 |
 |
 |
| |
Summary from virtual CROI 2020
for HIV and liver disease
HIV and the liver: what is new?
|
| |
| |
Jurgen K. Rockstroh M.D., Professor of Medicine
University of Bonn, Germany
Correspondence:
Prof. Dr. J.K. Rockstroh
Department of Medicine I
University of Bonn
Sigmund-Freud-Str. 25
53105 Bonn
Germany
Introduction
This year, following the worldwide spread of COVID-19, including first cases in Boston, a last minute decision changed the CROI 2020 meeting from a physical to a virtual meeting. The final decision - clearly a very difficult one - was only made on Friday 6th March. CROI are therefore to be congratulated on pulling a virtual conference together so quickly. Abstracts are available on the CROI website and posters have been uploaded as PDF files. Nearly all talks were streamed in real time, and oral presentations are now available as webcasts (see www.croiconference.org). The oral liver session even included a very animated live Q and A session where almost all speakers were able to address questions sent in by email during the live stream of their respective talks. The oral liver session included several highlights this year including reports on outbreaks of HIV and HCV as well as continued new HCV infections in intravenous drug user communities and networks, data on the HCV prevalence in newborn babies in New York State, data on risk factors for HCV reinfections, impact of external HCV introductions complicating HCV microelimination, treatment data for DAA therapy in acute HCV infection and risk factors for development of fatty liver disease in HIV-coinfection (1-7). This report aims at summarizing the main findings around viral hepatitis coinfection but also on new data around fatty liver disease in HIV-infected.
What continues to fuel the HCV-epidemic?
Misuse of opioids is rapidly becoming a global epidemic with >80% of drug seizures in Africa/Asia in part due to increased use of opioids to manage pain and expansion of heroin trafficking routes. Little is known about the drug using networks in these settings, which contribute to HIV/HCV transmission. The study presented at CROI therefore studied prevalence and predictors of incident HIV and HCV infections in PWIDs from New Delhi in India and the role of egocentric, sociometric and sociospatial networks on transmission (2). India indeed, has become home of the largest number of opioid users globally with an estimated 2.14 million PLHIV (prevalence 0.22%). 2512 people who inject drugs (PWID) were recruited (2017-19) into a cohort by a chain referral approach. Index participants were asked to name and recruit people they injected with in the past month (egocentric network of the index). Each recruit was asked to name and recruit their recent injection network members (egocentric network of recruit; sociometric network of index). Biometrics was used to identify duplicates and cross-network linkages. Participants underwent a survey and blood draw semi-annually. Blood was tested for HIV and HCV antibodies, HIV RNA and HCV RNA. Network viral load was calculated as the number of egocentric network members with HIV RNA>150 copies/ml. Poisson regression was used to identify predictors of incident HIV.
At baseline, 37% had HIV infection (928/2506) of whom only 7.4% were virologically suppressed; HCV prevalence was 65.1% (1634/2512),the proportion of individuals with chronic HCV (HCV-RNA positive) was 79.6%; recent heroin and other opioid use were 26.6% and 95.3%, respectively. 787 of 1578 HIV negative at baseline had at least one follow-up. 14 were confirmed dead. The median number of times injected in the prior 6 months was 360 (IQR 160-540). 42% shared syringes in the prior 6 months. The HIV and HCV incident infections during follow-up are summarized in table 1.
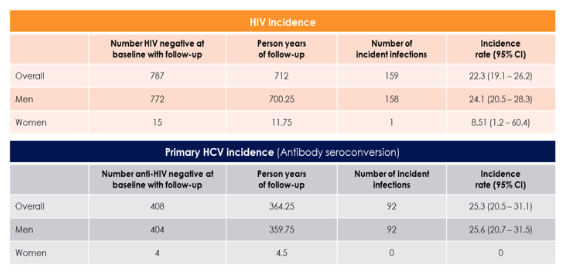
The majority of incident HIV cases were directly connected to at least one viremic person in their egocentric network. Predictive factors were recent injection/needle sharing, injection frequency, network viremia, sociometric path distance and injecting at spatial hotspot.
These data clearly highlight that epidemic control of HIV and HCV will not be possible without addressing the growing burden of these diseases in PWIDS also in low and middle income countries. Networks are dynamic and complicated and can contribute to new incident infections. The importance of harm reduction programs, HIV and hepatitis testing and high rates of virological suppression was also underlined from the results of a study which reported back from the recent increase in HIV diagnoses among people who inject drugs (PWID) in Cabell County (1). Clearly, cluster and outbreak response require increased coordination and creativity to improve service delivery to vulnerable communities. In places where these services become jeopardized or services are no longer financed, risk of viral outbreaks remains high.
Another group which is still contributing to new incident HCV infections are HIV positive as well as HIV negative men who have sex with men (MSM). An interesting study on risk factors for HCV reinfections among HIV-infected MSM was presented from New York (4). The HCV reinfection rate among HIV-infected MSM in NYC has previously been reported to be 4.4/100 PY. The main behavioral risk factors for primary HCV infection are receipt of semen into the rectum with condomless anal intercourse (semen in rectum) and/or sex while using crystal methamphetamine (sex on CM). Risk factors for HCV reinfection however are unknown. Therefore this study aimed at assessing behavioral risk factors in the context of HCV reinfections which is important to know to understand which interventions might be needed to prevent further HCV transmissions. Overall, 244 HIV-infected MSM in NYC with clearance of primary HCV between 1 Jan 2000 and 31 Dec 2018 were enrolled into this cohort study. Start of observation was defined as end of treatment, or first aviremic measurement in spontaneous control and the end of observation was defined as clinical onset of reinfection, or last undetectable viral load in those not reinfected. Each participant was prospectively queried about: 1) semen in rectum; 2) sex on crystal meth (CM); and 3) injection use of CM. Semen in rectum was significantly associated with 1st HCV reinfection in Cox Proportional Hazards Model. The findings from the multivariate analysis are shown in table 2. The multivariable Cox proportional hazards model included all three behavioral risk factors as independent exposure variables, adjusted for age, race, ethnicity, and year of HCV clearance.
Table 2: Mulivariate analysis
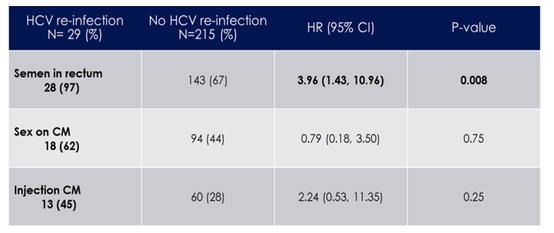
These results clearly underline that recommending condom use has not worked to prevent HCV. Indeed, new interventions are needed to prevent deposition of seminal HCV into the rectum. Currently, behavioral interventions are studied within the Swiss cohort study on HCV microelimination in HIV-positive MSM in Switzerland which may help to better define potential future interventions.
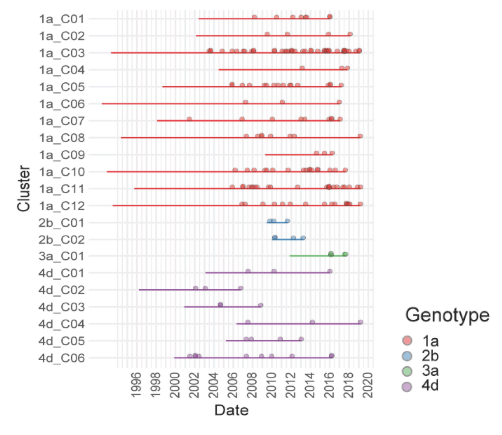
The topic of HCV reinfections among MSM remains of great interest as it represents one of the biggest barriers in achieving HCV microelimination in this particular patient group. With recent studies demonstrating that early DAA combination therapy for recently acquired hepatitis C infection may reduce new incident HCV infections by 50%, the question becomes is there still a risk of new HCV infections through external HCV introductions, particularly with a travel enriched and community event driven social agenda among MSM (8-10)? Along these lines the Amsterdam group presented results from a phylogenetic study which tries to address where the new HCV infections come from and whether they are phylogenetically related or not clustered and the role of external introductions over time (5). HVR1 sequences of 232 primary HCV infections and 56 reinfections were obtained from 244 MSM in care in Amsterdam. Maximum-likelihood phylogenies were constructed for each HCV genotype separately, and time-scaled phylogenies were constructed using a Bayesian coalescent approach. Transmission clusters were determined by Phydelity, which utilizes a statistically-principled and phylogeny-informed framework. The proportion of unclustered sequences over time was calculated using year-specific transmission trees inferred from sequences up to that year. For subtype 1a (n=191) the investigators found 12 transmission clusters ranging from 3 to 8 sequences. Transmission clusters of subtypes 2b and 3a were introduced more recently than clusters of subtypes 1a and 4d. For subtype 1a and 4d, clusters were introduced between 1998 and 2011, whereas for subtype 2b and 3a this was between 2009-2012 (see figure 1). Some transmission clusters appear extinct currently ((2b_C01, 2b_C02, 4d_C02, 4d_C03).
Figure 1: Transmission cluster characteristics
The proportion of external introductions has clearly increased over time as can be seen in figure 2.
Figure 2: Proportion external introductions increases over time
These data suggest that local micro-elimination will be complicated if contribution of external introductions is high. Also high DAA uptake and therefore less local virus will make it more likely to acquire external virus. Clearly, having similar treatment guidelines or at best universally immediate treatment of all new incident hepatitis C cases within Europe would be instrumental for achieving HCV microelimination in this patient group.
From abstract: The proportion of unclustered sequences has increased among HCV infections in recent years. The most likely explanation for this is that transmission of local strains has declined as a result of intense treatment efforts whereas external (possibly international) introductions of HCV into the MSM population in Amsterdam has increased. Frequent international transmission events will complicate national micro-elimination efforts and therefore international collaboration combined with international scale-up of treatment of all diagnosed HCV infections (including reinfections) is important.
Is there a need for newborn HCV screening?
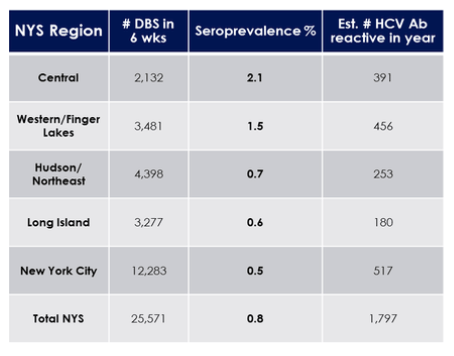
HCV infections in New York State have been rising among young adults due to increased injection drug use. In 2018 in NYS (excluding NYC), 61% of new female cases were in women of child-bearing age (15-44 years old). Increased HCV infections in this age group are concerning as 6% of HCV RNA-positive pregnant women will transmit HCV to their baby. To plan effective public health actions, accurate HCV prevalence rates among pregnant women are needed; however, many HCV infections go undiagnosed and unreported. Babies passively acquire maternal IgG antibodies. Therefore, testing newborn blood for HCV antibodies can reveal moms serostatus. The aim of the presented study was therefore to perform a large-scale HCV serosurvey of pregnant women in NYS by testing newborn dried blood spots (DBS) using a high-throughput, low-cost Luminex HCV immunoassay (3).
All DBS submitted to NYS's newborn screening program over 6 weeks were sampled by punching a 3mm circle into microplates. Aggregate data on birth weight, gestational age and mother's county of residence were recorded, and samples were blinded. A generic patient code was included to identify duplicate samples. HCV antigen-coupled beads were used to test eluted blood for HCV antibodies using a low-cost (<$0.80/well) newly developed Luminex-based immunoassay in 384-well plates. Repeated median fluorescence intensity (MFI) >1000 was considered HCV antibody reactive. Newborn testing revealed a high HCV Prevalence of 0.8% in pregnant women from NY State. The seroprevalences found by region are shown in table 3.
Table 3: HCV seroprevalence by NYS region
These findings clearly suggest that seroprevalence is high enough to recommend regular screening. Under consideration of the almost 1800 HCV-positive mother identified and an estimated spontaneous HCV clearance rate of 50% there still would be 900 HCV RNA positive mothers which would lead with a given perinatal transmission rate of 6% to 54 HCV infected babies. This is clearly much higher than what has been seen for HIV where in NYS of 343 HIV-exposed newborns none was infected with HIV. Maternal HCV detection and potentially DAA treatment once established for treatment during pregnancy (trials are currently being conducted) could be pivotal in preventing further HCV cases in newborn.
Is shorter treatment duration of acute or recently acquired hepatitis C possible?
Several small non-randomized studies on shortened duration therapy for acute and recently acquired HCV infection with direct-acting antiviral agents (DAAs), have shown high efficacy rates but there are also other studies with shorter treatment durations which demonstrated lower SVR rates because of increased risk for HCV relapse particularly in patients with high baseline HCV viral load (11-13). Therefore, until now guidelines have remained conservative in their recommendations with no currently approved regimens for this indication.
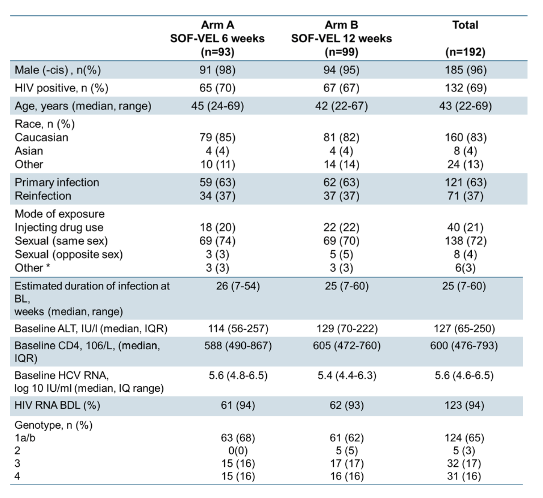
The REACT study was an NIH-funded multicentre international, open-label, randomised, phase 4 non-inferiority trial examining the efficacy of short course (6 weeks, Arm A) versus standard course (12 weeks, Arm B) therapy with sofosbuvir/velpatasvir for recently acquired HCV infection (estimated duration of infection <= 12 months) (6). Randomisation was at week 6 and stratified by site and HIV status. The primary endpoint was sustained virologic response at 12 weeks post-treatment (SVR12) reported in the intention-to treat (ITT) population. A total of 250 participants were planned for enrolment and DSMB analysis was scheduled after the first 50 participants in each arm reached SVR12. Following the initial DSMB review, a second review was scheduled following 60 participants through SVR12 in each arm. At second review (May 2019), the DSMB then recommended study cessation for evidence of inferiority in the short arm (A). All patients subsequently received the longer treatment duration. At CROI the final results of the trial were presented. The baseline characteristics of the 192 subjects from the primary analysis are summarized in Table 4.
Table 4: Baseline characteristics of the REACT study
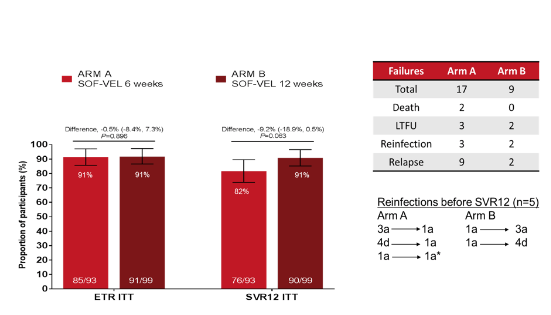
The majority of patients was male (96%), HIV-coinfected (69%) and had a genotype 1 infection (65%). At end of treatment no difference in end-of-treatment response rates was observed between the two treatment arms (see figure 3). However 12 weeks after stopping therapy SVR rate in the shorter treatment arm of 6 weeks SOF/VEL was considerably lower than in the longer treatment arm of 12 weeks. This was statistically however, not significant in the ITT analysis. However, in the modified ITT analysis which excluded LTFU (5), death (2) and confirmed reinfections (5) the difference in SVR rates 89 vs 98% became statistically significant (p=0.021). The main difference was the relapse rate of 9% in the short arm versus 2% in the standard arm. HCV reinfections occurred in both arms, 3 in Arm A and 2 in arm B. In 4 individuals reinfection was diagnosed after genotype switch. In the 5th case a genotype 1a was found with both infections. Reinfection with a different GT 1a strain was confirmed by showing a genetic distance in core-E2 by 11%.
Figure 3: Results: ITT analysis (n=192)

Of note the median HCV-RNA at baseline in the 11 relapsers was relatively high with 6.7 log. Level of adherence was numerically higher in the short treatment duration arm making this an unlikely explanation for the higher relapse rate. Indeed, a >90% adherence was found in 93% of study participants and only 5% were lost to follow-up. Overall, 11 SAEs (1 treatment related) were documented. There were no treatment related discontinuations. 3 deaths were documented which all occurred post treatment. In summary, the relapse rate of 9% in the 6 week arm was deemed unacceptably high compared to 2% in the standard arm. Despite the higher relapse in short arm, overall treatment in REACT was highly successful and safe supporting the feasibility and importance of treating early after new HCV infection.
What is new in treatment of chronic hepatitis C?
Direct acting antiviral (DAA) therapies for the treatment of chronic HCV infection offer excellent sustained virologic response (SVR) rates, regardless of HIV infection. Drug interactions with antiretrovirals (ARVs) however, remain a complicating factor when treating HIV/HCV coinfected patients with DAA. Ledipasvir is an inhibitor of P-gp and BCRP intestinal efflux transporters, resulting in increased exposure to tenofovir when dosed concomitantly with tenofovir disoproxil fumarate (TDF)-containing regimens. There is potential for further increased exposure when TDF is part of a pharmacologically boosted ARV regimen or in the case of coadministration with other comedications known to increase TDF exposure. Clinicians have approached this drug interaction differently, with some switching the ARV regimen and some continuing while monitoring renal function. Switches in ARV regimens due to drug interactions with DAA may put patients at risk of HIV treatment failure and has been reported to increase DAA failure (14). Indeed, in this study from an urban clinic in Baltimore, Maryland, patients who had changes to their HIV treatment regimen to avoid the potential for drug interaction with HCV DAA were less likely to achieve SVR compared to those who were on stable ART (14). The reason for this observation was not clear because incidence of blips in HIV RNA to detectable levels during HCV treatment was similar in both groups. The authors hypothesized that the combination of a relatively new antiretroviral regimen and a new HCV‐DAA regimen may have led to confusion about which medications to take or not take in the period surrounding an ART switch and HCV‐DAA initiation (15). The aim of the current study was therefore to assess the proportion of patients who are free of HIV treatment failure during the study period. The study design is shown in figure 3.
Figure 3: Study design of the switch registry
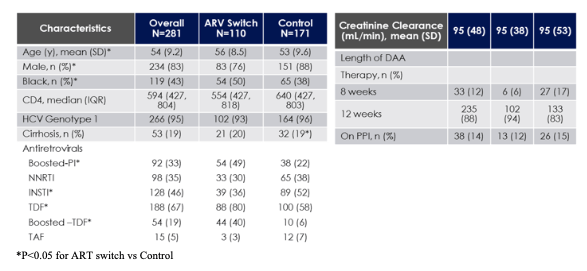
Overall, 281 subjects were enrolled, 110 in the switch arm and 171 in the control arm. The baseline characteristics of the study subjects are listed in table 5.
Table 5: Baseline characteristics in the switch registry
*P<0.05 for ART switch vs Control
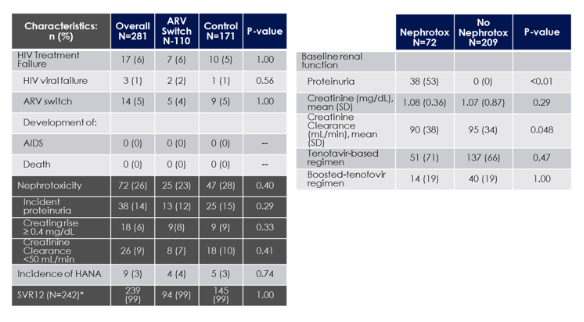
Patients who had their ARVs switched prior to DAA therapy did not experience more HIV treatment failure than controls. Switches were more common in patients on PI or TDF. HIV virologic failure was rare. HIV treatment failure was primarily due to ARV switches made while on DAA therapy. One switch occurred in a patient meeting nephrotoxicity criteria. Nephrotoxicity criteria were met in 72 (26%) of patients. These events were independent of ARV switch and were not associated with TDF or boosted protease containing regimens. Nephrotoxicity was more common in patients with lower baseline creatinine clearance or baseline proteinuria, although it was not associated with discontinuation of ARV or DAA therapy. HCV treatment response was excellent in this real world cohort of patients with HIV/HCV co-infection, and independent of ARV switch. The various outcome studied are summarized in table 6.
Table 6: Outcomes in the switch registry
Very rarely, patients with HIV/HCV coinfection undergoing all oral DAA combination therapy fail their DAA-based therapy. Indeed at this year CROI several real-life cohort reports were presented which looked at SVR12 rates in HIV/HCV coinfected patients which were treated with modern all oral DAA pangenotypic regimens. Two Spanish studies, one on outcome of 12 weeks SOF/VEL therapy in coinfected subjects (n=115) as well as one on 12 weeks glecaprevir/pibrentasvir therapy also in HIV/HCV coinfected subjects (n=104), reported over 93% and 97% HCV cure rates in ITT analysis, respectively (16,17).
In the "Interactive case-based workshop: What`s eating your liver?" David Wyles presented a patient who failed 4 previous HCV therapies including two all oral DAA regimens (SOF/LDV and SOF/VEL + ribavirin (18). The patient was then successfully cured with a triple drug combination of sofosbuvir, glecaprevir/pibrentasvir and 1200 mg of ribavirin for 24 weeks. A similar regimen was used in a GT1b patient with prior failure to three other HCV regimens who also achieved SVR12 and was just published very recently as a case report (19). In general, there is very little clinical data to guide retreatment of multiple DAA regimen failure. But three drug class therapy with ribavirin for as long as possible is the general rule and at least successful in reported individual cases. Resistance testing seems to have a very limited to no role in selecting therapy and is mostly only done for research questions. Late HCV RNA recurrences are almost always reinfections and can be separated from relapses by either genotype switch or phylogenetic analysis looking at genetic distance to the baseline viral strain.
CROI: HCV Retreatment, Multiple DAA Failures - Case Study (03/18/20)
What was new for hepatitis B?
One big challenge in the area of hepatis B prevention is the significantly lower immune response to HBV vaccination in particular in patients with HIV, HCV and on chronic dialysis. Current HBV vaccines have a positive response rate upwards of 85% in the general population, but the same vaccines only provide immunity for 20-70% of people living with HIV and 40-60% for the HCV population, emphasizing the need for improved hepatitis B vaccines. At this year CROI, results after vaccination with a novel, adjuvanted 2-dose HBV vaccine series (HepBCpG) were presented for these vulnerable populations (20). Previous studies with HepB-CpG demonstrated improved immune response (>90%) in non-HIV and non-HCV cohorts. This study now evaluated the immune response to HepB-CpG among HIV and HCV patients at an outpatient virology clinic. HIV and HCV patients who received at least one dose of HepB-CpG between October 1, 2018 and September 30, 2019 were included into the analysis. HBV vaccinations were given a minimum of 4 weeks apart and timed with routine clinical appointments. HBV surface antibody was tested at least 4 weeks after last immunization with a result ≥10mIU/mL deemed immune. The baseline characteristics of the different study groups are summarized in table 7.
Table 7: Baseline characteristics
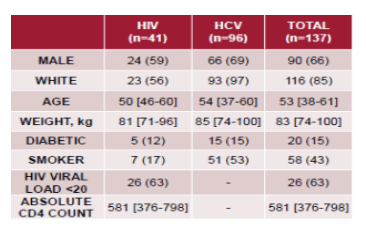
Currently, 29 (71%) of the HIV cohort have been tested for immunity and 25 (86%) are immune. Another 42 (44%) have been tested in the HCV cohort and 34 (81%) are immune. Of the 71 (52%) that were tested for immunity, 16 (23%) were found to be immune after just one dose, and a total positive immune response in 59 (83%). Of the 25 immune patients with HIV, 17 (68%) were non responders to a full, prior HBV vaccination series No adverse events reported From the HCV cohort, 3 patients have died due to unrelated causes. The immune response results are captured in figure 4.
Figure 4: Immune responses for the different study groups
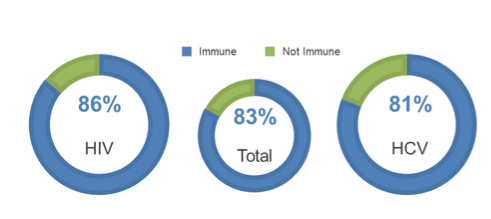
In summary, these results show an overall immune response to HepB-CpG of 83%, which is considerably higher than historical data using the non-adjuvanted vaccine. As part of a robust immunization program to protect HIV and HCV patients, HepB-CpG may become an alternative to the traditional HBV vaccination series.
Since hepatitis B vaccination can be co-administered with hepatitis A vaccine, this offers the opportunity to check and vaccinate against both hepatitis viruses simultaneously. This is particularly important in light of the recent outbreaks of hepatitis A virus (HAV) infection among men who have sex with men, which occurred globally in many different countries including the USA. An estimated critical immunity threshold against HAV is ≥ 70% to prevent outbreaks in MSM populations. National HIV Pre-exposure prophylaxis (PrEP) and post exposure prophylaxis (PEP) guidelines however, do not recommend HAV serology testing among MSM for PrEP/PEP initiation. At New York City sexual health clinics (SHC), all patients initiating PrEP or PEP receive HAV serology testing. At CROI a study was presented from NYC which aimed at determining the prevalence of HAV immunity among MSM initiating PrEP/PEP at sexual health clinics and determine subsequent HAV vaccine uptake (21). Electronic medical record (EMR) data was extracted for HIV-negative MSM PrEP/PrEP patients who had HAVST for the first time at SHC from September 2016 to March 2019, with a follow up through July 2019. Demographics, immunization history and EMR administered vaccines were analyzed, respectively. Patients reactive for HAV IgG were considered immune. Patients were considered vaccinated against HAV if they received at least one dose of HAV vaccine (Havrix™) or two doses of hepatitis A/B combination vaccine (Twinrix™) at the clinic (or self-reported vaccination at other clinics).
Overall, 4233 subjects were enrolled into the study. At time of PrEP/PEP initiation, 65% of MSM were immune to HAV (n=2733). On bivariate analysis, immunity to HAV varied by age, race/ethnicity, country of birth, and HAV vaccination status. Of 1500 MSM patients not immune to HAV, 49.6% (745/1500) received ≥1 dose of Havrix (n= 455) or Twinrix (n=290) within a year after HAV serology. At time of PrEP/PEP initiation, a total of 2437 (57.6%) patients self-reported receiving hepatitis A vaccination at non-NYC SHC settings; 36.8% (897/2437) were not immune. Table 8 summarizes the hepatitis A vaccinations over time.
Table 8: hepatitis A vaccinations over time in the NYC cohort.
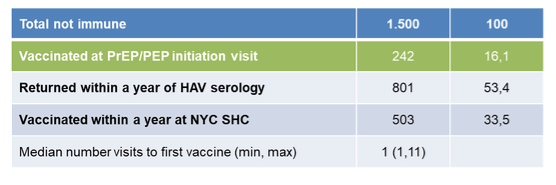
In summary, at time of PrEP/PEP initiation, HAV immunity among this NYC MSM cohort was below the critical immunity threshold of 70% against HAV. Subsequent HAV vaccination of half of the non-immune cohort likely increased MSM cohort immunity to ≥ 70% as one dose of Havrix was 95% immunogenic to HAV. Patient self-report of vaccination to HAV was not a reliable method within this study to assess patient's immunity to HAV.
HIV and fatty liver disease: what are the driving factors?
Nonalcoholic fatty liver disease (NAFLD) - ranging from steatosis to steatohepatitis to fibrosis - is a major cause of liver disease in HIV and is gaining increasing interest. It is however, still unclear how much HIV itself contributes and how much may derive from ART-associated toxicity. While simple steatosis is regarded as relatively benign, hepatic fibrosis has been linked to all-cause and liver-specific mortality. The natural history of NAFLD in HIV, including which patients are likely to develop clinically overt disease, is not well known. At CROI now findings from a study, which collected repeated liver biopsies samples from a clinical trial of HIV-associated NAFLD was presented (7). Aim of the study was to identify predictors of fibrosis presence and progression (7). The liver biopsies were all collected in a randomized trial of the growth hormone-releasing hormone analogue tesamorelin to treat NAFLD in HIV. In this study, the authors previously found that tesamorelin reduced liver fat and prevented fibrosis progression (22). After 12 months, 35% of individuals receiving tesamorelin and 4% receiving placebo had a hepatic fat fraction of less than 5% (p=0·0069). Sixty-one participants with HIV and NAFLD were randomized to tesamorelin or placebo for 12 months. NAFLD was defined as hepatic fat fraction (HFF) ≥ 5% by magnetic resonance spectroscopy in the absence of active hepatitis B or C or excess alcohol consumption. Individuals with cirrhosis were excluded. Participants underwent liver biopsy at baseline and 12 months; histologic evaluation was performed by a single expert pathologist blinded to treatment and biopsy order.
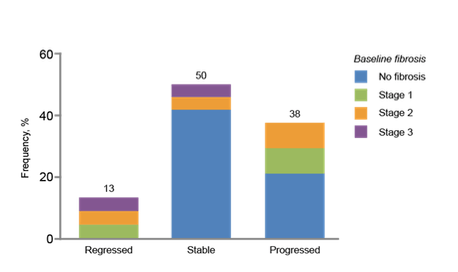
Among 58 participants with baseline biopsies, 43% had hepatic fibrosis (stage 1, 36%; stage 2, 40%; stage 3, 24%). Fibrosis was associated with greater visceral fat content at baseline (284 ± 91 cm2 vs. 212 ± 95 cm2, P = 0.005), but not subcutaneous fat or BMI. While hepatic fat fraction did not differ between groups, individuals with fibrosis had higher NAFLD Activity Score (3.6 ± 2.0 vs. 2.0 ± 0.8, P < 0.0001), ALT (41 ± 30 U/L vs. 23 ± 8 U/L, P = 0.002), and AST (44 ± 27 U/L vs. 24 ± 10 U/L, P = 0.0003). Among 24 participants randomized to placebo with paired liver biopsies, 38% had progression of fibrosis over 12 months. The progression of fibrosis in HIV-Associated NAFLD
for the placebo-treated participants over 12 months is shown in figure 5. Mean rate of fibrosis progression was 0.2 ± 0.8 stages per year. A total of 56% of participants with fibrosis progression had no evidence of fibrosis at baseline.
Figure 5: Progression of fibrosis in the placebo-treated participants
Higher visceral fat content at baseline (306 ± 119 cm2 vs. 212 ± 89 cm2, P = 0.04) was the only clinical predictor of fibrosis progression, which remained significant upon adjusting for BMI, HFF, and NAS Score (see table 9). Age, sex, race, duration of HIV, and CD4 count did not relate to fibrosis presence or progression. In conclusion, high rates of liver fibrosis presence and progression were observed in a cohort with HIV and NAFLD. Individuals with greater visceral fat content at baseline were more likely to have baseline fibrosis and progression of fibrosis. Patients with signs of fatty liver disease need to carefully monitored and targeted for hopefully new interventions in the future.
Table 9: Odds of Fibrosis Progression in Placebo-Treated Participants
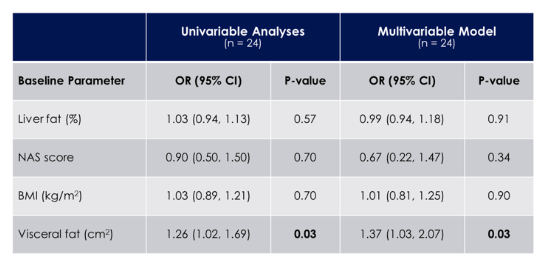
Odds ratios correspond to a 1-unit change in the baseline parameter, except for visceral fat which is calculated based on a 25 cm2 increment.
Non-alcoholic fatty liver disease (NAFLD) is strongly associated with cardiovascular disease (CVD) in the general population. In people living with HIV (PLWH), this association has not yet been investigated yet. At CROI findings from a large cohort study on the impact of NAFLD and liver fibrosis on cardiovascular risk in PLWH was presented for the first time (23). 1410 HIV infected patients from three prospective cohorts (LHIVPA in Palermo, LIVEHIV in Montreal, MHMC in Modena) were evaluated with Transient Elastography (TE). Exclusion criteria were: significant alcohol intake, coinfection with hepatitis B or C virus and failure of TE examinations defined as IQR value > 30%. NAFLD and significant liver fibrosis were defined as controlled attenuation parameter (CAP) ≥ 288 dB/m and as liver stiffness measurement (LSM) > 7 kPa, respectively. Cardiovascular risk was assessed with Atherosclerotic Cardiovascular Disease (ASCVD) Risk Estimator, according to American College of Cardiology, in patients aged 40 - 75 years, and categorized as: low if < 5%, borderline if 5 - 7.4%, intermediate if 7.5 - 19.9% and high if ≥ 20%. Patients with previous cardiovascular events were considered as high risk, regardless of age. Overall, 941 HIV mono-infected subjects were included with a mean age of 53 years. The majority were males (74%) and 98% were on ART. 423 (45%), 128 (13,6%), 260 (27,6%) and 130 (13,8%) patients were categorized at low, borderline, intermediate and high ASCVD risk, respectively. Previous cardiovascular events were recorded in 8.5% of study subjects. Prevalence of NAFLD and liver fibrosis by transient elastography were 20% and 17%, respectively. The distribution of ASCVD risk categories by NAFLD and liver fibrosis status are shown in figure 6. Overall, intermediate and high risk ASCVD risk were more frequent in patients with NAFLD (p< 0.0001) and liver fibrosis (p<0.05).
Figure 6: Distribution of ASCVD risk categories by NAFLD and liver fibrosis status
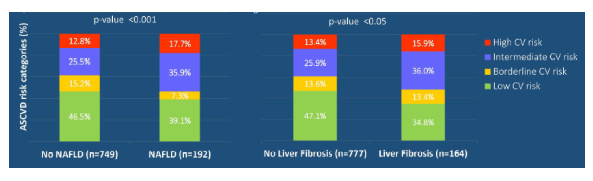
Both NAFLD and liver fibrosis are predictors of cardiovascular disease in PLHIV. Therefore, HIV mono-infected patients with NAFLD and/or liver fibrosis should always be screened for cardiovascular risk. Prevention of CVD, possibly with lifestyle modifications, should be strengthened in PLHIV with NAFLD, in particular in those with longer HIV duration.
Summary
• An extremely high incidence of HIV and HCV can be observed in PWIDs from India. Improved access to testing, harm reduction services and successful antiviral therapy are crucial in minimizing network linked transmission.
• Sex, with receipt of semen into the rectum, rather than drug (methamphetamine) use, was the behavior associated with HCV reinfection in HIV-infected MSM in NYC.
• As condom use has not been successful as an HCV prevention strategy, there is a need for novel interventions to prevent deposition of seminal HCV into the rectum.
• External HCV introductions are rising among MSM in Amsterdam with a high degree of MSM specific clustering.
• Newborn DBS testing using a Luminex-based immunoassay is an effective way to generate a population-based estimate of HCV burden among pregnant women.
• Short-term treatment of acute or recent hepatitis C with 6 weeks SOF/VEL leads to a higher relapse rate than the standard 12 weeks of SOF/VEL. Thus, shorter treatment durations are not recommended with this DAA combination.
• Patients who have their ARVs switched prior to DAA therapy do not experience
more HIV treatment failures than patients who have no ART switch prior to starting DAA therapy. Overall, switches are more common in patients on boosted PI or TDF.
• Real-life cohorts demonstrate very high HCV cure rates after treatment with modern pangenotypic all oral DAA therapy in HIV/HCV coinfection.
• After multiple DAA treatment failure three drug class therapy with ribavirin for as long as possible is the general rule and at least successful in reported individual case reports.
• The immune response to a novel, adjuvanted 2-dose HBV vaccine series (HepBCpG) is considerably higher than historical data using the three-dose vaccine, and is demonstrating a response in previous non-responders.
• HAV serological testing for MSM initiating PrEP/PEP and subsequent hepatitis A vaccination of non-immune patients is an effective intervention to prevent future HAV outbreaks.
• National guidelines for PrEP and PEP should consider adding Hepatitis A serology testing among MSM initiating PrEP and PEP and vaccinating the non-immune to HAV.
• Patients with HIV and NAFLD are at high risk of liver fibrosis presence and progression. Higher visceral fat content at is the only clinical predictor of fibrosis progression.
• Both NAFLD and liver fibrosis are predictors of cardiovascular disease in PLHIV. Prevention of CVD, possibly with lifestyle modifications, should be strengthened in PLHIV with NAFLD, in particular in those with longer HIV duration.
References
1. McClung P et al. Large HIV outbreak among people who inject drugs, West Virginia, 2018-2019. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 123
2. Clipman S et al. Explosive HIV and HCV epidemics driven by network viremia among PWID 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 147
3. Styer LM et al. Newborn testing reveals high HCV seroprevalence in pregnant women from New York State. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 125
4. Factor SH et al. Sex, not drug use, is driving HCV reinfection among HIV-infected MSM in New York City. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 594
5. Koopsen J et al. HCV transmission among MSM: External introductions could complicate microelimination. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 124
6. Matthews G et al. Inferiority of short duration sofosbuvir-velpatasvir for recent HCV (REACT Study). 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 121
7. Fourman L et al. Clinical predictors of liver fibrosis presence & progression in HIV-associated NAFLD. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 124
8. Boerekamps A, et al. Declining Hepatitis C Virus (HCV) Incidence in Dutch Human Immunodeficiency Virus-Positive Men Who Have Sex With Men After Unrestricted Access to HCV Therapy. Clin Infect Dis 2018;66:1360-1365.
9. Braun D, et al., for the Swiss HIV Cohort Study. A treatment as prevention trial to eliminate HCV in HIV+ MSM: The Swiss HCVREE trial. 25th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2018, Boston; abstract LB87
10. Garvey L, et al. Fall in HCV incidence in HIV+ MSM in London following expansion of access to DAA therapy. 26th Conference on Retroviruses and Opportunistic Infections, March 04-07, 2019, Seattle; abstract 0085
11. Naggie S, et al. Acquired Immunodeficiency Syndrome Clinical Trials Group (ACTG) A5327 Study Team. Ledipasvir/Sofosbuvir for 8 Weeks to Treat Acute Hepatitis C Virus Infections in Men With Human Immunodeficiency Virus Infections: Sofosbuvir-Containing Regimens Without Interferon for Treatment of Acute HCV in HIV-1 Infected Individuals. Clin Infect Dis. 2019;69(3):514-522.
12. Boerekamps A, et al. Treatment of acute hepatitis C genotypes 1 and 4 with 8 weeks of grazoprevir plus elbasvir (DAHHS2): an open-label, multicentre, single-arm, phase 3b trial. Lancet Gastroenterol Hepatol. 2019;4(4):269-277.
13. Rockstroh J et al. Ledipasvir-sofosbuvir for 6 weeks to treat acute hepatitis C virus genotype 1 or 4 infection in patients with HIV coinfection: an open-label, single-arm trial. Lancet Gastroenterol Hepatol. 2017;2(5):347-353.
14. Falade-Nwulia O, et al. High hepatitis C cure rates among black and nonblack human immunodeficiency virus-infected adults in an urban center. Hepatology. 2017;66(5):1402-1412.
15. Mauss S et al. A MULTICENTER REGISTRY IN PATIENTS WITH HIV/HCV COINFECTION ON LEDIPASVIR/SOFOSBUVIR. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 573
16. Aldámiz-Echevarría T et al. Real-world effectiveness of sofosbuvir/velpatasvir for hepatitis C virus infection. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 572.
17. Domínguez-Domínguez L et al. Real-worl effectiveness of glecaprevir/pibrentasvir for hepatitis C virus infection. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 571.
18. Wyles D. When at third you don`t succeed. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 11.
19. Fierer D and Wyles D. Re-treatment of Hepatitis C Infection After Multiple Failures of Direct-acting Antiviral Therapy. Open Forum Infectious Diseases 16th March 2020
20. Kimball B et al. Effectiveness of the novel adjuvanted hepatitis B vaccine among HIV and HCV patients. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 605
21. Mikati T et al. Suboptimal imunity to hepatitis A among NYC MSM initiating PrEP or PEP, 2016-2019. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 617
22. Stanley TL, et al. Torriani M, Kleiner DE, Hadigan CM, Grinspoon SK. Effects of tesamorelin on non-alcoholic fatty liver disease in HIV: a randomised, double-blind, multicentre trial. Lancet HIV 2019;6(12):e821-e830.
23. Mazzola G et al. NAFLD and liver fibrosis predict high cardiovascular risk in HIV-monoinfected subjects. 27th Conference on Retroviruses and Opportunistic Infections, March 08-11, 2020, Boston; abstract 543
|
| |
|
 |
 |
|
|