomized to the 3-drug regimen had a viral load below 50 copies, results meeting noninferiority criteria for the 2-drug combination. Snapshot analysis also showed similar proportions of participants below 50 copies at week 96 among those starting treatment below 100,000 copies (87% DTG+3TC, 90% DTG+TDF/FTC) or above 100,000 copies (84% DTG+3TC, 86% DTG+TDF/FTC).
Among participants beginning therapy with more than 200 CD4s, 96-week snapshot response rates were similar with the two therapies (88% DTG+3TC, 90% DTG+TDF/FTC). But among people starting with fewer than 200 CD4s, those randomized to DTG+3TC had a lower snapshot response rate than those randomized to the 3-drug regimen (68% DTG+3TC, 87% DTG+TDF/FTC). In a preplanned week 96 treatment-related-discontinuation-equals-failure analysis, sub-50-copy response rates in people starting with fewer than 200 CD4s were similar with the two regimens (93% DTG+3TC, 96% DTG+TDF/FTC).
In the week-96 snapshot analysis, the 68% response rate with DTG+3TC reflected nonresponse in 20 of 63 people randomized to DTG+3TC. Only 3 of those 20 had a confirmed virologic withdrawal (as did 1 person in the DTG+TDF/FTC group). Other reasons for nonresponse to DTG+3TC were withdrawn consent (4), loss to follow-up (3), protocol violation (3), viral load above 50 copies in window (2, with 1 resuppression), nontreatment-related adverse event (2), treated-related adverse event (1), change in antiretroviral therapy (1), and investigator discretion (1).
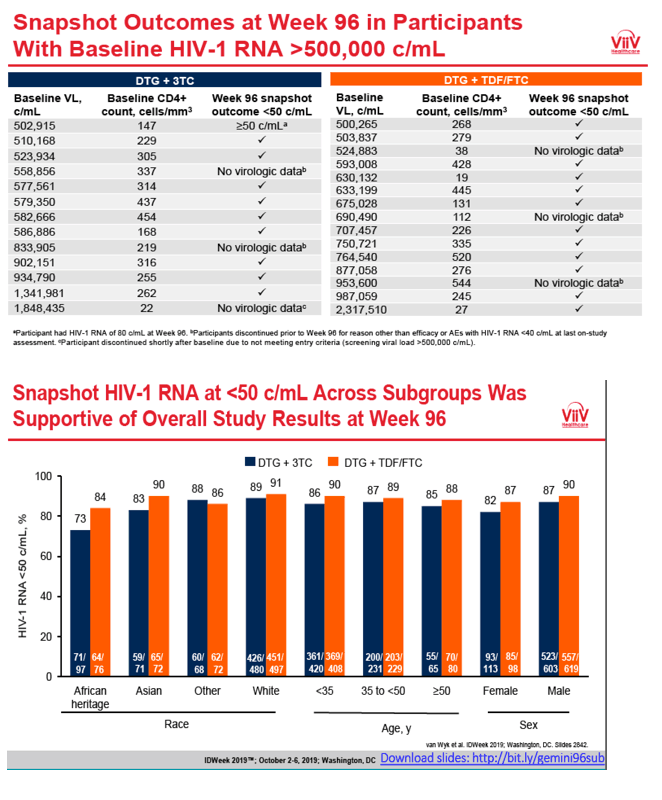
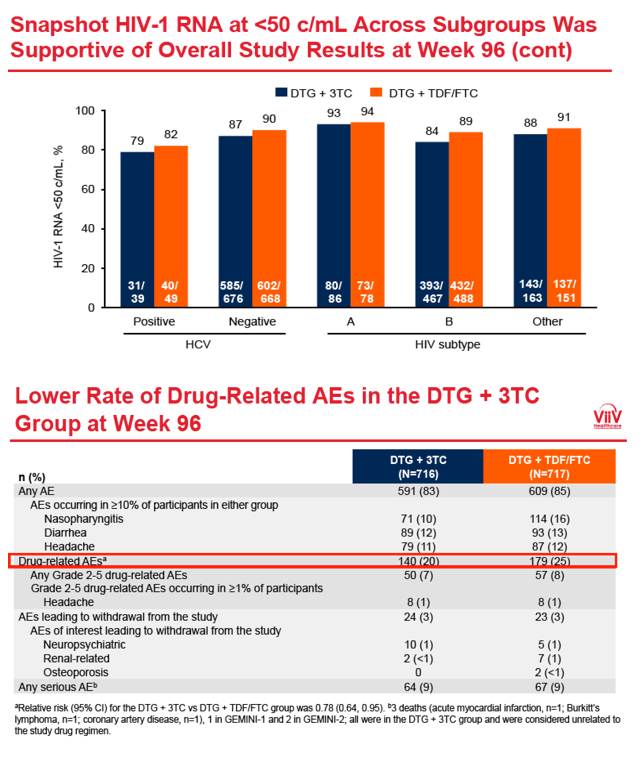
In other subgroup analyses, week-96 sub-50-copy response rates were marginally lower with DTG+3TC than with the 3-drug regimen in people of African heritage (73% DTG+3TC, 84% DTG+TDF/FTC) but similar with the two regimens for whites, Asians, and others. Response rates did not differ between treatment arms when the researchers stratified participants by age (under 35, 35 to under 50, 50+), sex, HCV positivity, or HIV subtype (B or other).
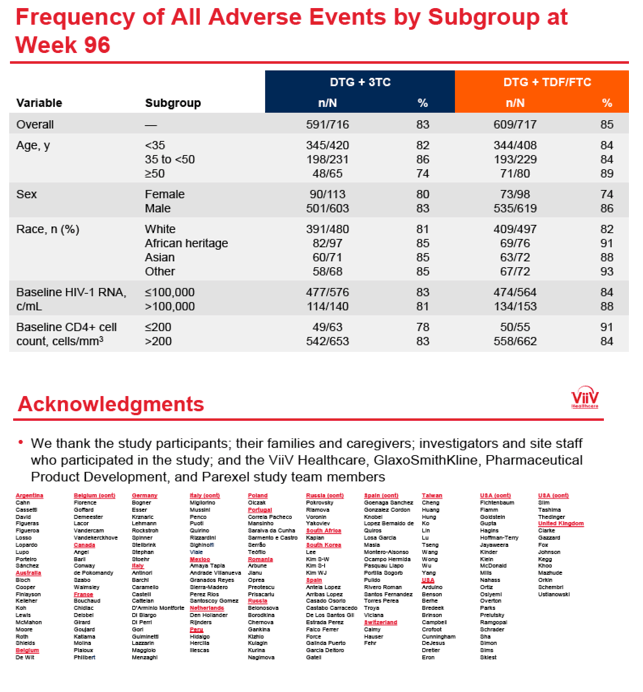
The overall drug-related adverse event rate was slightly lower with DTG+3TC than with the control regimen (20% versus 25%). The same proportions in the two study arms had any serious adverse event (9%) or an adverse event leading to discontinuation (3%).
References
1. van Wyk J, Man C, Sievers J, et al. Durable efficacy of two-drug regimen (2DR) of dolutegravir (DTG) plus lamivudine (3TC) in antiretroviral treatment-naive adults with HIV-1 infection at 96 weeks: subgroup analyses in the GEMINI studies. IDWeek, October 2-6, 2019, Washington, DC. Abstract 2842. Slides at http://bit.ly/gemini96sub
2. Cahn P, Madero JS, Arribas JR, et al. Dolutegravir plus lamivudine versus dolutegravir plus tenofovir disoproxil fumarate and emtricitabine in antiretroviral-naive adults with HIV-1 infection (GEMINI-1 and GEMINI-2): week 48 results from two multicentre, double-blind, randomised, non-inferiority, phase 3 trials. Lancet. 2019;393:143-155. Erratum in Lancet. 2019;393:132.


|
| |
|
 |
 |
|
|