 |
 |
 |
| |
START OR SWITCH OF INTEGRASE INHIBITORS ON DEPRESSIVE SYMPTOMS IN WOMEN WITH HIV
|
| |
| |
CROI 2020
Reported by Jules Levin
Jane A. O'Halloran1, Kunbo Wang2, Dionna W. Williams3, Raha Dastgheyb3, Kathryn Fitzgerald3, Asante R. Kamkwalala3, Amanda B. Spence4, Anjali Sharma5, Deborah Gustafso
1Washington University in St Louis, St Louis, MO, USA,2Johns Hopkins University, Baltimore, MD, USA,3Johns Hopkins University School of Medicine, Baltimore, MD, USA,4Georgetown University, Washington, DC, USA,5Albert Einstein College of Medicine, Bronx, NY, USA,6SUNY Downstate Medical Center, Brooklyn, NY, USA,7University of Illinois at Chicago, Chicago, IL, USA,8Cook County Health & Hospitals System, Chicago, IL, USA,9University of North Carolina at Chapel Hill, Chapel Hill, NC, USA,10University of Miami, Miami, FL, USA
Abstract Body:
Depressive symptoms are associated with use of some antiretroviral therapy (ART) agents. Recently, concerns regarding neuropsychiatric symptoms with integrase strand transfer inhibitor (INSTI) use have been raised. We examined INSTI-associated changes on the profile of depressive symptoms in women with HIV (WWH).
Women's Interagency HIV Study (WIHS) participants who started or switched to INSTI-based ART and had two consecutive records (one before and one after the start/switch) with a completed Center for Epidemiologic Studies Depression Scale (CES-D) were included in the present analysis. We examined the adverse effects of INSTI start or first switch as a drug class on subscale-level (interpersonal, somatic, negative and positive affect) CES-D symptoms using linear mixed effects models adjusting for relevant covariates (e.g., age, race, substance use, body mass index, HIV RNA). Subsequent models examined each of the INSTIs separately.
1036 WWH, median age 48 (interquartile range 36, 60) years, 697 (67%) black, non-Hispanic were included in the analysis. Twenty-one percent were INSTI starts (30% raltegravir [RAL]; 29% elvitegravir [EVG]; 41% dolutegravir [DTG]) and the remainder of observations were switches to INSTI-based regimens (35% RAL; 27% EVG; 38% DTG). The majority of switches were from a protease inhibitor (PI) (56%) or a non-nucleoside reverse transcriptase inhibitor (NNRTI) (34%) based regimen. Overall, INSTI use was not associated with subscale changes in depressive symptoms after start/switch. Similarly, start/switch to individual INSTIs did not affect depressive symptoms. When analysis was restricted to those who switched to INSTIs from any ART, again no effects on depressive symptoms were observed. Similarly, switch from a NNRTI-based or PI-based regimen to any INSTI or individual INSTIs did not impact depressive symptoms. Starting any INSTI on the other hand improved positive affect, when stratified by INSTI type EVG but not RAL or DTG improved positive affect (p=0.004).
Among WWH, switching to INSTI based therapy did not have an impact of depressive symptoms. Initiation of INSTIs did result in improvement in positive affect symptoms and this was predominantly driven by EVG use.

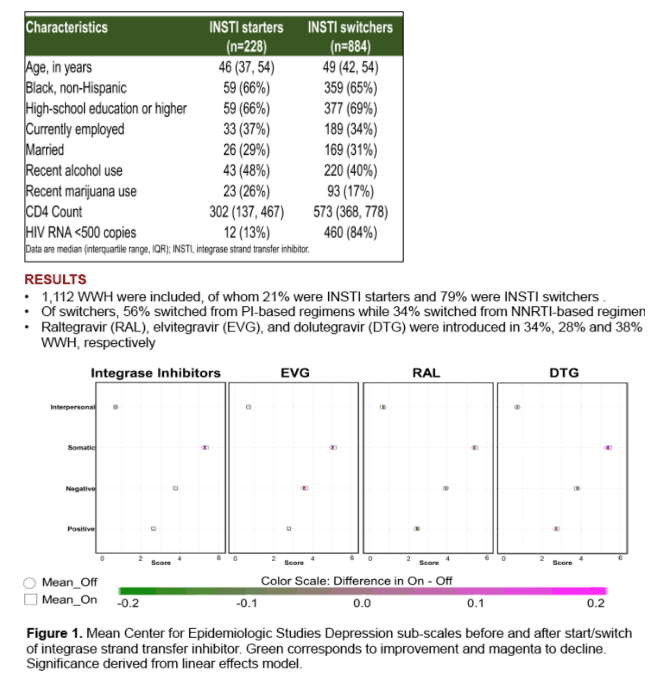

|
| |
|
 |
 |
|
|