 |
 |
 |
| |
Results of the First Prophylactic
Hepatitis C Virus Vaccine Efficacy Trial
|
| |
| |
Reported by Jules Levin
IDWeek 2019; October 2-6, 2019; Washington, DC, USA
Kimberly Page, Michael Melia, Rebecca T. Veenhuis, Guido Massaccesi, William O. Osburn, Katherine Wagner, Linda Giudice, Ellen Stein, Alice Asher, Ventzislav
Vassilev, Lan Lin, Alfredo Nicosia, Stefania Capone, Elisa Scarselli, Antonella Folgori, Richard Gorman, Soju Chang, Peter A. Wolff, T. Jake Liang, Marc Ghany, Michael R. Wierzbicki, Paula J. Lum, and Andrea L. Cox

program abstract
Background:
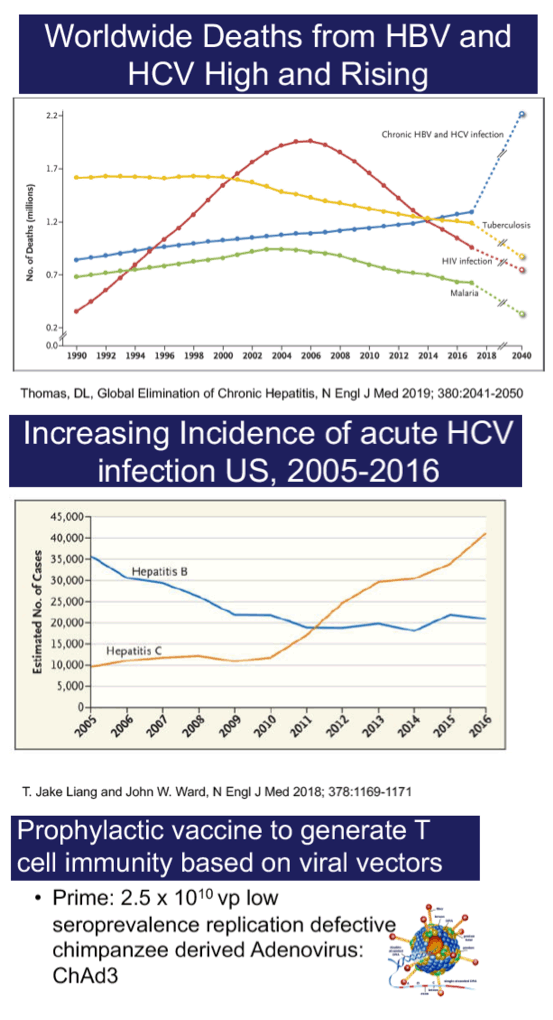
The development of a safe and effective vaccine to prevent chronic hepatitis C virus (HCV) infection is a critical component of elimination efforts, providing the rationale for the first HCV vaccine efficacy trial.
Methods:
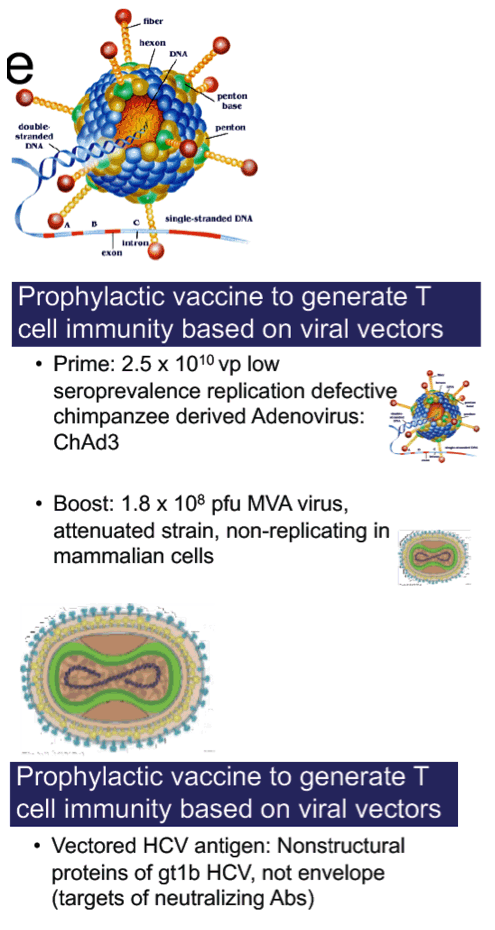
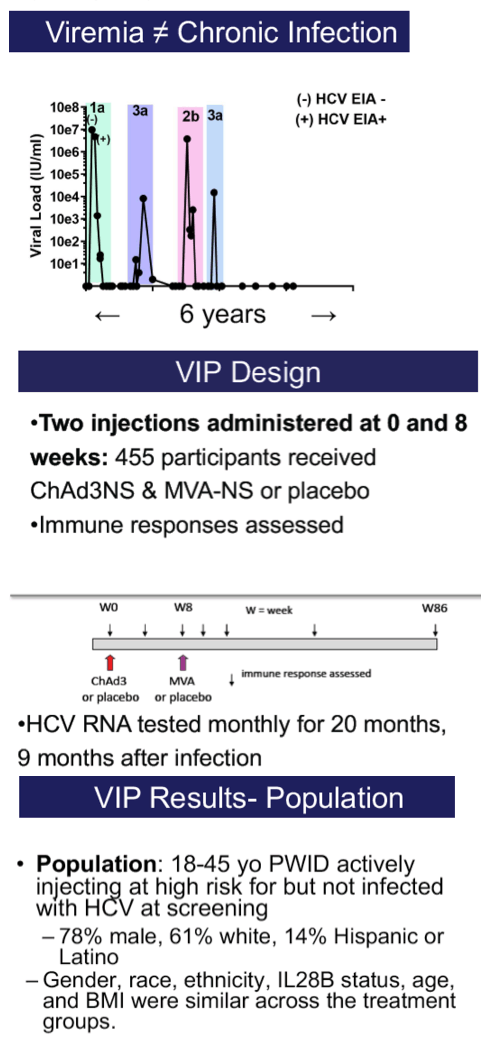
In a randomized, multicenter, double-blind, placebo-controlled efficacy trial (NCT01436357), we evaluated a recombinant chimpanzee adenovirus 3 vector vaccine prime followed by a recombinant modified vaccinia Ankara boost, both encoding nonstructural proteins of HCV. HCV-uninfected adults 18-45 years old at-risk for HCV infection due to injection drug use were randomized to receive the prime-boost regimen or placebo at Days 0 and 56. Trial participants were monitored for vaccine reactogenicity, adverse events, and HCV viremia. Vaccine safety, immunogenicity, and efficacy against progression to chronic HCV infection were assessed.
Results:
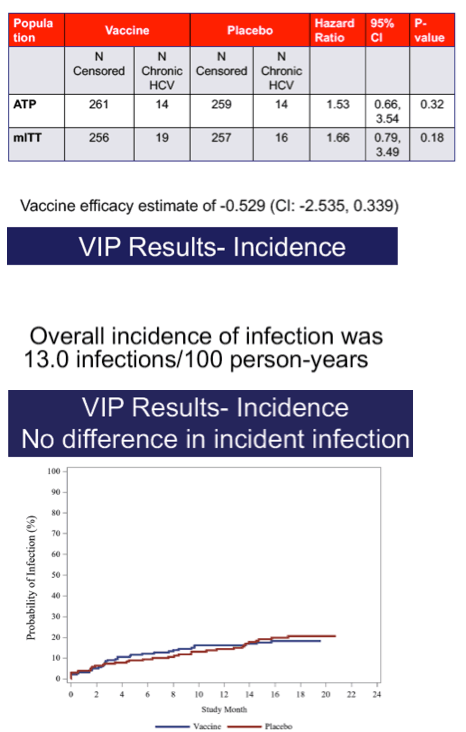
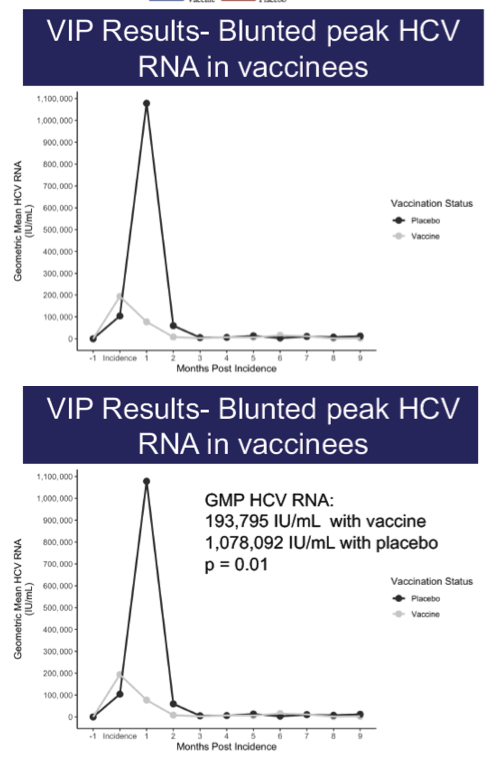
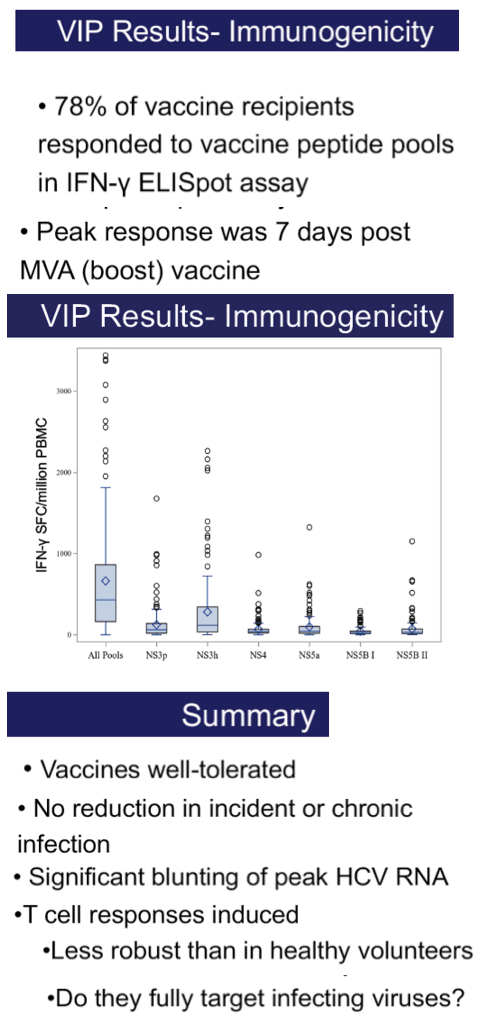
A total of 455 subjects received the prime-boost regimen or two doses of placebo, with 202 and 199 in the respective groups included in the according-to-protocol efficacy cohort. Overall incidence of infection was 14.1 infections per 100 person-years. There were no differences in development of chronic infection between vaccine and placebo arms, with 14 chronically infected subjects in each group. Specifically, the vaccine efficacy in preventing chronic infection was -0.53 (95% confidence interval [CI], -2.5 to 0.34). Of vaccinated subjects, 78% generated T cell responses to ≥1 vaccine-encoded HCV antigens. The vaccine was generally safe and well tolerated with no serious vaccine-related adverse events. There were more solicited reports of adverse events after either injection in the vaccine group (81%) than in the placebo group (59%), with the difference mainly due to injection-site reactions. Serious adverse events and deaths occurred with similar frequencies in the two groups.
Conclusion:
A randomized, placebo controlled, Phase I/II trial of a prime-boost vaccine to prevent chronic HCV infection was completed in an at-risk population, demonstrating the feasibility of conducting rigorous vaccine research in people who inject drugs. The regimen elicited robust immune responses without evident safety concerns, but did not provide protection against chronic HCV infection.



|
| |
|
 |
 |
|
|