 |
 |
 |
| |
DOSE-RESPONSE RELATIONSHIP OF SUBCUTANEOUS LONG-ACTING HIV CAPSID INHIBITOR GS-6207
|
| |
| |
Gilead will be initiating enrollment of two new clinical trials of GS-6207 in combination with other antiretroviral agents in people living with HIV - a Phase 2/3 study (NCT03739866) in heavily treatment-experienced people living with multidrug resistant HIV-1, as well as a Phase 2 study (NCT04143594) in treatment-naïve people living with HIV. GS-6207 will be administered via a two-week oral lead-in, followed by a subcutaneous injection every six months.
....https://www.gilead.com/news-and-press/press-room/press-releases/2019/11/gilead-presents-data-on-investigational-hiv-1-capsid-inhibitor-gs-6207-as-a-potential-component-of-long-acting-hiv-therapy
CROI 2020 March 8-11
Reported by Jules Levin
Eric Daar1, Cheryl McDonald2, Gordon Crofoot3, Peter Ruane4, Gary Sinclair5, Edwin DeJesus6, Mezgebe Berhe7, Moti Ramgopal8, Heena Patel9, Ya-Pei Liu9, Rebecca Begley9, Diana Brainard9, Robert H. Hyland9, Martin Rhee9
1Harbor-UCLA Medical Center, Torrance, CA, USA, 2Tarrant County Infectious Disease Associates, Fort Worth, TX, USA, 3Crofoot Research Center, Houston, TX, USA, 4Peter J Ruane, MD Inc, Los Angeles, CA, USA, 5AIDS Arms, Inc, Dallas, TX, USA, 6Orlando Immunology Center, Orlando, FL, USA, 7North Texas Infectious Diseases Consultants, Dallas, TX, USA, 8Midway Immunology and Research Center, Fort Pierce, FL, USA, 9Gilead Sciences, Inc, Foster City, CA, USA
program abstract
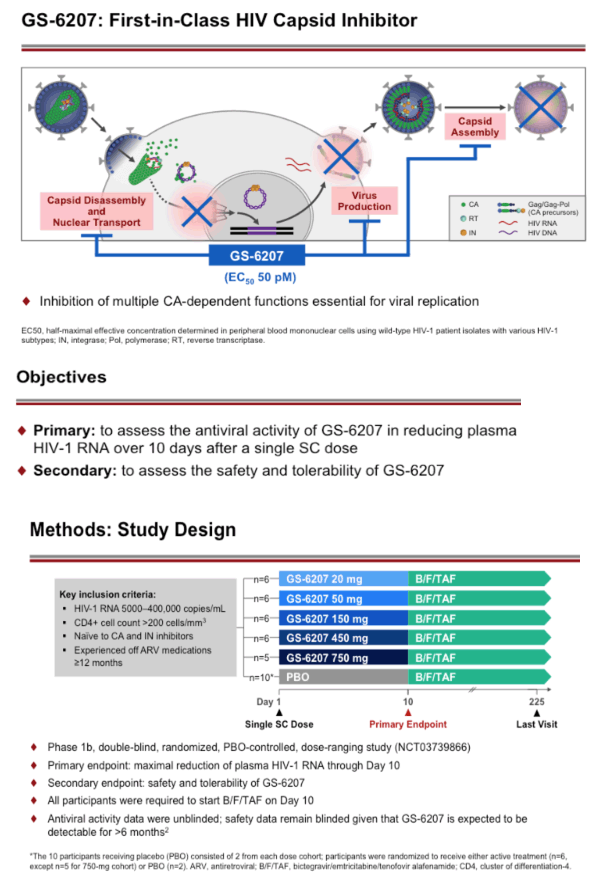
Background: GS-6207, a potent, selective, first-in-class, multi-stage inhibitor of HIV-1 capsid function that inhibits HIV at picomolar concentrations and is in development as a long-acting agent for treatment of HIV-1 infection. The safety, antiviral activity and pharmacokinetics (PK) of GS-6207 were evaluated in people living with HIV (PLWH) in this Phase 1b study.
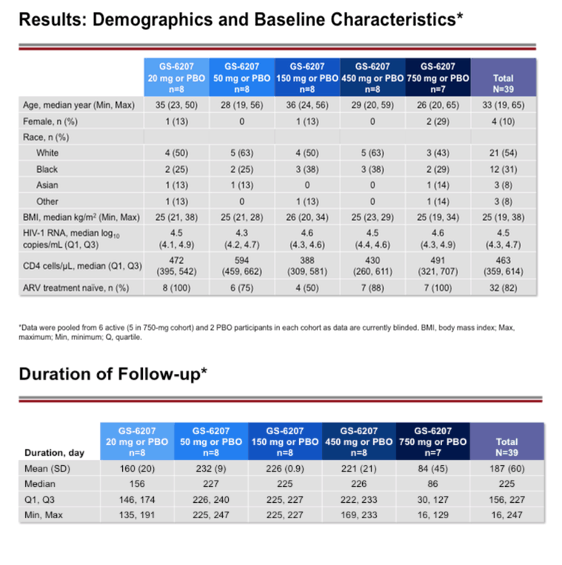
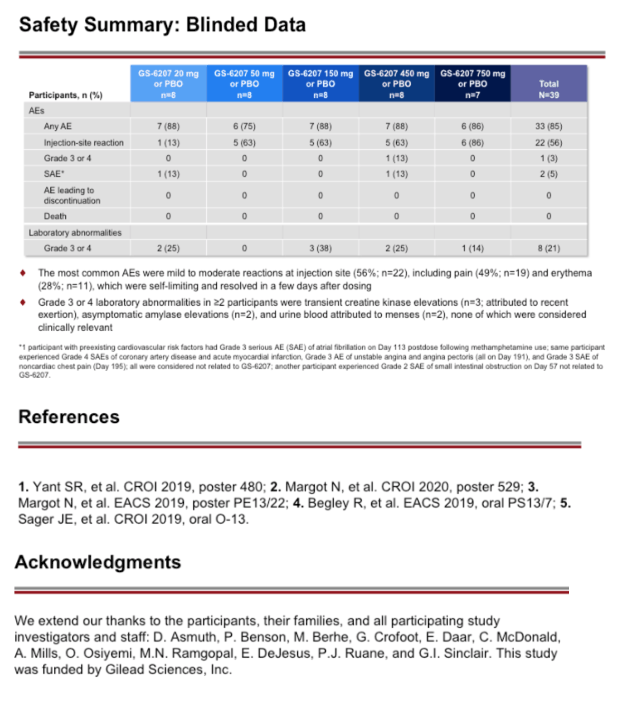
Methods: This is an ongoing, Phase 1b, randomized, double-blinded, placebo-controlled dose-ranging study of GS-6207 in HIV capsid-inhibitor naive PLWH who are not taking antiretroviral therapy. A single subcutaneous (SC) dose of GS-6207 (20, 50, 150, 450, or 750 mg; N=6/cohort) or placebo (N=2/cohort) was administered. The primary endpoint was maximum reduction of plasma HIV-1 RNA through post dose day 10 (D10). Safety was assessed using laboratory tests and adverse event (AE) reporting. We present antiviral activity, blinded safety, and dose-response relationship for the 20 to 450 mg dose cohorts; enrollment of the 750 mg cohort is ongoing.
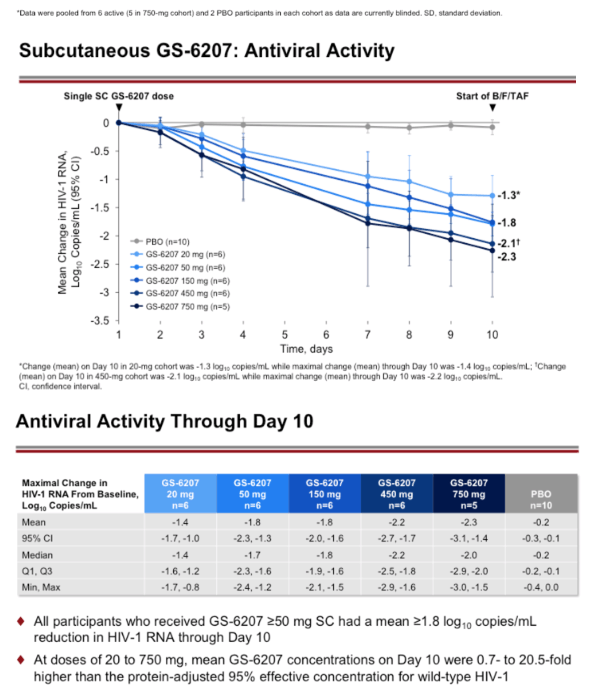
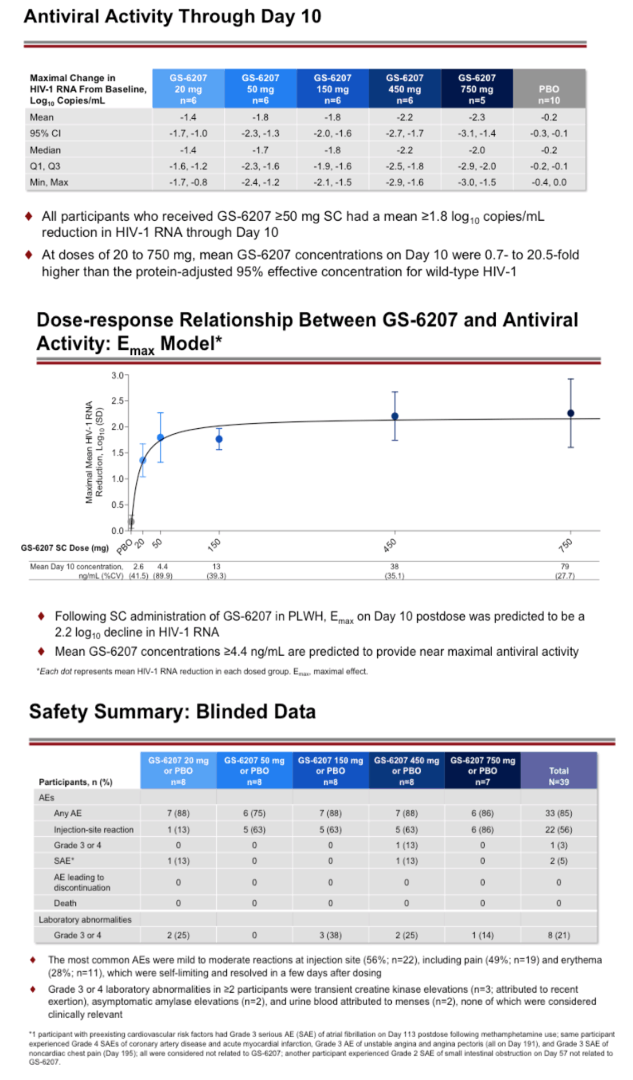
Results: Demographics and baseline characteristics were similar across groups (N=32, n=8 per group). All PLWH who received active drug had significantly greater reductions in HIV-1 RNA by D10 than the placebo (all p<0.0001). The 50 to 450 mg groups had a numerically greater mean reductions in HIV-1 RNA through D10 (range: 1.8 to 2.2 log10copies/mL) than the 20 mg group (1.4 log10copies/mL). At these doses, the inhibitory quotients (mean GS-6207 concentrations for each group/protein adjusted 95% maximal effective concentration in MT-4 cells for wild type HIV-1) on D10 ranged from 0.7 to 9.9. Using a maximum effect (Emax) model for GS-6207 (SC 20 to 450 mg) and antiviral activity, Emax was ∼2.1 log10copies/mL decline in HIV-1 RNA, and a dose inhibiting viral replication by 50% (ED50) was ∼10 mg. One participant experienced a serious AE (Grade 3) of atrial fibrillation after using methamphetamine; no other SAEs, Grade 3 or 4 AEs, AEs leading to discontinuation, or clinically relevant Grade 3 or 4 laboratory abnormalities were reported. The most common AEs were injection site reactions that were mostly mild and transient (50%).
Conclusion: In PLWH, GS-6207 demonstrated potent antiviral activity, with up to a 2.2 log10 copies/mL decline at Day 10, and was generally safe and well tolerated. These results support further clinical evaluation of GS-6207 as a long-acting antiretroviral agent.

|
| |
|
 |
 |
|
|