 |
 |
 |
| |
SAFETY AND EFFICACY OF DTG VS EFV AND TDF VS TAF IN PREGNANCY: IMPAACT 2010 TRIAL
|
| |
| |
CROI 2020
Reported by Jules Levin
Lameck Chinula1, Sean S. Brummel2, Lauren Ziemba2, Lynda Stranix-Chibanda3, Anne Coletti4, Chelsea Krotje5, Patrick Jean-Philippe6, Lee Fairlie7, Tichaona Vhembo3
1University of North Carolina Project–Malawi, Lilongwe, Malawi,2Harvard T.H. Chan School of Public Health, Boston, MA, USA,3University of Zimbabwe, Harare, Zimbabwe,4FHI 360, Durham, NC, USA,5Frontier Science & Technology Research Foundation, Inc, Amherst, NY, USA,6DAIDS, NIAID, Rockville, MD, USA,7Wits Reproductive Health and HIV Institute, Johannesburg, South Africa,8Makerere University–Johns Hopkins University Research Collaboration, Kampala, Uganda,9University of California Los Angeles, Los Angeles, CA, USA,10Brigham and Women's Hospital, Boston, MA, USA,11University of North Carolina at Chapel Hill, Chapel Hill, NC, USA
Abstract
We compared the safety and virologic efficacy of dolutegravir (DTG) + emtricitabine (FTC)/tenofovir alafenamide fumarate (TAF) vs. DTG + FTC/tenofovir disoproxil fumarate (TDF) vs. efavirenz (EFV)/FTC/TDF in pregnant women.
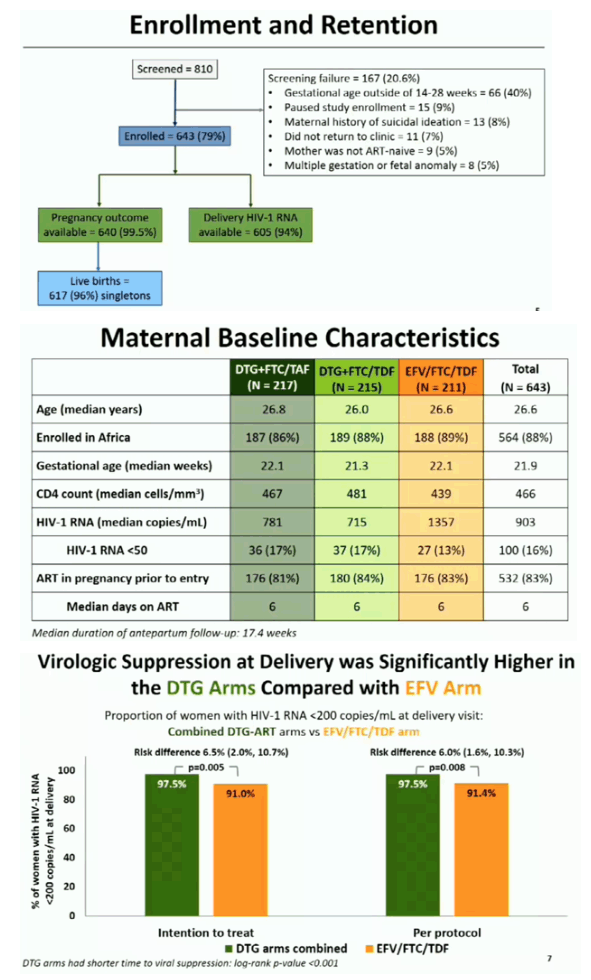
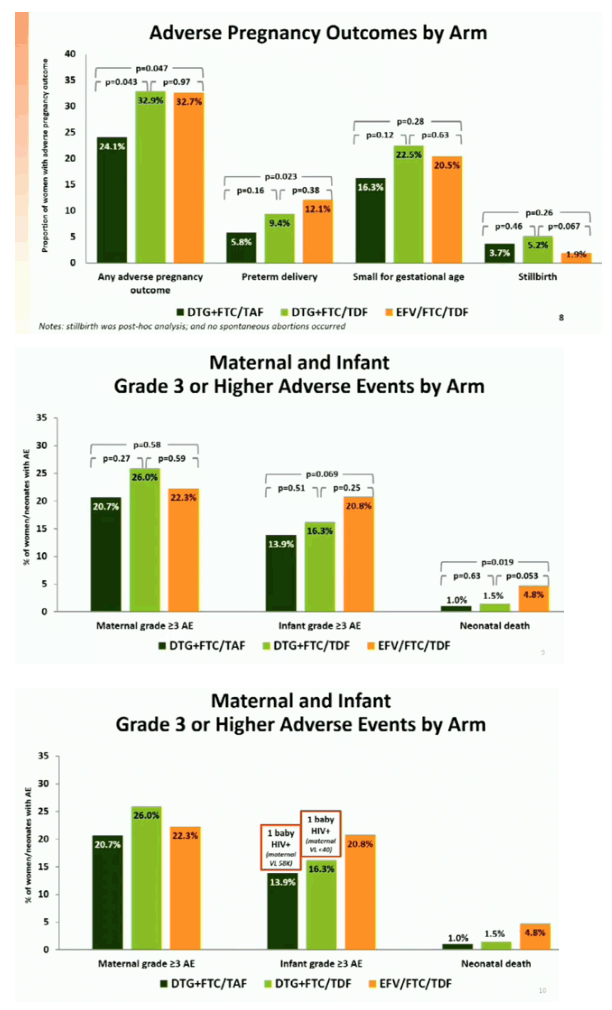
Pregnant women with HIV-1 in 9 countries were randomized 1:1:1 to start open-label DTG+FTC/TAF, DTG+FTC/TDF, or EFV/FTC/TDF at 14-28 weeks gestational age (GA). Up to 14 days’ pre-entry antiretroviral treatment (ART) was permitted. In primary efficacy analysis, we compared the combined DTG-containing arms to the EFV arm for non-inferiority (-10% margin), then superiority, with regard to delivery HIV RNA<200 cp/mL. Safety outcomes compared between all arms were a) composite adverse pregnancy outcome (preterm delivery [PTD]<37 weeks, small for GA [SGA]<10th centile, stillbirth [SB] or spontaneous abortion [SAB]); b) maternal grade>3 adverse event (AE) through 14 days postpartum; and c) infant grade>3 AE through 28 days. Neonatal death (NND, <28 days) was also evaluated.
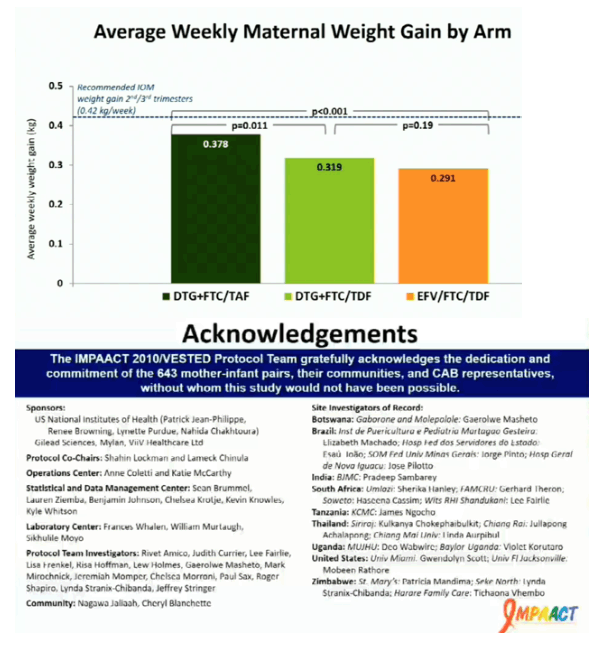
We randomized 643 women: 217 to DTG+FTC/TAF, 215 to DTG+FTC/TDF, and 211 to EFV/FTC/TDF. Baseline medians were: GA 21.9 weeks, HIV RNA 903 cp/mL, CD4 count 466 cells/uL; 83% took ART prior to entry (median 6 days). Median antepartum follow-up was 17.4 weeks. Delivery HIV RNA, available for 605 (94.1%) women, was <200 cp/mL in 395 of 405 (97.5%) in the combined DTG arms vs 182 of 200 (91.0%) in the EFV/FTC/TDF arm (difference 6.5% [95%CI 2.0%, 10.7%]; p=0.005). Pregnancy outcomes were available for 640 (99.5%). Fewer women in the DTG+FTC/TAF arm (24.1%) had an adverse pregnancy outcome than in DTG+FTC/TDF (32.9%, p=0.043) or EFV/FTC/TDF (32.7%, p=0.047) arms. Although SB was more frequent with DTG+FTC/TAF (3.7%) and DTG+FTC/TDF (5.2%) than EFV/FTC/TDF (1.9%) (all by-arm p-values 0.05; post-hoc), NND was more frequent with EFV+FTC/TDF (4.8%) than DTG+FTC/TAF (1.0%, p=0.019) or DTG+FTC/TDF (1.5%, p=0.053). Combined SB or NND rates were similar by arm (post-hoc analysis). At least one grade>3 AE occurred in 148 (23.0%) women and 105 (17.0%) infants (all by-arm p-values 0.05). Two babies were diagnosed with HIV at <14 days, one each in DTG+FTC/TAF and DTG+FTC/TDF arms (maternal delivery HIV-1 RNA 58,590 and <40 cp/mL, respectively).
DTG-containing ART started at GA 14-28 weeks had superior virologic efficacy at delivery to EFV/FTC/TDF. DTG+FTC/TAF had the lowest composite frequency of adverse pregnancy outcomes. Maternal and infant AE outcomes were similar by arm.
http://www.croiwebcasts.org/console/player/44813?mediaType=slideVideo&


|
| |
|
 |
 |
|
|