 |
 |
 |
| |
Barriers To Direct-Acting Antiviral Therapy Among HIV/HCV-Coinfected Adults
|
| |
| |
CROI 2020
Reported by Jules Levin
Gina M Simoncini1, Q Hou2, E Tedaldi1, K Carlson2, FJ Palella, Jr4, J Fuhrer5, RM Novak6, C Mayer7, DE Koren1, LA Battalora8, K Buchacz3, S Mayer9, S Purinton2, J Li3, for the HIV Outpatient Study Investigators 1Temple University, Philadelphia, PA, USA; 2Cerner Corp, Kansas City, MO, USA; 3Centers for Disease Control and Prevention, Atlanta, GA, USA; 4Northwestern University, Evanston, IL, USA; 5Stony Brook University, Stony Brook, NY, USA; 6University of Illinois at Chicago, Chicago, IL, USA; 7St. Joseph's Comprehensive Research Institute, Tampa, FL, USA; 8Colorado School of Mines, Golden, CO, USA; 9University of Illinois College of Medicine, Peoria, IL, USA
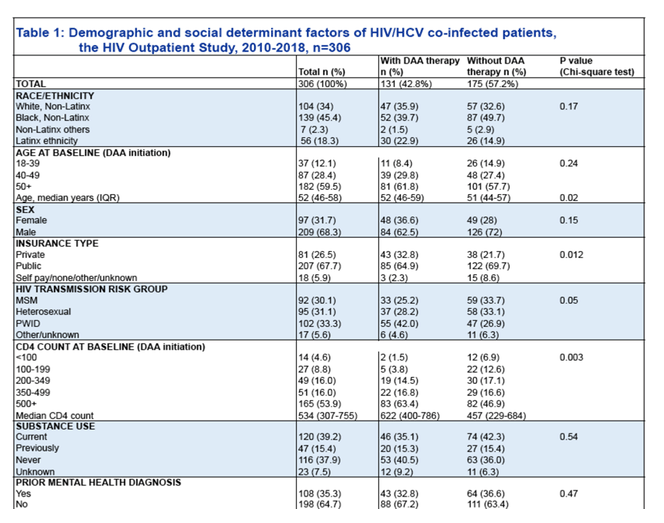
Only 42.8% of HIV/HCV-coinfected participants have been treated in the HOPS cohort. Factors associated with lower uptake of DAA therapy included younger age, sexual transmission as the HIV risk factor, lower CD4 range, and public or no insurance. Substance use, mental health diagnosis, race, and ethnicity were not associated with DAA treatment. Improved insurance coverage and expanded clinical care access to younger, presumably healthier individuals, is needed to bridge the treatment gap.
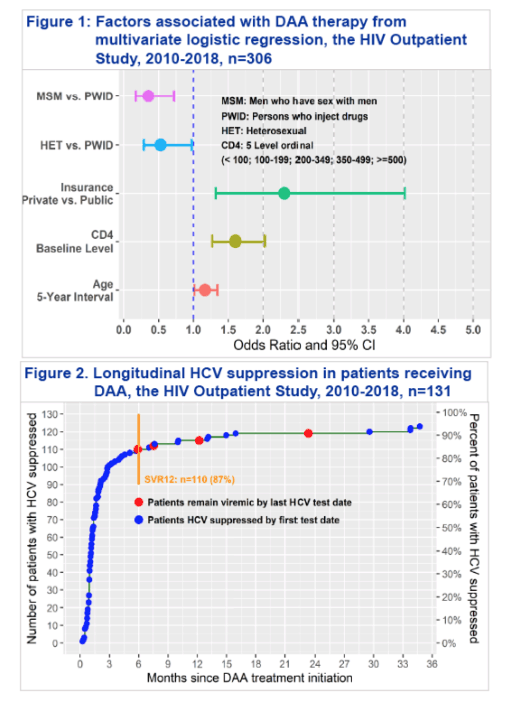
Among 306 eligible participants, median age was 52 years, median duration of follow up was 3.96 years, 97 (32%) were female, and 202 (66%) were non-white, 131 (42.8%) were prescribed DAA therapy, 127 (96.9%) had at least one follow-up HCV VL and 13 (9.9%) participants remained HCV viremic 12 months after initiating DAA therapy, resulting in an overall cure rate of 90.1%. DAA treatment was not associated with patients race and ethnicity (p=0.17), history of substance abuse (p=0.53), nor a mental health condition (p=0.43). Multivariable logistic regression analyses indicated that participants who were older (p=0.03), with health insurance (p=0.01), higher CD4 range (p<0.001), and injection drug use as the HIV risk category (p=0.03), were more likely to receive DAA therapy (Figure). Compared with the publicly insured, privately insured participants were more likely to receive DAA with an odds ratio of 2.30 (95% confidence interval: 1.32-4.12).


|
| |
|
 |
 |
|
|