 |
 |
 |
| |
PK, Food Effect, and Safety of Oral GS-6207, a Novel HIV-1 Capsid Inhibitor
|
| |
| |
CROI 2020 March 8-11
Reported by Jules Levin
Rebecca Begley, Martin S. Rhee, Steve K. West, Jessica Corpus, John Ling, Polina German — Gilead Sciences, Inc., Foster City, CA

Abstract Body:
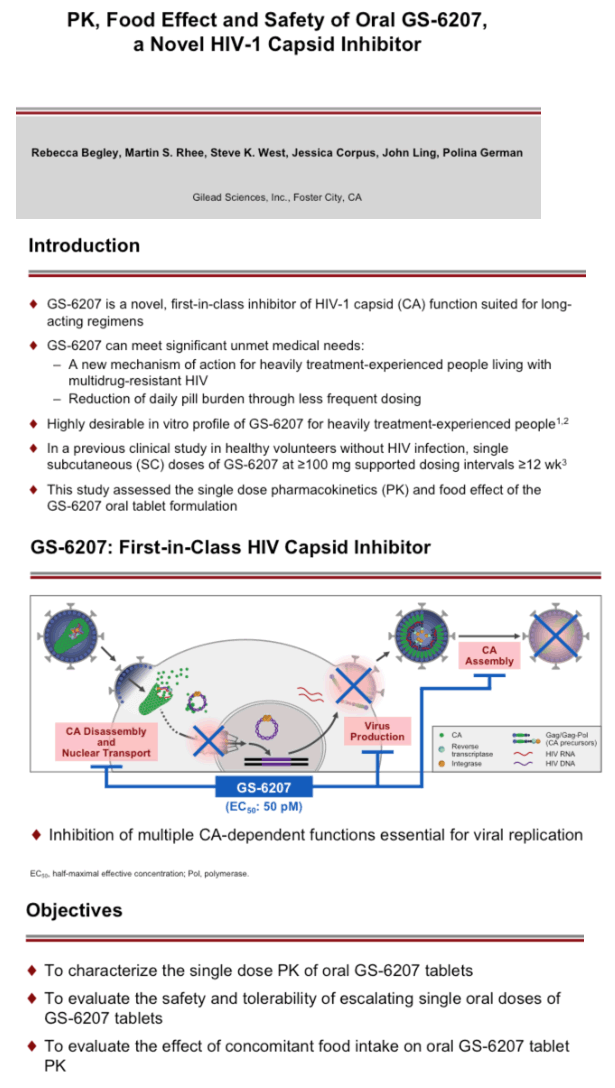
GS-6207, a potent, selective, first-in-class, multi-stage inhibitor of HIV-1 capsid function is in development for treatment of HIV-1 infection as a long acting agent. In people living with HIV (PWH), GS-6207 administered subcutaneously (SC) has shown potent antiviral activity, and is generally well tolerated. In addition to GS-6207 SC formulations, an oral tablet formulation is also in development. The safety, single ascending dose (SAD) pharmacokinetics (PK) and effect of food (FE) on GS-6207 oral tablets were evaluated in HIV negative participants.
This is an ongoing, blinded, placebo-controlled Phase 1 study with staggered SAD and open label FE cohorts. In each SAD cohort subjects were randomized (4:1) to receive single doses of GS‑6207 (n=8/cohort) or placebo (n=2/cohort), at 50, 300, 900 or 1800 mg. In the FE cohorts (n=8/cohort), subjects received GS-6207 300 mg following a high fat (∼1000 kcal; ∼50% fat) or low fat (∼400 kcal; ∼25% fat) meal. Intensive PK sampling will be performed for 64 days post-dose. Single dose PK parameters were estimated using noncompartmental methods using available data; dose proportionality and FE were assessed. Safety was evaluated throughout the study.
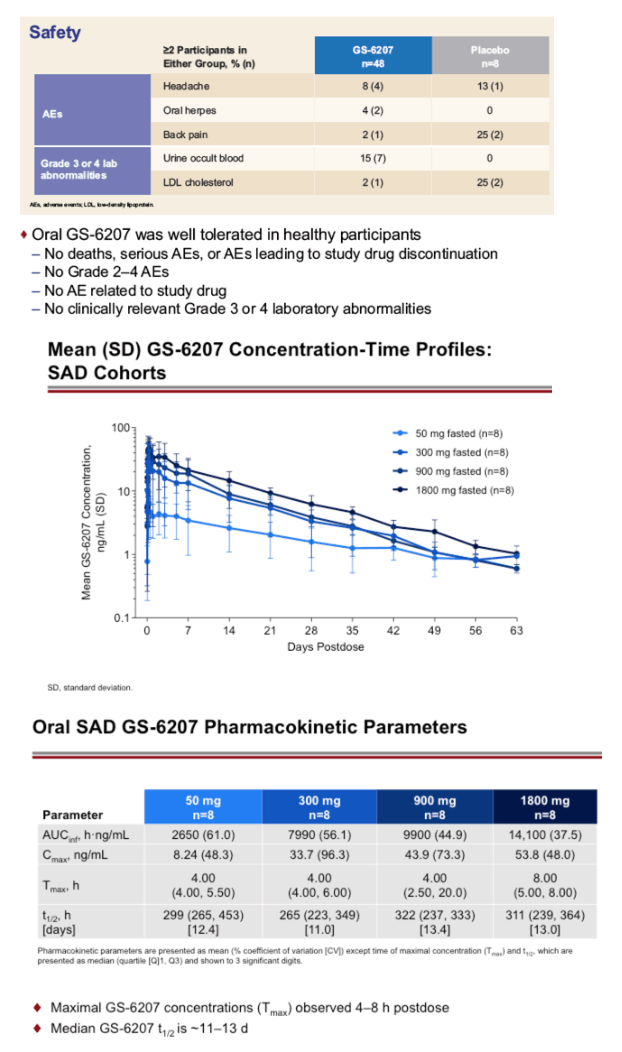
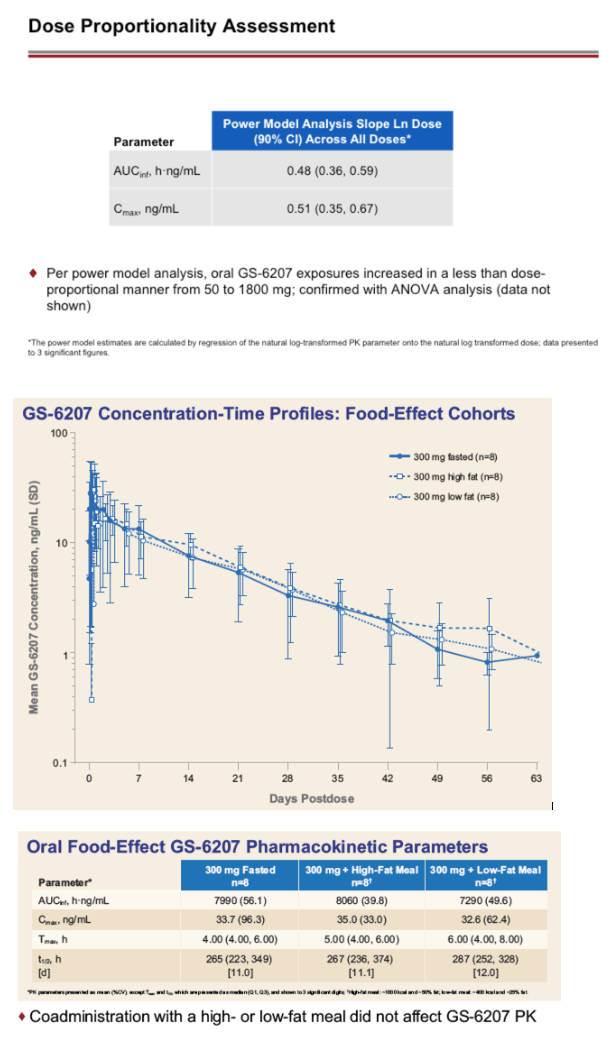
Interim safety and PK data are available through 35 (300 and 900 mg fasted) or 8 days post dose (50 and 1800 mg fasted, 300 mg high and low fat). 56 of 56 participants completed dosing. GS-6207 oral tablets were generally well tolerated. No serious adverse events (AEs), Grade 3 or 4 AEs, or discontinuations due to AEs were reported. The most common AEs were back pain (n=2) and headache (n=3); all Grade 1. Based on the available data, GS-6207 exposures increased in a less than dose-proportional manner over the dose range of 50 to 1800 mg. Maximal concentrations (Cmax) of GS-6207 were achieved ∼4 to 8 h post dose (Tmax), and GS-6207 t1/2 was ∼12 days. Exposure (Cmax and AUC0-D8) and time to maximal exposure (Tmax) were not affected by administration following a high or low fat meal, supporting dosing of GS-6207 tablets without regard to food (Table 1).
The preliminary PK and safety data suggest GS-6207 oral tablets are well tolerated following single oral doses up to 1800 mg, and can be dosed without regards to food. These data support ongoing clinical development of oral GS-6207 for use in PWH.


|
| |
|
 |
 |
|
|