 |
 |
 |
| |
Predicted Rise in Diabetes Risk With
TAF/FTC+DTG Versus TDF/FTC+DTG
|
| |
| |
Download the PDF here
"Risks of metabolic syndrome, diabetes, and cardiovascular disease in ADVANCE trial"
"Estimated rates of diabetes, myocardial infarctions and other
NCDs from the ADVANCE trial"

CROI 2020, March 8-11, 2020, Boston
Mark Mascolini
Taking tenofovir alafenamide/emtricitabine plus dolutegravir (TAF/FTC+DTG) rather than tenofovir disoproxil fumarate (TDF)/FTC+DTG in the South African ADVANCE trial resulted in a significantly higher risk of clinical obesity and metabolic syndrome, and increases in visceral and subcutaneous adipose tissue (VAT and SAT) [1]. Those changes led to a predicted additional 3 cases of diabetes per 1000 people taking TAF/FTC+DTG versus TDF/FTC+DTG. Predicted risk of myocardial infarction (MI) or coronary death rose more with TAF/FTC+DTG than with TDF/FTC + efavirenz (EFV).
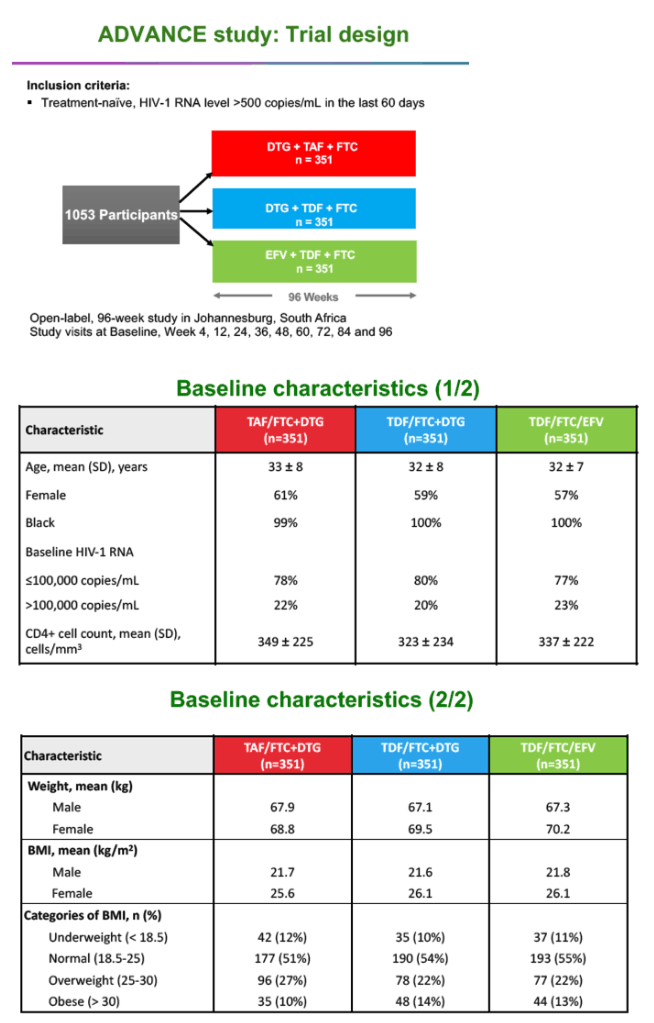
ADVANCE randomized 1053 antiretroviral-naive South Africans to TAF/FTC+DTG, TDF/FTC+DTG, or TDF/FTC+EFV in an open-label design. The three study arms did not differ substantially in age (about 32 years), proportions of women (about 59%), proportions with a pretreatment viral load above 100,000 copies (about 21%), or pretreatment CD4 count (about 340). About one quarter of participants (mostly women) were overweight at study entry, while about 12% were obese.
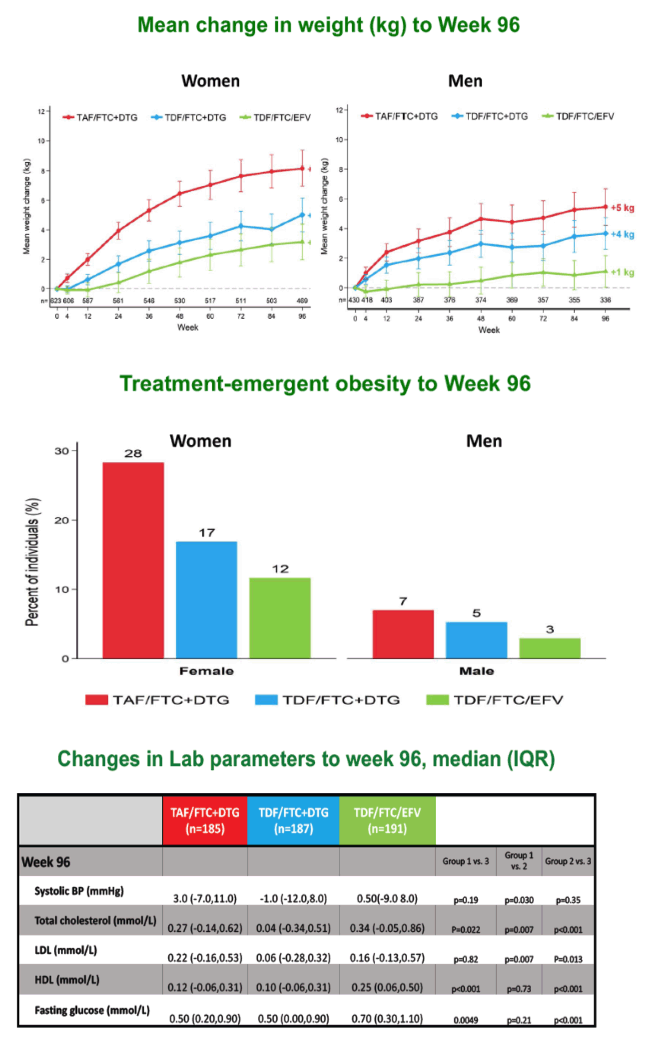
Women and men randomized to TAF/FTC+DTG gained more weight across 96 weeks than people in the other two study arms. Among women, 28% taking TAF/FTC+DTG had treatment-emergent obesity by week 96, compared with 17% taking TDF/FTC+DTG and 12% taking TDF/FTC+EFV. Respective proportions with treatment-emergent obesity among men were 7%, 5%, and 3%.
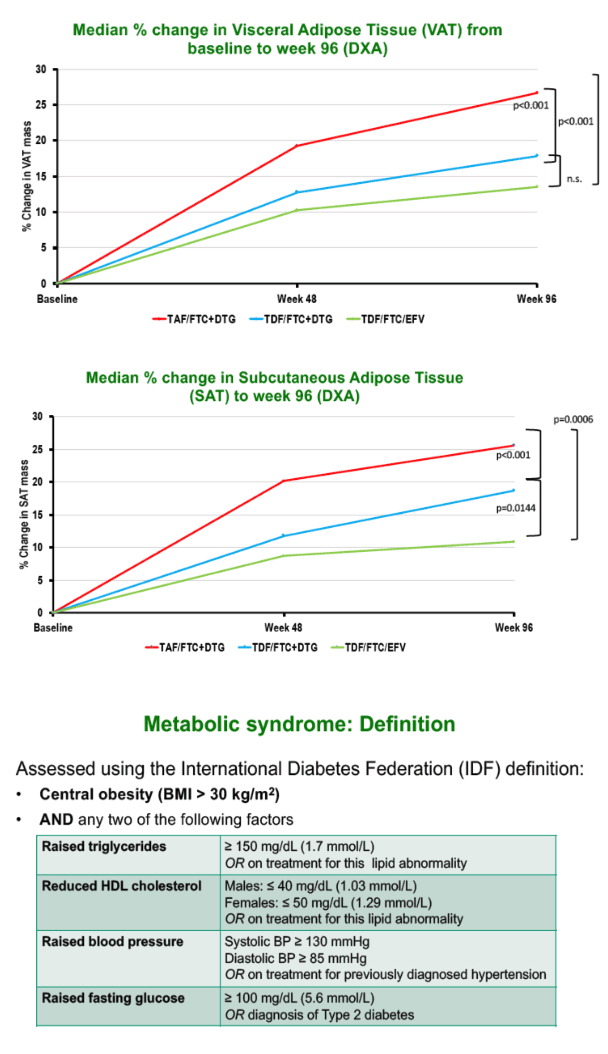
Median low-density lipoprotein (LDL) cholesterol rose significantly more with TAF/FTC+DTG than with TDF/FTC+DTG across 96 weeks (0.22 versus 0.06 mmol/L, P = 0.007), as did total cholesterol (0.27 versus 0.04 mmol/L, P = 0.007) and systolic blood pressure (3.0 versus -1.0 mmHg, P = 0.030). VAT rose significantly more through 96 weeks with TAF/FTC+DTG than with TDF/FTC+DTG (P < 0.001) or TDF/FTC+EFV (P < 0.001). SAT also climbed significantly more with TAF/FTC+DTG than with the other two regimens.
Prevalence of metabolic syndrome rose from 5% to 8% over 96 weeks with TAF/FTC+DTG, a greater change than with TDF/FTC+DTG (6% to 6%) or TDF/FTC+EFV (4% to 3%, P = 0.031). Changes in the Framingham Cardiovascular Risk Equation did not differ significantly between study arms through 96 weeks. But the QRISK equation, which estimates 10-year risk of MI or coronary death (https://qrisk.org/three/), stood significantly higher at week 96 in the TAF/FTC+DTG arm than in the TDF/FTC+EFV arm (+0.20% versus +0.10%, P = 0.027).
The QDIABETES Equation (https://qdiabetes.org) rose significantly more with TAF/FTC+DTG than with TDF/FTC+DTG through 96 weeks (+0.90% versus +0.50%, P = 0.004).
The ADVANCE team noted that these comparisons of MI and diabetes risk are limited because of the young age of the study population. They added that weight continued to rise through 96 weeks in women taking TAF/FTC+DTG, with no sign of a plateau, and the predictive models used did not account for additional weight gain after week 96.
The investigators estimated that people randomized to TAF/FTC+DTG would have 3 more cases of diabetes per 1000 people than participants randomized to TDF/FTC+DTG. They urged colleagues to repeat these analyses in other trials and patient populations taking TAF/FTC plus an integrase inhibitor.
In 2019 the World Health Organization revised its first-line antiretroviral guidelines, making the first choice TDF/(FTC or 3TC) + DTG [2]. TAF "may be considered [in first-line regimens] for patients with established osteoporosis and/or impaired renal function."
References
1. Hill A, McCann K, Qavi A, et al. Risks of metabolic syndrome, diabetes, and cardiovascular disease in ADVANCE trial. Conference on Retroviruses and Opportunistic Infections (CROI). March 8-11, 2020. Boston. Abstract 81.
2. WHO. Update of recommendations on first-and second-line antiretroviral regimens. July 2019. https://apps.who.int/iris/bitstream/handle/10665/325892/WHO-CDS-HIV-19.15-eng.pdf


|
| |
|
 |
 |
|
|