 |
 |
 |
| |
COMBINED ACTIVE AND PASSIVE IMMUNIZATION IN SHIV-INFECTED RHESUS MONKEYS
|
| |
| |
CROI 2020
Dan Barouch1, Noe Mercado1, Abishek Chandrashekar1, Erica Borducchi1, Joseph Nkolola1, Maria Pau2, Hanneke Schuitemaker2, Merlin L. Robb3, Nelson L. Michael4
1Beth Israel Deaconess Medical Center, Boston, MA, USA,2Janssen Prevention and Vaccines, Leiden, Netherlands,3Henry M Jackson Foundation, Rockville, MD, USA,4Walter Reed Army Institute of Research, Silver Spring, MD, USA,5Gilead Sciences, Inc, Foster City, CA, USA
http://www.croiwebcasts.org/console/player/44684?mediaType=slideVideo&
Abstract
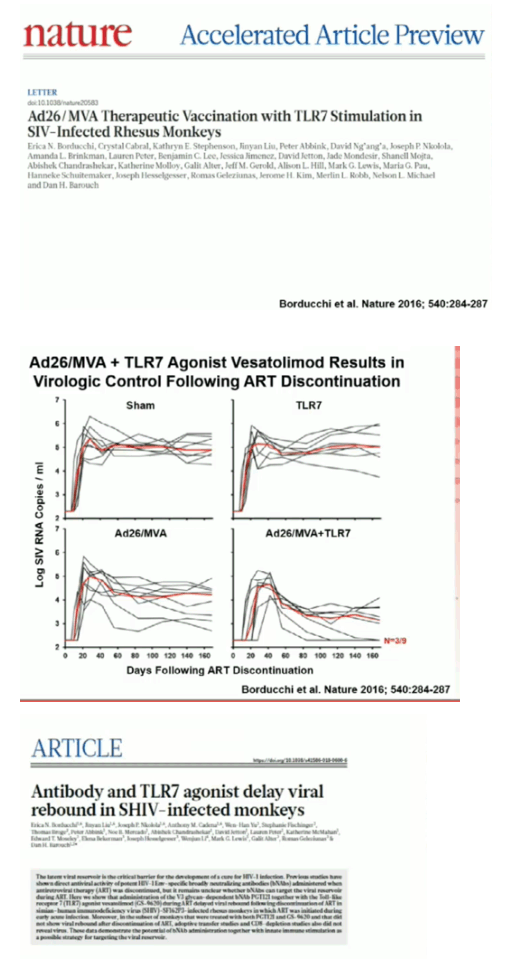
Our group and others have previously reported that therapeutic immunization can result in post-rebound virologic control in SHIV-infected rhesus monkeys following ART discontinuation, and that administration of broadly neutralizing antibodies (bNAbs) can delay or prevent viral rebound. The potential of combined active and passive immunization as an HIV-1 cure strategy has not previously been evaluated.
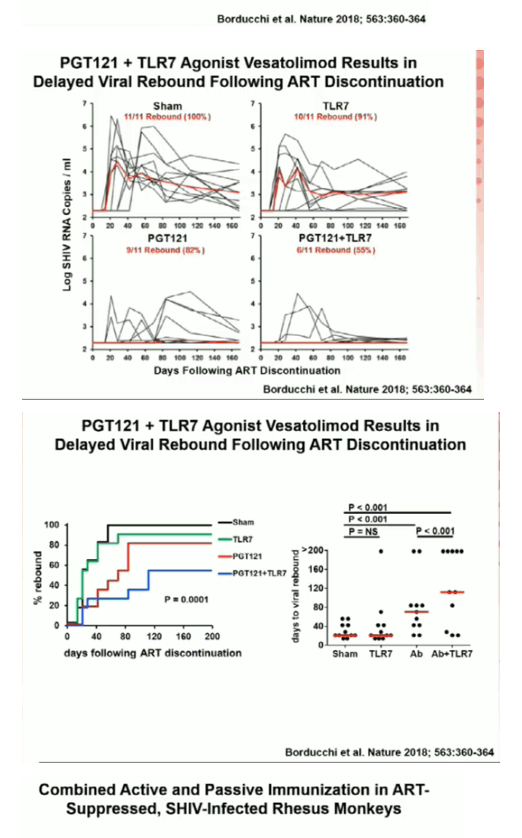
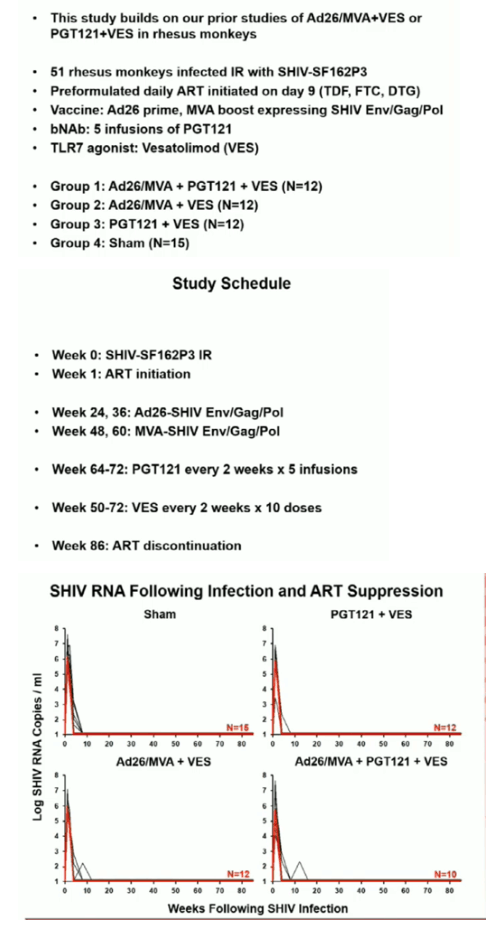
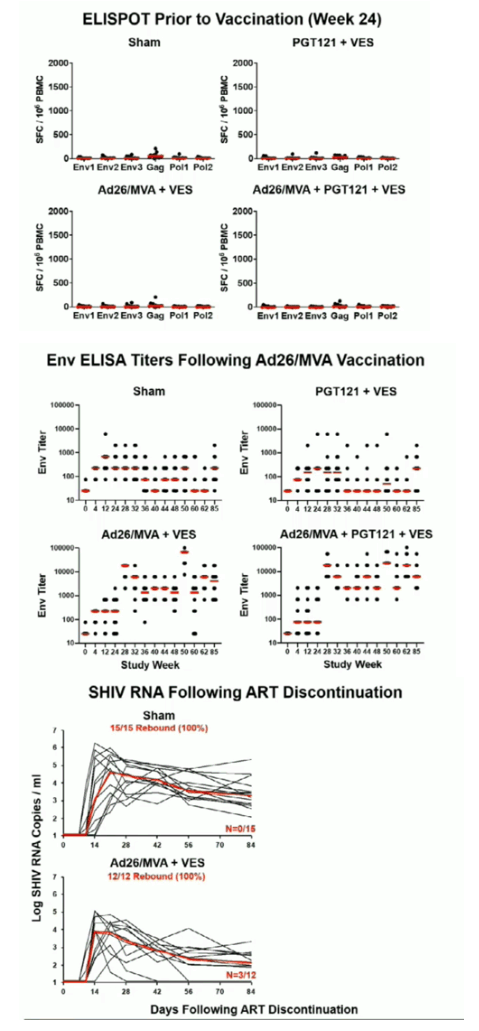
49 rhesus monkeys were infected with SHIV-SF162P3 and initiated ART (TDF/FTC/DTG) on day 9 of infection. Following 24 weeks of continuous daily suppressive ART, animals received 4 immunizations with Ad26/MVA vaccines at weeks 24/36/48/60 (N=12), 5 infusions of 10 mg/kg PGT121 every 2 weeks from weeks 64-72 (N=12), both Ad26/MVA vaccines and PGT121 (N=10), or sham controls (N=15). All groups except the sham controls received 10 doses of 0.15 mg/kg of the TLR7 agonist vesatolimod (VES) by oral gavage (every 2 weeks from weeks 50-72). At week 86, ART was discontinued and viral rebound was monitored for 140 days.
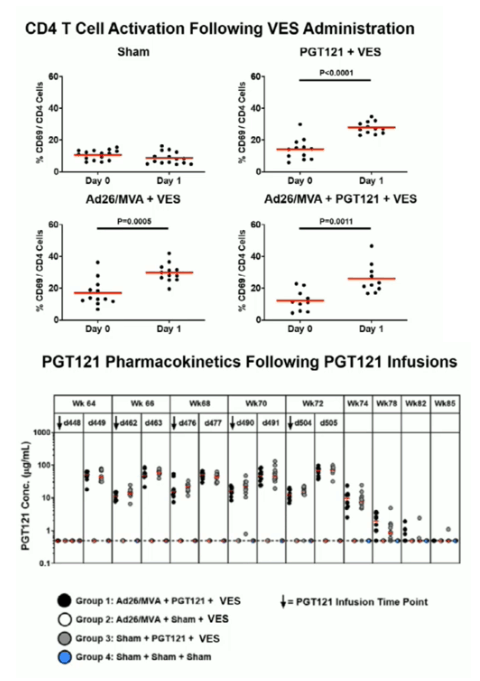
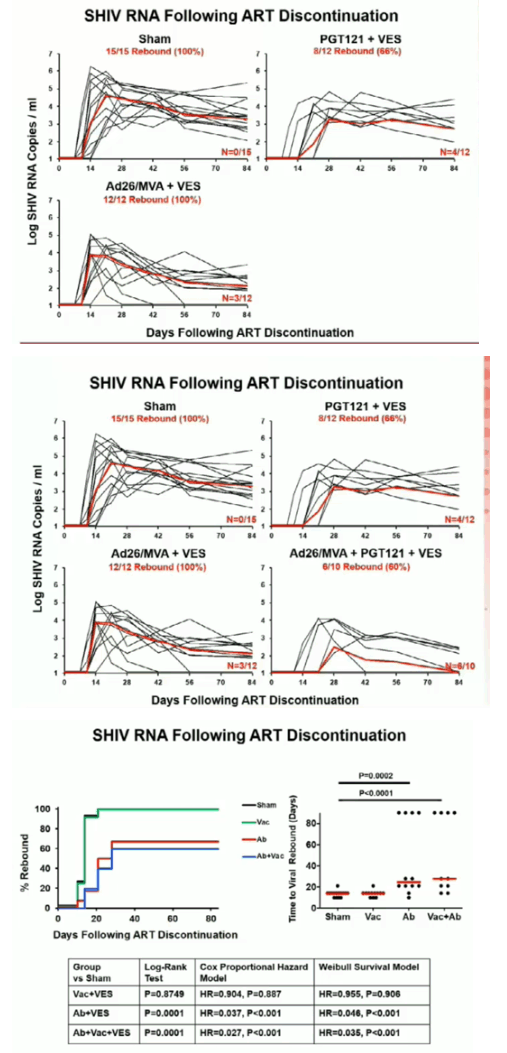
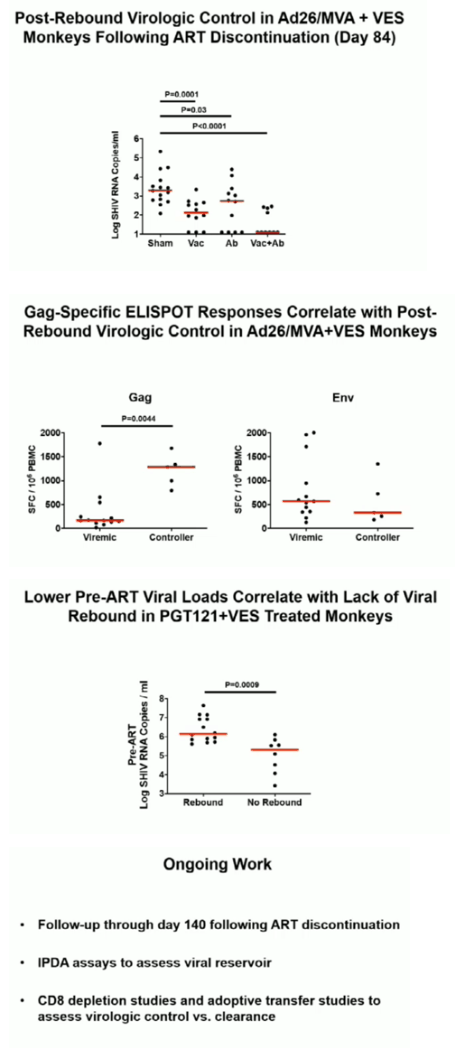
Ad26/MVA vaccination resulted in increased magnitude and breadth of SHIV-specific cellular and humoral immune responses. PGT121 infusion resulted in 14 weeks of therapeutic antibody levels followed by a decline to undetectable levels prior to ART discontinuation. VES administration led to activation of multiple cellular immune subsets including CD4+ T lymphocytes. Following ART discontinuation, 100% (15 of 15) of sham controls exhibited rapid viral rebound, and all animals in this group remained viremic by day 140 following ART discontinuation. 100% (12 of 12) of the Ad26/MVA + VES vaccinated animals also rebounded, but 3 animals demonstrated post-rebound virologic control to undetectable levels. In contrast, only 66% (8 of 12) of PGT121 + VES treated animals and 60% (6 of 10) of Ad26/MVA + PGT121 + VES treated animals rebounded (P=0.016, Fishers exact test compared with sham controls). Moreover, only 40% (4 of 10) of Ad26/MVA + PGT121 + VES treated animals were viremic by day 140 following ART discontinuation (P=0.001, Fishers exact test compared with sham controls).
Combined active and passive immunization with TLR7 stimulation resulted in both delayed viral rebound and post-rebound virologic control following ART discontinuation in SHIV-infected rhesus monkeys that initiated ART during acute infection. This multi-pronged approach represents a novel HIV-1 cure strategy.
Safety & Analytic Treatment Interruption Outcomes of Vesatolimod in HIV Controllers....http://www.natap.org/2020/CROI/croi_13.htm
SAFETY & PHARMACOKINETICS OF GS-9722 IN HIV-NEGATIVE PARTICIPANTS AND PEOPLE WITH HIV - (03/09/20)
GS-9722: First-in-Class, Effector-Enhanced, Broadly Neutralizing
Antibody for HIV Cure - and PGT121 in HIV+ / PGT121 bNAb in HIV+.....http://www.natap.org/2019/CROI/croi_110.htm
|
| |
|
 |
 |
|
|