 |
 |
 |
| |
DRUG LEVELS, ADHERENCE, AND RISKS FOR
LOW ADHERENCE IN THE DISCOVER PrEP STUDY
|
| |
| |
CROI 2020 March 11
Reported by Jules Levin

program abstract
Susanne Doblecki-Lewis1, Olamide Dosekun2, Moti Ramgopal3, Jay Gladstein4, Mezgebe Berhe5, Kevin Nguyen6, Jason Hindman6, Yongwu Shao6, Ramin Ebrahimi6, Diana Brainard6, Moupali Das6, Scott McCallister6, Adriano Lazzarin7, Gerold F. Lang8, Peter L. Anderson9
1University of Miami, Miami, FL, USA, 2Imperial College Healthcare NHS Trust, London, UK, 3Midway Immunology and Research Center, Fort Pierce, FL, USA, 4APLA Health, Los Angeles, CA, USE, 5North Texas Infectious Diseases Consultants, Dallas, TX, USA, 6Gilead Sciences, Inc, Foster City, CA, USA, 7Ospedale San Raffaele, Milano, Italy, 8Dr med unvi Gerold Felician Lang, Vienna, Austria, 9University of Colorado, Aurora, CO, USA
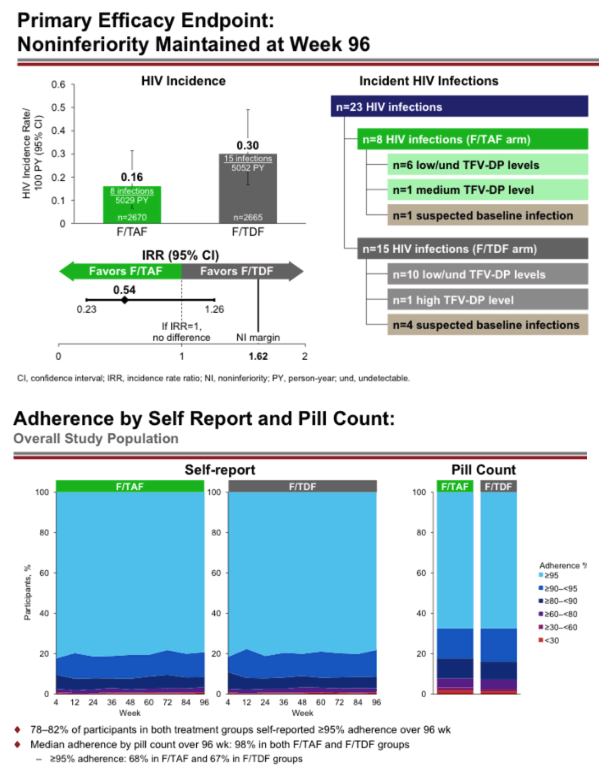
Background: In over 8,700 person-years (PY) follow up in the DISCOVER PrEP trial, the HIV incidence rates in the emtricitabine/tenofovir alafenamide (F/TAF) and emtricitabine/tenofovir disoproxil fumarate (F/TDF) arms were 0.16 and 0.34/100 PY, demonstrating noninferiority for HIV prevention. Study investigators and site staff provided comprehensive adherence support to study participants at all visits.
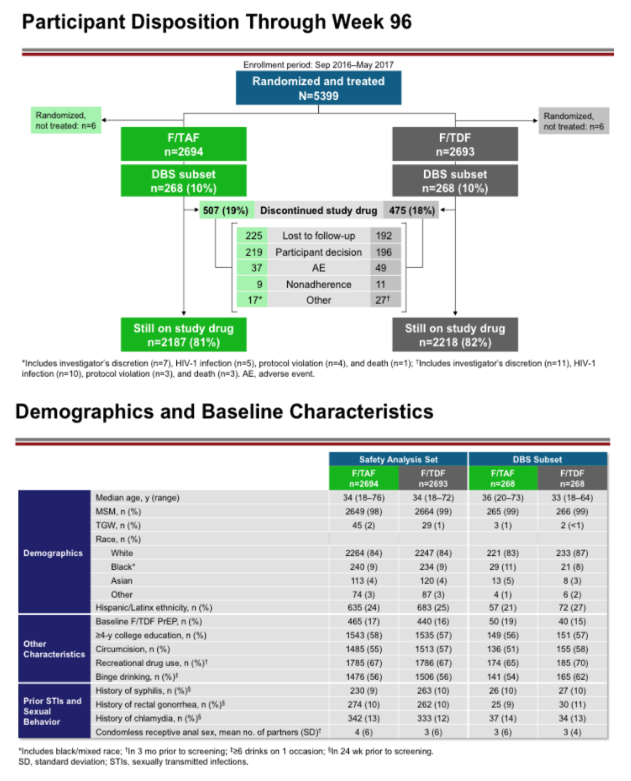
Methods: 5,387 men who have sex with men (MSM) and transgender women (TGW) at high HIV risk were randomized 1:1 to receive blinded once daily F/TAF or F/TDF. At all visits, adherence was assessed by self-report on a confidential questionnaire and also by pill count. Dried blood spot analyses of tenofovir diphosphate (TFV-DP) in red blood cells (RBCs) were assessed in a randomly selected subset of 10% participants, and in any participant who acquired HIV; peripheral blood mononuclear cell (PBMC) TFV-DP levels were assessed at W4 in the same subset. Adherence support included adherence counseling at each visit, personal communications from site staff as needed, optional text messaging daily, and email updates periodically.
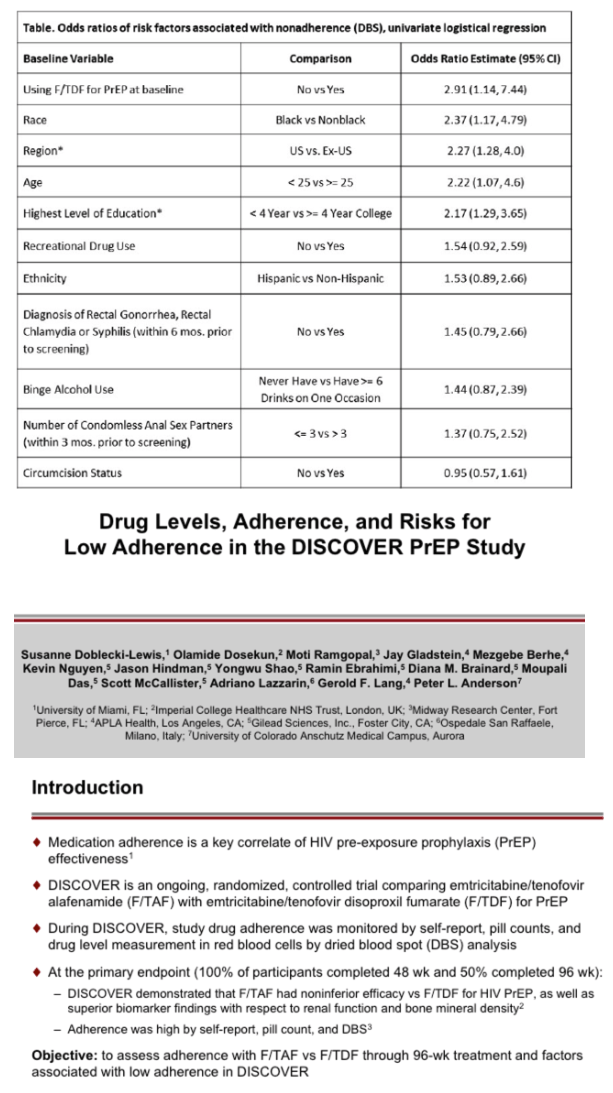
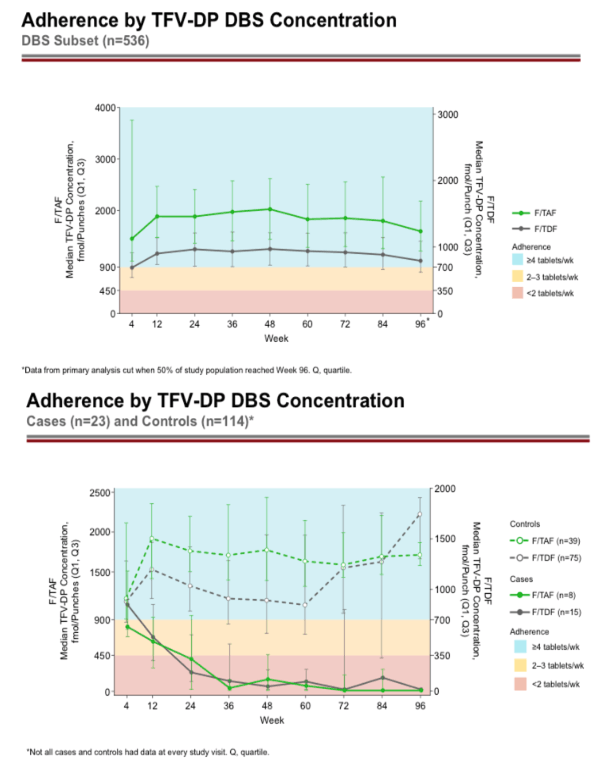
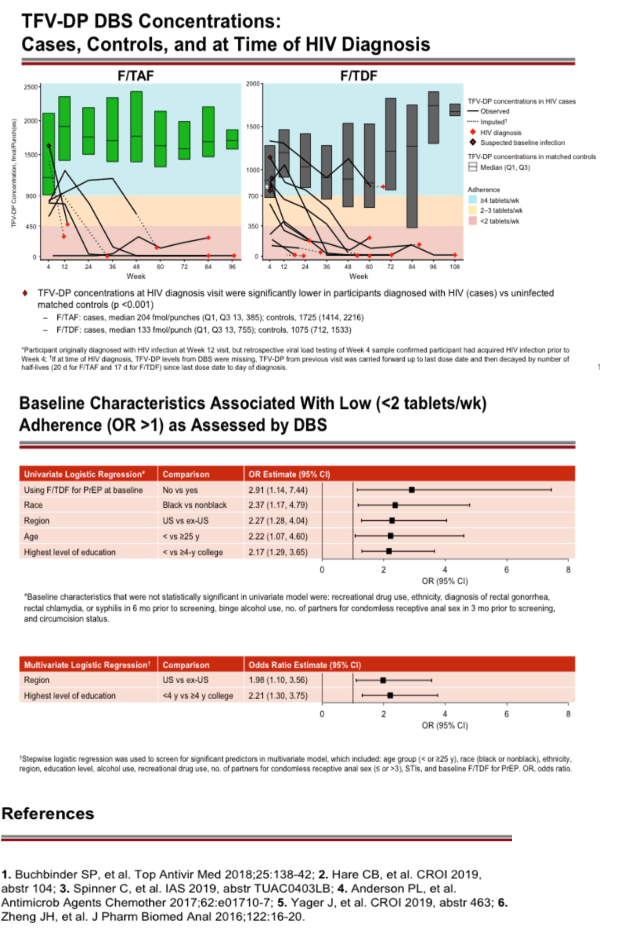
Results: 17 of the 22 HIV infections diagnosed in DISCOVER occurred while on study; 5/22 were suspected baseline infections. Of the 17 HIV infections that occurred on study, 6/17 occurred in the F/TAF arm and 11/17 occurred in the F/TDF arm. In 15 of the 17 on study HIV infections, DBS testing demonstrated that participants had undetectable or low TFV-DP levels in RBCs. By univariate logistical regression analysis, 5 baseline variables were significantly associated with low adherence by DBS (see Table); 2/5 were selected by multivariate stepwise analysis (asterisks). In both arms, adherence of at least 95% was >80% by self-report and was 69% by pill count. In the F/TAF and F/TDF arms respectively, 86-96% and 84-93% of participants were using at least 4 tablets/week, as measured by TFV-DP levels in RBCs. Levels of TFV-DP in PBMCs strongly correlated with tablets per week adherence TFV-DP levels in RBCs.
Conclusion: DISCOVER participants had very high adherence and very low HIV incidence rates. TFV-DP levels in RBCs were significantly lower in those with low adherence. The most important risk factor for acquisition of HIV on study was low adherence. Not using PrEP at baseline, black race, US residence, age below 25, and less than 4 years of college were significant risks for having low adherence to study drugs.

|
| |
|
 |
 |
|
|