 |
 |
 |
| |
TFV-DP in DBS for pregnant/postpartum
adolescent and young women on PrEP in Africa
|
| |
| |
CROI 2020
Reported by Jules Levin
Peter L. Anderson1, Lynda Stranix-Chibanda2, Sharon Huang3, Sybil Hosek4, Deborah Kacanek3, TeaclerNematadzira2, Frank Taulo5, Violet Korutaro6, ClemensiaNakabiito7, MaseboleMasenya8, Kathryn Lypen9, Nahida Chakhtoura10, Hans M. Spiegel11, Benjamin H. Chi12, on behalf of the IMPAACT 2009 team
1University of Colorado Anschutz Medical Campus, Aurora, CO, USA, 2University of Zimbabwe, Harare, Zimbabwe, 3Harvard University, Cambridge, MA, USA,4John H. Stroger Jr. Hospital of Cook County, Chicago, IL, USA, 5Malawi College of Medicine-Johns Hopkins University Research Project, Blantyre, Malawi, 6Baylor College of Medicine Children's Foundation, Kampala, Uganda,7Makerere University, Kampala, Uganda, 8Wits Reproductive Health and HIV Institute, Johannesburg, South Africa, 9FHI 360, Durham, NC, USA, 10National Institute of Child Health and Human Development, Bethesda, MD, USA, 11Kelly Government Solutions, Contractor to DAIDS/NIAID/NIH/HHS, Rockville, USA, 12University of North Carolina at Chapel Hill, Chapel Hill, NC, USA

abstract
Pregnant/postpartum adolescent girls and young women (AGYW) in Africa are one of the populations at highest risk for HIV acquisition; yet, pharmacokinetic (PK) data for pre-exposure prophylaxis (PrEP) remains limited. Intracellular tenofovir-diphosphate (TFV-DP) concentration in red blood cells, measured via dried blood spots (DBS), has been used to monitor cumulative PrEP adherence in many settings.
The first phase of IMPAACT 2009 evaluated PK characteristics of daily oral PrEP (FTC 200mg/TDF 300mg) among pregnant/postpartum AGYW (16-24 years) in Malawi, South Africa, Uganda, and Zimbabwe. Daily FTC/TDF was administered under direct observation for 12 weeks in two groups: pregnant AGYW starting at 14-24 weeks gestation (pregnancy) or 6-12 weeks after delivery (postpartum). Weekly TFV-DP was measured from DBS using a validated liquid chromatography-tandem mass spectrometry assay. TFV-DP distributions were determined at 12 weeks and groups compared with the Wilcoxon test. Population PK models were fit to estimate half-life and steady state concentrations.
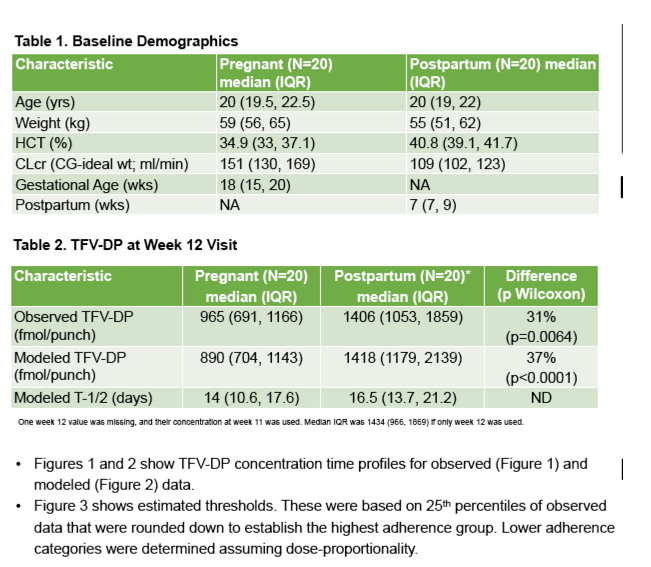
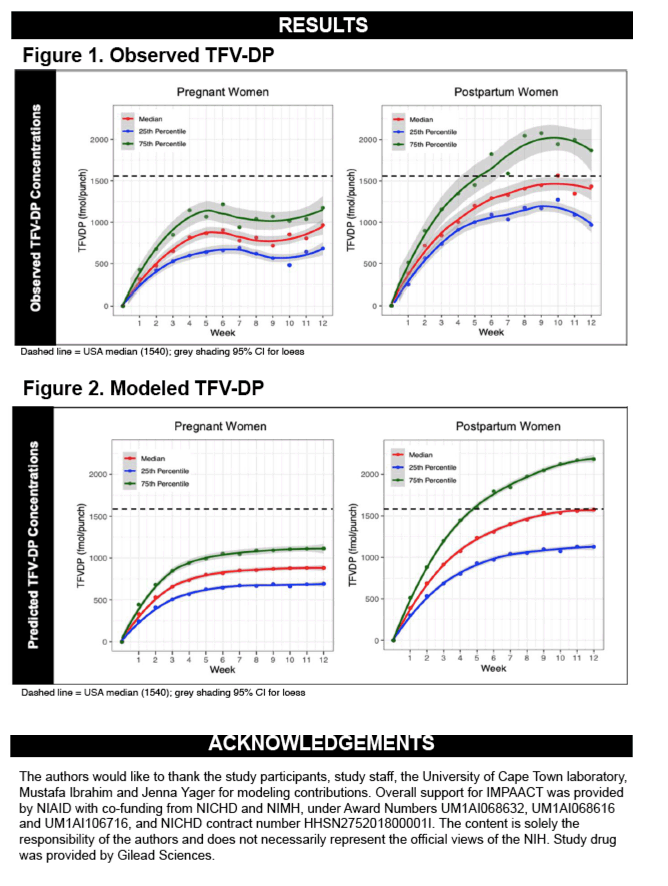
From March to June 2019, we enrolled 20 pregnant (median gestational age: 18 wks) and 20 postpartum (median time after delivery: 7 wks) women at a median age of 20 years (IQR:19,22). Of 3360 doses, 3348 (>99%) were directly observed. TFV-DP accumulated with a half-life of 15.3 days (95%CI: 12.8,17.8) in pregnancy and 18.0 days (95%CI: 15.3,20.7) postpartum, with steady state achieved by 8-10 weeks in both groups. Median TFV-DP was 965 fmol/punch (IQR: 691,1166) in pregnancy vs 1406 fmol/punch (IQR: 1053,1859) postpartum (p=0.006). Predicted median TFV-DP was 890 fmol/punch (IQR: 704,1143) in pregnancy vs 1418 fmol/punch (IQR: 1179,2139) postpartum (Figure). Two fetal demises (unrelated to study agent), two newborns <10%tile birthweight, and one preterm birth were recorded. No HIV transmissions occurred during follow-up.
Under conditions of near perfect adherence, TFV-DP in African AGYW was 31-37% lower in pregnancy than postpartum. With sequential measurements and a novel measure of cumulative drug exposure, these findings extend prior studies showing lower plasma TFV during pregnancy. There are few data correlating HIV protection and TFV-DP concentrations in women; however, our results suggest that strict adherence is needed during pregnancy. They also provide guidance for assessing PrEP adherence using TFV-DP levels in DBS for pregnant/postpartum African women.

|
| |
|
 |
 |
|
|