 |
 |
 |
| |
COST-EFFECTIVENESS OF ANTENATAL RESCREENING AMONG PREGNANT WOMEN FOR HEPATITIS C IN THE UNITED STATES
|
| |
| |
AASLD 2020 Nov 11-16 virtual
Reported by Jules Levin
Antoine Chaillon1, Adriane Wynn1, Tatyana Kushner2, Nancy Reau3 and Natasha Martin1,4, (1)Division of Infectious Diseases and Global Public Health, University of California San Diego, (2)Icahn School of Medicine at Mount Sinai, (3) Medicine Department, Rush University, (4)Population Health Sciences, University of Bristol, United Kingdom
Antoine Chaillon1, Adriane Wynn1, Tatyana Kushner2, Nancy Reau3 and Natasha Martin1,4, (1)Division of Infectious Diseases and Global Public Health, University of California San Diego, (2)Icahn School of Medicine at Mount Sinai, (3) Medicine Department, Rush University, (4)Population Health Sciences, University of Bristol, United Kingdom
Background: Prevalence of Hepatitis C Virus (HCV) among pregnant women in the United States (U .S.) U .S. doubled nationally from 2009-2014 (~0 .7%), yet many remain undiagnosed. Studies have found screening in pregnant women cost-effective, but none explicitly examined rescreening strategies. Screening pregnant women is not recommended by the Society of Maternal-Fetal Medicine, and although new draft recommendations by the Center for Disease Control (CDC) and U .S. Preventive Services Taskforce recommend screening in pregnant women, explicit polices surrounding rescreening may differ. We assessed the cost-effectiveness of HCV antenatal rescreening for women in the U.S. previously screened during pregnancy and without evidence of past HCV exposure.
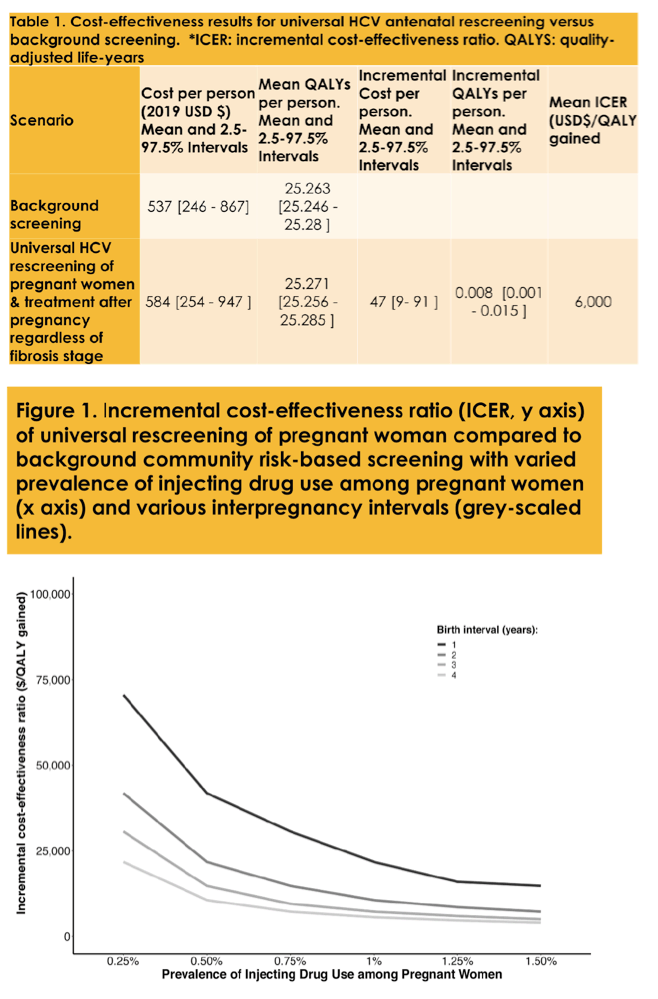
Methods: A HCV natural history Markov model was used to evaluate the cost-effectiveness of universal HCV rescreening among pregnant women previously screened during pregnancy and without evidence of past HCV exposure in the U.S. compared to background risk-based screening from a public sector health care payer perspective. At baseline we assumed no treatment restrictions by fibrosis stage, 1.25% with ongoing injecting drug use risk, and 3 year average interpregnancy interval, but varied in sensitivity analyses (0 .5-1 .5% injecting drug use, 1-4 year interpregnancy interval). We assessed cost (USD$) and health outcomes (quality-adjusted life years, QALYs) over a lifetime horizon. We assess mean incremental cost-effectiveness ratios (ICERs) under a willingness to pay threshold (WTP) of $50,000/QALY gained.
Results: Universal rescreening was cost-effective (mean ICER $9,390 /QALY gained). Rescreening remained cost-effective in settings with lower prevalence of ongoing risk (0 .5%) and with lower interpregnancy intervals (1 year). In settings with fibrosis restrictions (METAVIR F1 and beyond, or F2 and beyond), rescreening was cost-effective under a $50,000 WTP unless both prevalence of injecting drug use was 0 .5% and interpregnancy interval was 1 year.
Conclusion: Universal rescreening for HCV among pregnant women in the U.S. is likely cost effective and should be recommended nationall .



|
| |
|
 |
 |
|
|