 |
 |
 |
| |
Results from a PrEP Demonstration Project for At-Risk Cisgender Women in the US – PrEP/PEP at CROI
|
| |
| |
CROI 2020
Reported by Jules Levin
At CROI 2020:
Longer Term Efficacy and Safety of F/TAF and F/TDF For HIV PrEP: DISCOVER Trial Week 96 Results - (03/11/20)
IMPACT OF PrEP AND TasP ON INCIDENCE OF HIV DIAGNOSES IN 48 HIGHEST-BURDEN US AREAS (03/16/20)
DISCOVER: 96-Week Follow-up of Black and Hispanic/Latinx Study Participants - (03/11/20)
DRUG LEVELS, ADHERENCE, AND RISKS FOR LOW ADHERENCE IN THE DISCOVER PrEP STUDY (03/13/20)
WEEKLY ORAL ISLATRAVIR PROVIDES EFFECTIVE PEP AGAINST IV CHALLENGE WITH SIVMAC251 - (03/11/20)
FTC/TAF + BIC Postexposure Prophylaxis Protects Macaques Against Rectal SHIV Infection - (03/11/20)
F/TAF and F/TDF Efficacy and Safety as PrEP for 96 Weeks - (03/11/20)
Jill Blumenthal*1, Sheldon Morris1, Sonia Jain1, Feng He1, Ryan Kofron2, Eric Ellorin1, Jamila K. Stockman1,Gifty M. Ntim2,
Peter L. Anderson3, Richard Haubrich4, Katya Corado5, K. Rivet Amico6, David J. Moore1, Raphael J. Landovitz3
1University of California, San Diego, La Jolla, CA, 2University of California Los Angeles, Los Angeles, CA, 3University of Colorado, Denver, CO, 4Gilead Sciences, Foster City, CA, 5Harbor UCLA Medical Center, Torrance, CA, 6University of Michigan, Ann Arbor, MI

Abstract
Data on TDF/FTC PrEP use by cisgender women have largely been from Africa. We report the primary results from the first US demonstration project of oral PrEP among at-risk cisgender women.
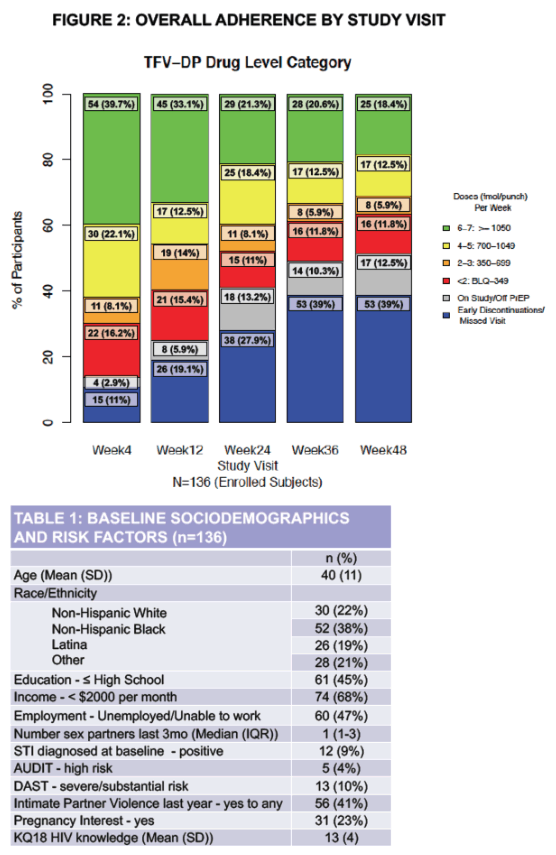
Adherence Enhancement Guided by Individualized Texting and Drug Levels (AEGiS) was a 48-week PrEP demonstration project in cisgender women ≥18 years old at-risk for HIV conducted at 5 Southern California sites. Adherence was supported using two-way text messaging (Individualized Texting for Adherence Behavior; ITAB) and titrated adherence counseling based on rapid-turnaround tenofovir diphosphate (TFV-DP) concentrations. Study visits occurred at baseline, week 4, week 12, then quarterly through week 48. Demographics were collected with computer surveys. Outcomes included PrEP adherence, retention and persistence. Adherence was assessed by quantifying TFV-DP concentrations in dried blood spots.
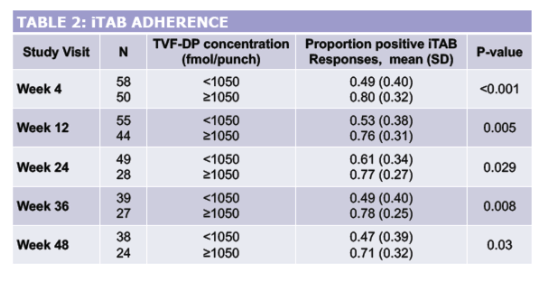
Concentrations ≥1050 fmol/punch were considered protective, suggesting ≥6 doses on average per week. Self-reported PrEP adherence was determined by the proportion of participants responding positively to daily iTAB text prompts over 30 days prior to study visits.
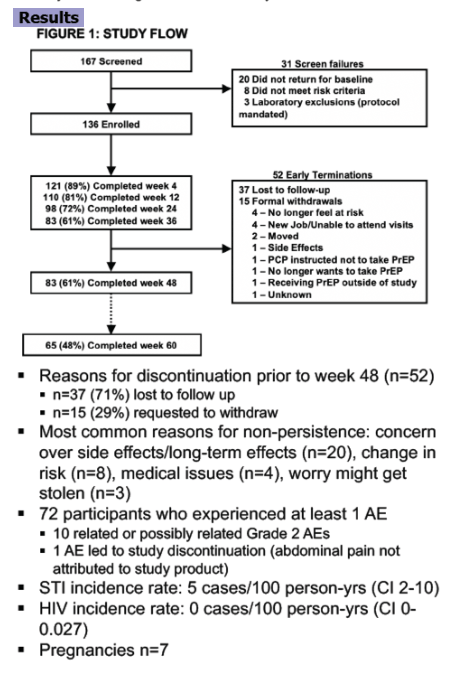
Between 6/2016 and 10/2018, 136 ciswomen enrolled with mean age 40 (SD 11); 38% were non-Hispanic (NH) Black and 19% Latina. Over 48 weeks, 84 (62%) participants were retained and 62 (74%) remained on PrEP. Over one-third (12/31) of those on study but off PrEP discontinued TDF/FTC due to self-reported side effects; one led to study discontinuation. Of 120 participants with drug concentrations measured, 67 (56%) had at least one protective concentration; 22 (18%) had consistently protective drug concentrations across all available study visits attended. For all visits, women with protective TFV-DP were more likely to have a higher proportion of positive iTAB responses compared to those with TFV-DP<1050 fmol/punch (p<0.05 at all visits except week 24). There were no incident HIV infections and 4 incident bacterial STIs.
Cisgender women in a PrEP demonstration project had mixed adherence and retention; many had non-protective TFV-DP concentrations and over 25% were lost to follow up. US PrEP programs may need to consider offering prevention alternatives for women who discontinue or struggle with PrEP adherence. In particular, integrating PrEP delivery within other valued medical or social services may promote and augment HIV prevention efforts.

|
| |
|
 |
 |
|
|