 |
 |
 |
| |
Real world single center experience on the efficacy of stopping long term nucleos(t)ide analog therapy in patients with chronic hepatitis B.
|
| |
| |
Stopping Anti-HBV NUCs "Feasible" if HBeAg-Negative and HBsAg Below 100 IU/mLå
AASLD The Liver Meeting Digital Experience, November 13-16, 2020
Mark Mascolini
Stopping nucleos(t)ides (NUCs) for chronic HBV infection proved feasible and generally safe in people negative for HBeAg and with quantitative HBsAg (qHBsAg) below 100 IU/mL, according to results of a retrospective single-center study in Canada [1]. Six of 47 people (13%) who stopped NUCs had to restart because of virologic or biochemical flares.
University of Calgary researchers who conducted this study noted that NUC therapy for chronic HBV infection rarely leads to loss of hepatitis B surface antigen (HBsAg), which indicates current infection. As a result, therapy becomes prolonged for most people with chronic HBV. The Calgary team pointed out that some guidelines say hepatitis B e antigen (HBeAg)-positive people may stop NUCs after HBeAg seroconversion, undetectable HBV DNA for more than 6 to 12 months, and consolidation therapy [2], yet many relapse when NUCs stop. (HBeAg indicates ongoing HBV replication and the possibility of transmission.)
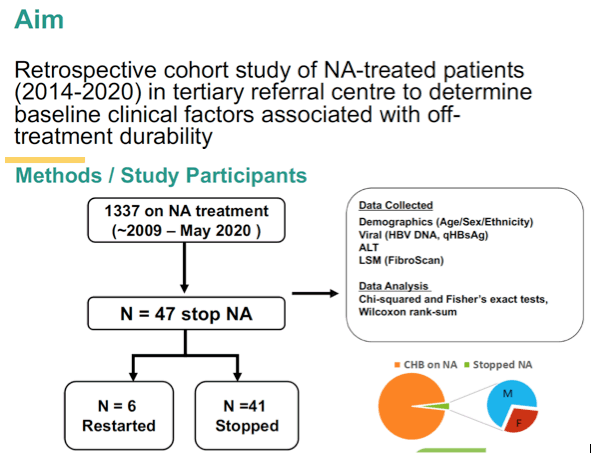
Because some evidence suggests NUCs can be stopped safely in HBV patients without cirrhosis, with undetectable HBV DNA, and with qHBsAg levels under 100 IU/mL, the Calgary researchers analyzed that approach in their long-term NUC patients, aiming to determine baseline factors that predicted off-treatment durability.
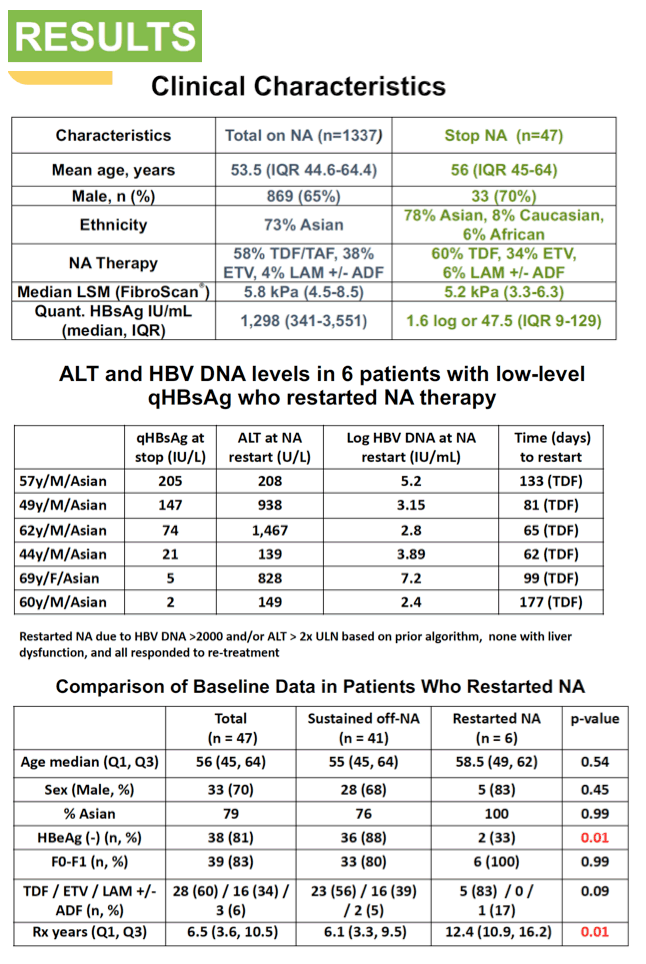
The analysis involved 1337 people taking long-term NUCs for chronic HBV infection. The researchers determined that 47 in this group (3.5%) stopped NUCs on their clinician's advice. Fourteen of the 47 (30%) were women, 37 (78%) were East Asian, and median age stood at 56 years (range 18 to 74). While 28 people (60%) were taking tenofovir disoproxil fumarate (TDF), 16 (34%) took entecavir and 3 (6%) lamivudine (2 of them combined with adefovir). All 47 people were HBeAg-negative when they stopped therapy, and 46 had undetectable HBV DNA. Liver stiffness ranged from 3.3 to 6.6 kPa (median 5.2 kPa).
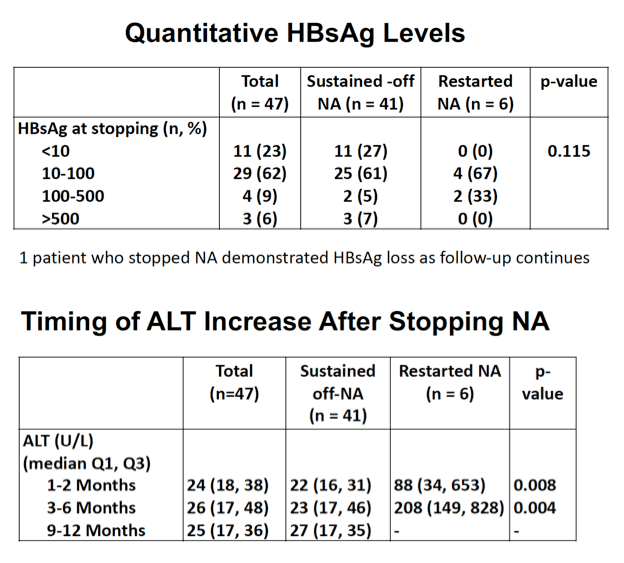
Six of these 47 people (13%) had to restart NUC therapy because of virologic flare (defined as HBV DNA above 2000 IU/mL) and/or biochemical flare (defined as alanine aminotransferase [ALT] more than 2 times the upper limit of normal). When these 6 stopped therapy their qHBsAg levels stood at 205, 147, 74, 21, 5.4, and 2.3. At their virologic flare their HBV DNA ranged from 265 to 152 million IU/mL (median 4663 IU/mL), and at biochemical flare their ALTs were 208, 938, 1467, 139, 828, and 149 IU/L. Time to restarting NUCs ranged from 62 to 177 days.
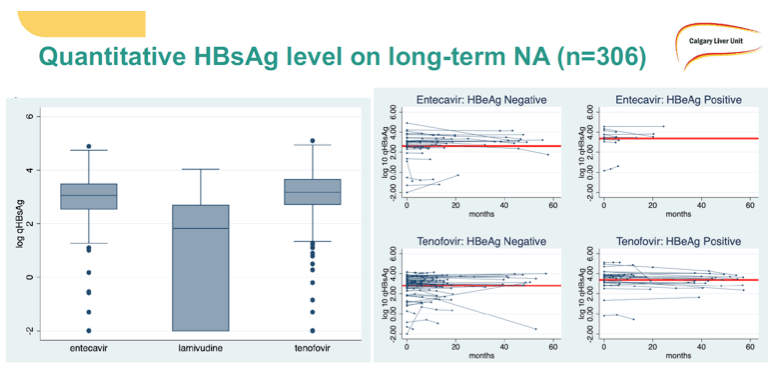
Hepatic dysfunction developed in no one who stopped NUC therapy, and all suppressed HBV DNA when treatment resumed (all with TDF). Two variables distinguished the 6 people who had to restart NUC therapy from the 41 who did not: (1) HBeAg-negative at baseline: 2 (33%) who restarted versus 36 (88%) who did not (P = 0.01); (2) longer NUC duration: median 12.4 years in restarters versus 6.1 years in sustained responders (P = 0.01). Age, sex, ethnicity, fibrosis stage F0-F1, and NUC taken did not affect relapse risk. qHBsAg when stopping therapy did not correlate with relapse. So far, 1 person who stopped therapy has lost HBsAg.
The Calgary investigators think their findings suggest that NUC withdrawal "is feasible" in HBeAg-negative people with chronic HBV infection, minimal fibrosis, and low qHBsAg levels. But because ALT flares can be severe, people who stop NUCs need close monitoring.
References
1. Azhari H, Frolkis A, Shaheen AA, et al. Real world single center experience on the efficacy of stopping long term nucleos(t)ide analog therapy in patients with chronic hepatitis B. AASLD The Liver Meeting Digital Experience, November 13-16, 2020. Abstract 24.
2. Consolidation therapy "generally involves treatment for at least 12 months of persistently normal ALT levels and undetectable serum HBV DNA levels." Terrault NA, Bzowej NH, Chang KM, et al. AASLD guidelines for treatment of chronic hepatitis B. Hepatology. 2016;63:261-83. doi: 10.1002/hep.28156. https://www.aasld.org/sites/default/files/2019-06/Terrault_et_al-2016-Hepatology.pdf
Real World Single Centre Experience on the Efficacy of Stopping Long Term Nucleos(t)ide Analog Therapy in Patients with Chronic Hepatitis
AASLD 2020 Nov 11-16 virtual
H Azhari, AD Frolkis, AA Shaheen, H Israelson, J Pinto, SE Congly, MA Borman, AA Aspinall, LM Stinton, MG Swain, KW Burak, SS Lee, MD Sadler and CS Coffin


|
| |
|
 |
 |
|
|