 |
 |
 |
| |
Real world utilization of ibalizumab (IBA) without
an optimized background regimen (OBT)
|
| |
| |
Reported by Jules Levin
ID Week 2019 Oct 2-6 Wash DC
Jeannette Bouchard, PharmD; Caroline Derrick, PharmD, BCPS; Joseph Horvath, MD
(1) University of South Carolina School of Pharmacy, Columbia, SC, (2) University of South Carolina School of Medicine, Colum, SC

Program Abstract
Background : It is difficult to treat multidrug resistant (MDR) human immunodeficiency virus (HIV). Trogarzo® (ibalizumab) a novel monoclonal antibody was approved in 2018 for heavily treatment-experienced HIV patients. Data support IBA use with at least one fully active agent, an OBR. Real-world IBA data are lacking. We report a successful case of reaching and maintaining suppression with IBA in a patient without an OBR.
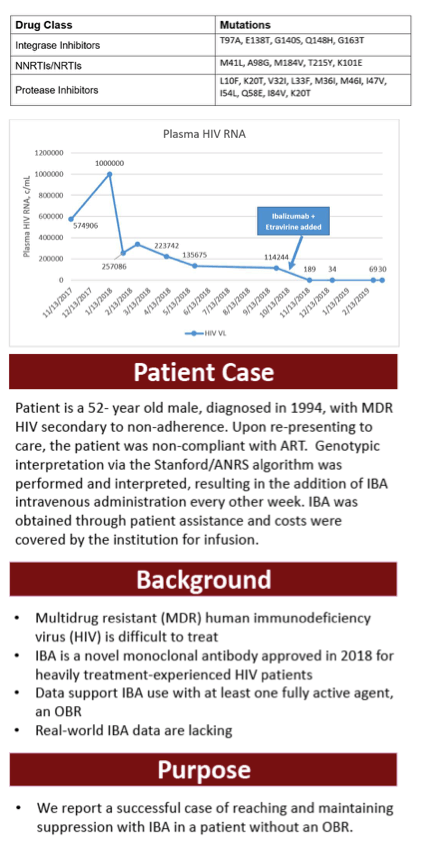
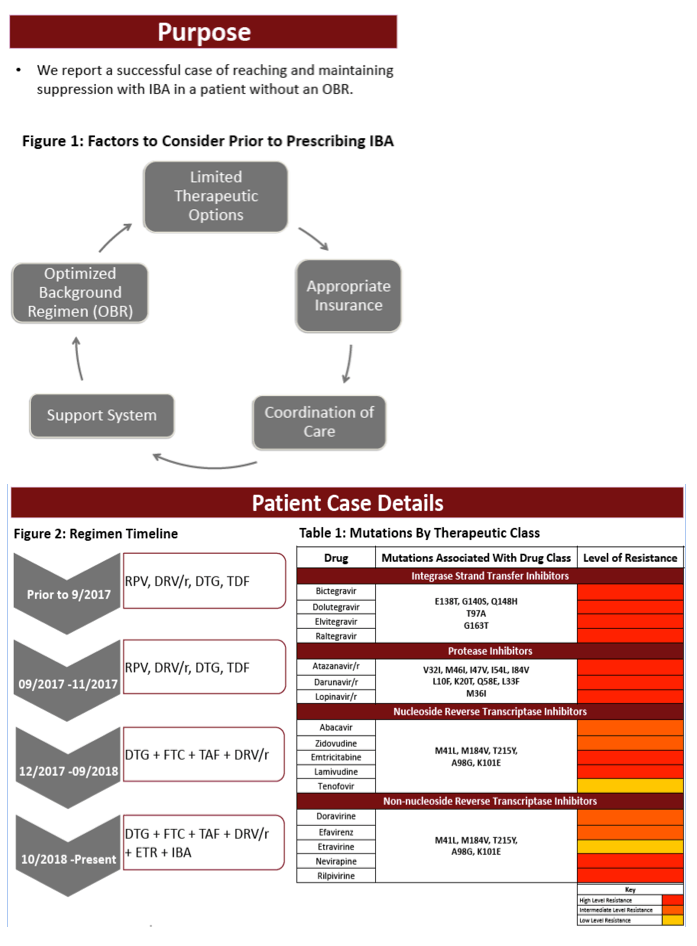
Methods : Mutations were reviewed for the patient, Table 1, and evaluated for treatment. The patient is a 52- year old male, diagnosed in 1994, with MDR HIV secondary to non-adherence. Upon re-presenting to care, the patient was non-compliant with ART. Genotypic interpretation via the Stanford/ANRS algorithm was performed and interpreted, resulting in the addition of IBA intravenous administration every other week. IBA was obtained through patient assistance and costs were covered by the institution for infusion.
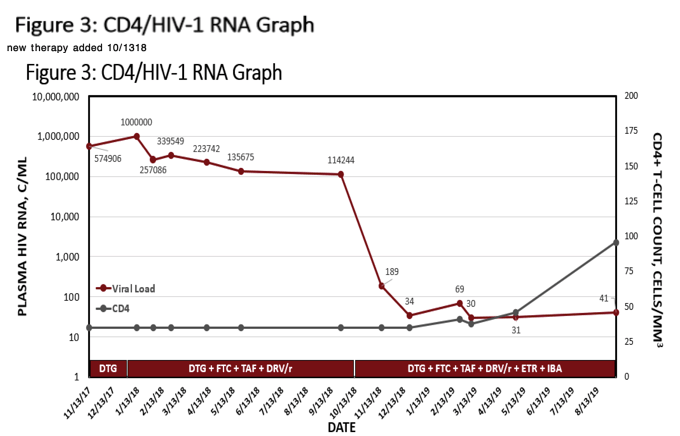
Results : Evaluation of the resistance profile indicated varying resistance to all available ART. More specifically, high-level resistance to all FDA-approved INSTIs, PIs, and low to high-level resistance to all NNRTIs and NRTIs. Table 2 outlines the ART history and viral load (VL) trends. The patient was initiated on daruanvir/ritonavir twice daily, etravirine twice daily, emtricitabine/tenofovir alafenamide and did not reach suppression. IBA was added off-label to a failing regimen. The patient reached VS (VL < 200 copies/mL) at Week 4 and has had an undetectable VL for 8 weeks. Notably his CD4 count has risen to 46, first detectable number since re-presenting to care.
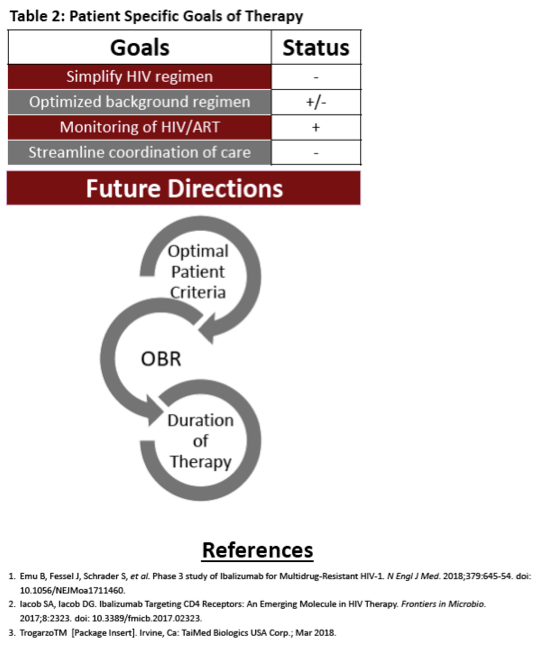
Conclusion : We describe a heavily treatment experienced patient with an MDR HIV virus who achieved an undetectable VL without an OBT and the addition of intravenous IBA. Fostemsavir, was utilized in IBA's phase III trial for similar patients, however, it is not currently FDA-approved nor available. Further data are needed to ensure continued susceptibility to IBA without an OBT. This patient required high level coordination to reach each visit and receive this therapy alongside his oral agents. We conclude, IBA has allowed this patient to reach and maintain VS.
|
| |
|
 |
 |
|
|