 |
 |
 |
| |
Tenofovir Diphosphate in Dried Blood Spots Predicts Virologic Failure and Resistance
|
| |
| |
CROI 2020
Poster pdf attaced here
Download the PDF here
Jose R. Castillo-Mancilla1, Yuan Zhao2, JaysinghBrijkumar3, Brent Johnson4, Alex Edwards2, Pillay Selvan3, MelendhranPillay3, PraviMoodley3, Mahomed-YunusMoosa3, Daniel R. Kuritzkes5, Henry Sunpath3, Lane R. Bushman1, Lucas Ellison1, Peter L. Anderson1, Vincent C. Marconi21University of Colorado Anschutz Medical Campus, Aurora, CO, USA, 2Emory University, Atlanta, GA, USA, 3University of KwaZulu-Natal, Durban, South Africa, 4University of Rochester, Rochester, NY, USA, 5Brigham and Women's Hospital, Boston, MA, USA

abstract
Tenofovir diphosphate (TFV-DP) in dried blood spots (DBS) is a measure of cumulative adherence and exposure to TFV-based antiretroviral therapy (ART). This adherence biomarker has been associated with viral suppression and found to predict future viremia in persons living with HIV (PLWH) receiving tenofovir disoproxil-fumarate (TDF)-based ART. However, little is known about its utility in the context of virologic failure (VF) and drug resistance in resource-limited settings.
Participants within a prospective clinical cohort of PLWH who initiated TDF-based ART in 2 clinical sites in KwaZulu-Natal, South Africa, were evaluated. DBS samples were collected from participants who received at least 6 months of ART and developed VF, defined as an HIV VL >1000 copies/mL (cases), and in a selected group of participants who had an HIV VL <1000 copies/mL (controls, matched by site, age, gender, race and duration of ART). Cases were categorized as having VF without resistance or having VF with resistance using genotypic resistance testing. Concentrations of TFV-DP in DBS were quantified using a validated LC-MS/MS method. One-way ANOVA was used to compare the concentrations of TFV-DP in DBS at the time of the last study visit between controls, participants with VF without resistance and participants with VF with resistance. Data are presented as mean [SD] or median (IQR).
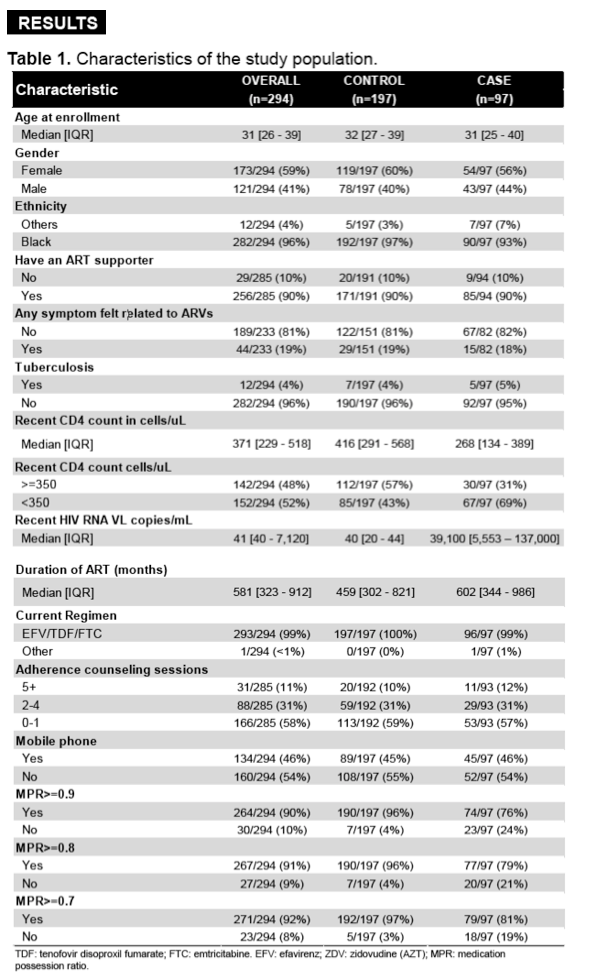
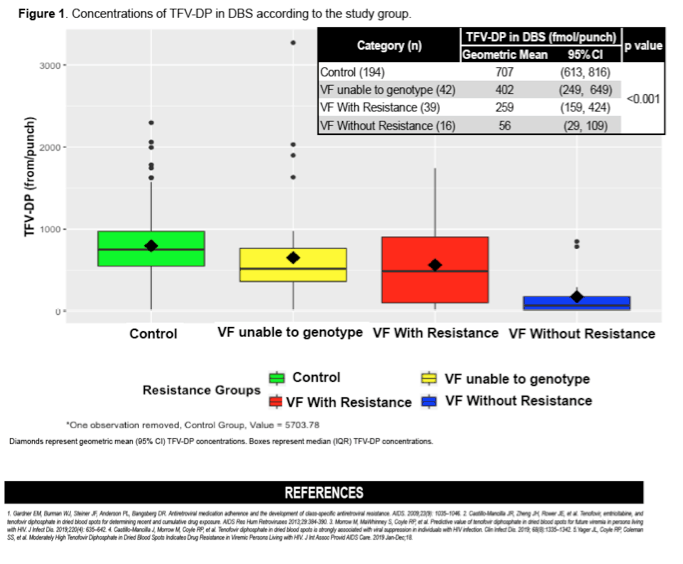
A total of 1000 participants (500 at each site) were enrolled in the cohort. Of these, 288 (45 cases) had available DBS samples, which were included in the analysis. Median age was 31 (26, 38) years and 170 (59%) were women. TFV-DP concentrations in DBS in controls were higher than in participants with VF with resistance and VF without resistance (808 [503] vs. 589 [495] vs. 527 [555] fmol/punch; P<0.01; Table), respectively.
TFV-DP in DBS was associated with VF and drug resistance in South African PLWH on TDF-based ART. Participants with VF who developed drug resistance had TFV-DP concentrations in the mid-range of cumulative exposure, but higher than those who did not develop resistance. These results suggest that moderate cumulative drug exposure is required to develop ART resistance. Future research on the clinical utility of TFV-DP in DBS to prevent the development of VF and drug resistance is needed.

|
| |
|
 |
 |
|
|