 |
 |
 |
| |
Efficacy and safety of subcutaneous semaglutide once-daily versus placebo in patients with nonalcoholic steatohepatitis
Semaglutide Resolves NASH in Big Placebo-Controlled Trial
|
| |
| |
AASLD The Liver Meeting Digital Experience, November 13-16, 2020
Mark Mascolini
Semaglutide, a glucagon-like peptide-1 agonist (GLP-1RA) licensed for treatment of type 2 diabetes, resolved nonalcoholic steatohepatitis (NASH) without worsening fibrosis significantly more often than placebo at all 3 subcutaneous doses used in a 320-person 72-week trial [1]. The medication did not significantly improve fibrosis compared with placebo, but it did improve weight, lipids, and the diabetes metric HbA1c.
Besides lowering glucose, GLP-1RAs have beneficial effect on heart, brain, and pancreas, according to an international team that conducted the semaglutide trial. This 17-country phase 2 double-blind placebo-controlled trial enrolled people with biopsy-confirmed NASH, stage 1 to 3 fibrosis, body mass index above 25 kg/m2 (the overweight threshold), HbA1c at or below 10% and NAFLD Activity Score at or above 4. A hierarchical testing procedure [2] randomized participants to subcutaneous* doses 0.4 mg, 0.2 mg, or 0.1 mg of semaglutide once daily versus placebo for 72 weeks. The primary endpoint was resolution of NASH with no liver fibrosis worsening in people with initial stage 2 or 3 fibrosis.
The trial included 80 people randomized to 0.1 mg of semaglutide, 78 to 0.2 mg, 82 to 0.4 mg, and 80 to placebo [3]. All participants also received counseling on nutrition and physical activity. Age averaged 55 years across the four study group, and 55% to 66.7% across the four groups were women. About 62% in the four study arms had diabetes, and body mass index averaged 35.8 kg/m2. Proportions with stage 1, 2, and 3 fibrosis were 28.1%, 22.5%, and 49.4%.
In the 230 participants with stage 2 or 3 fibrosis, 40.4% receiving 0.1 mg of semaglutide daily (P = 0.01 vs placebo), 35.6% receiving 0.2 mg (P = 0.0359 vs placebo), and 58.9% receiving 0.4 mg (P < 0.0001 vs placebo), compared with 17.2% getting placebo, had resolution of NASH without worsening fibrosis. In an analysis of all randomized participants regardless of fibrosis stage, significantly higher proportions receiving each dose of semaglutide than placebo had NASH resolution with no fibrosis worsening.
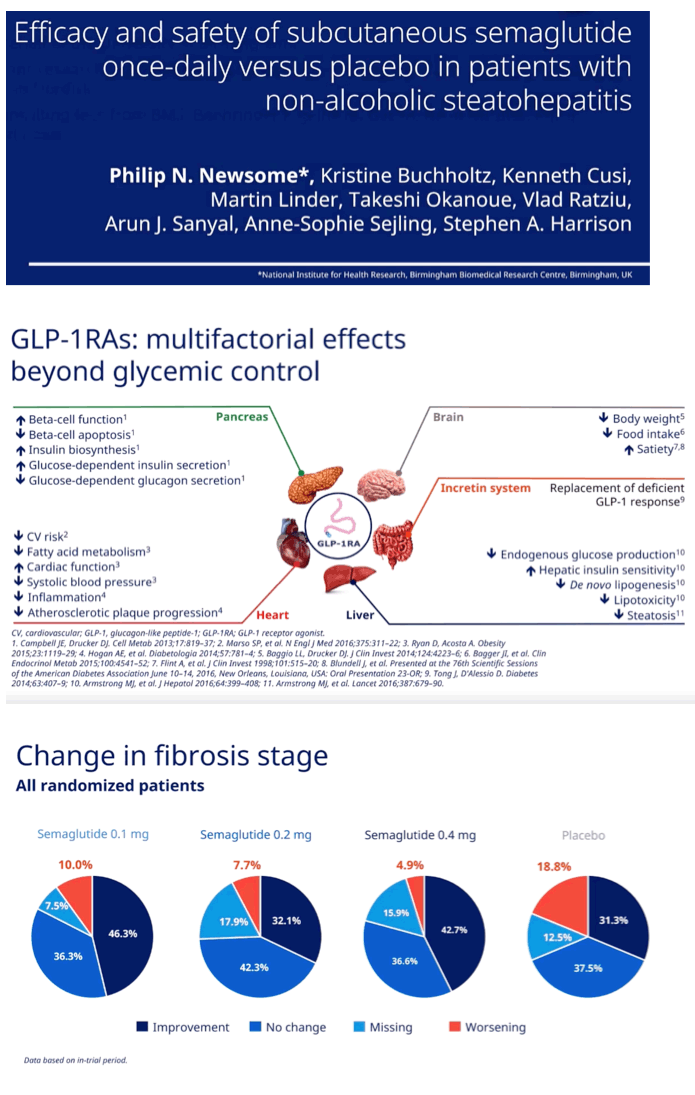
Among participants with stage 2 or 3 fibrosis at baseline, fibrosis did not improve significantly more with any dose of semaglutide than with placebo. Proportions with improved fibrosis were 46.3% with 0.1 mg of semaglutide, 32.1% with 0.2 mg, 42.7% with 0.4 mg, and 31.3% with placebo. Researchers recorded worsening fibrosis in 10% taking 0.1 mg of semaglutide, 7.7% taking 0.2 mg, 4.9% taking 0.4 mg, and 18.8% taking placebo.
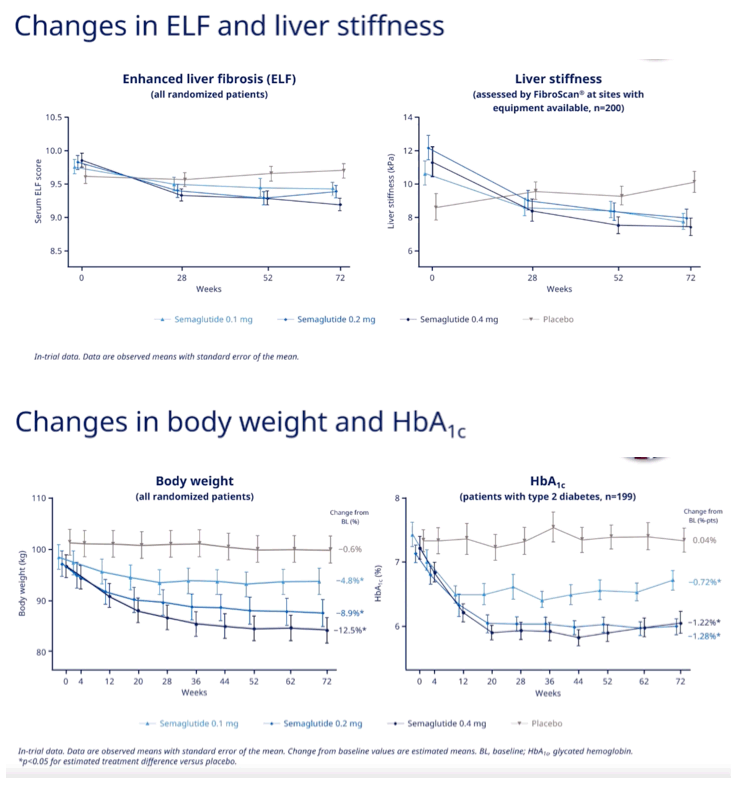
Semaglutide yielded dose-dependent 72-week improvements in alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transferase (GGT), and two fibrosis markers (ELF and liver stiffness by FibroScan).
Through 72 weeks in all randomized participants, body weight dropped 4.8% with 0.1 mg of semaglutide, 8.9% with 0.2 mg, 12.5% with 0.4 mg, and 0.6% with placebo (P < 0.05 for all differences from placebo). Over 72 weeks HbA1c fell 0.72% with 0.1 mg of semaglutide, 1.28% with 0.2 mg, 1.22% with 0.4 mg, and 0.04% with placebo (P < 0.05 for all differences from placebo).
Compared with placebo. 0.4 mg of semaglutide yielded a significant gain in "good" HDL cholesterol (P = 0.0064) and significant drops in triglycerides (P < 0.0001), VLDL cholesterol (P < 0.0001), and free fatty acids (P = 0.0029).
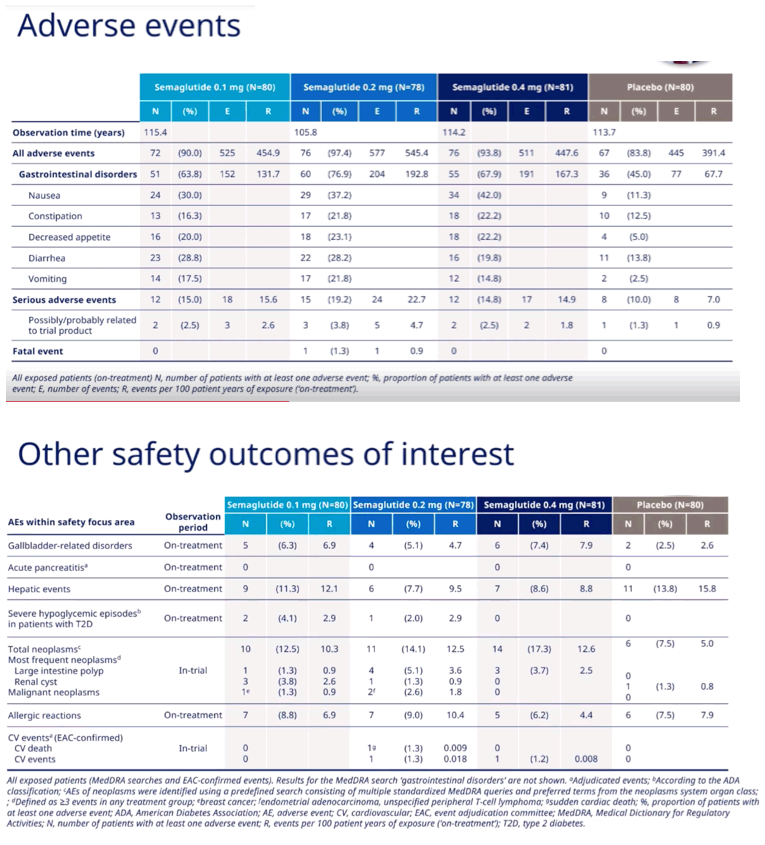
Rates of serious adverse events possibly or probably related to study drug were 2.5% with 0.1 mg of semaglutide, 3.8% with 0.2 mg, 2.5% with 0.4 mg, and 1.3% with placebo. Nausea was the most frequent problem, reported in 30% making 0.1 mg of semaglutide, 37.2% taking 0.2 mg, 42% taking 0.4 mg, and 11.3% taking placebo. The researchers believe the overall safety profile of semaglutide is similar to that seen in people with type 2 diabetes, "with no new safety concerns."
*A tablet formulation of semaglutide is now licensed for type 2 diabetes.
References
1. Newsome PN, Buchholtz K, Cusi K, et al. Efficacy and safety of subcutaneous semaglutide once-daily versus placebo in patients with nonalcoholic steatohepatitis. AASLD The Liver Meeting Digital Experience, November 13-16, 2020. Abstract 10.
2. ClinicalTrials.gov. Investigation of efficacy and safety of three dose levels of subcutaneous semaglutide once daily versus placebo in subjects with non-alcoholic steatohepatitis. ClinicalTrials.gov identifier NCT02970942.
3. Bretz F, Xun X. Introduction to multiplicity in clinical trials. Novartis. http://www2.cscc.unc.edu/impact7/system/files/Bretz_Slides.pdf

|
| |
|
 |
 |
|
|