 |
 |
 |
| |
Starting ART With a PI or an InSTI (Mainly Raltegravir)
May Heighten Risk of Diabetes
|
| |
| |
CROI 2020, March 8-11, 2020, Boston
Mark Mascolini
Starting antiretroviral therapy (ART) with a protease inhibitor (PI) or an integrase inhibitor (InSTI) may boost the risk of newly diagnosed diabetes, according to results of a 23,000-person North American analysis [1]. Weight gain in the first year of ART only partly explained the higher diabetes risk.
Previous work linked starting an InSTI-containing first antiretroviral regimen to weight gain, but in this context the impact of InSTIs and other antiretrovirals on metabolic changes remained poorly understood. To address these questions, NA-ACCORD investigators analyzed antiretroviral-naive adults starting a regimen based on an InSTI, a PI, or a nonnucleoside (NNRTI) in 2007-2016. People included in the weight analysis had to have a weight measure 12 months after starting ART, while the analysis excluded people with diabetes diagnosed during the first 12 months of therapy. Cox regression analysis identified predictors of newly diagnosed (incident) diabetes after adjustment for age, sex, race, HIV transmission risk, year ART began, and pre-ART weight, CD4 count, and viral load.
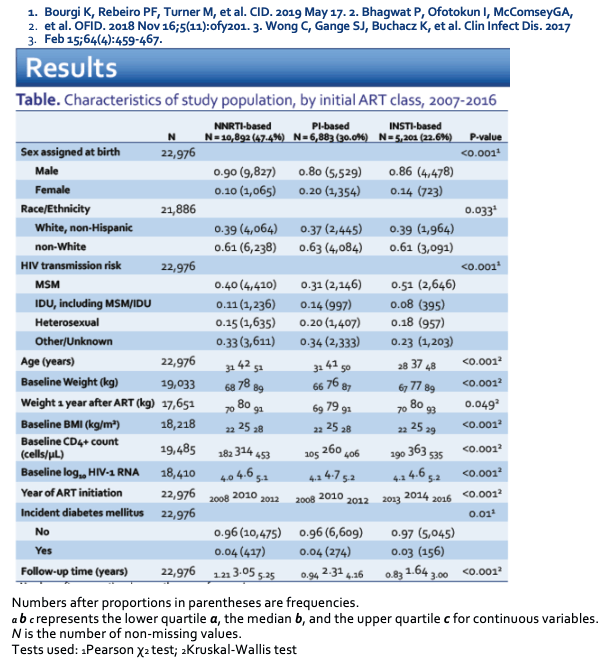
The 22,976 study participants included 5201 (22.6%) starting an InSTI, 6883 (30%) starting a PI, and 10,892 (47.4%) starting an NNRTI. The NNRTI group included a higher proportion of men than the PI group or the InSTI group (90%, 80%, 86%, P < 0.001), had a higher median age (42, 41, 37, P < 0.001), and had a higher baseline weight (78, 76, 77 kg, P < 0.001). The NNRTI and PI groups started ART in 2010 (median), compared with 2014 for the InSTI group (P < 0.001). Median follow-up time was longer with NNRTIs (3.05 years) versus PIs (2.31 years) or InSTIs (1.64 years) (P < 0.001). Diabetes incidence was slightly but significantly greater in the NNRTI and PI groups (4% for both) than in the InSTI group (3%) (P = 0.01).
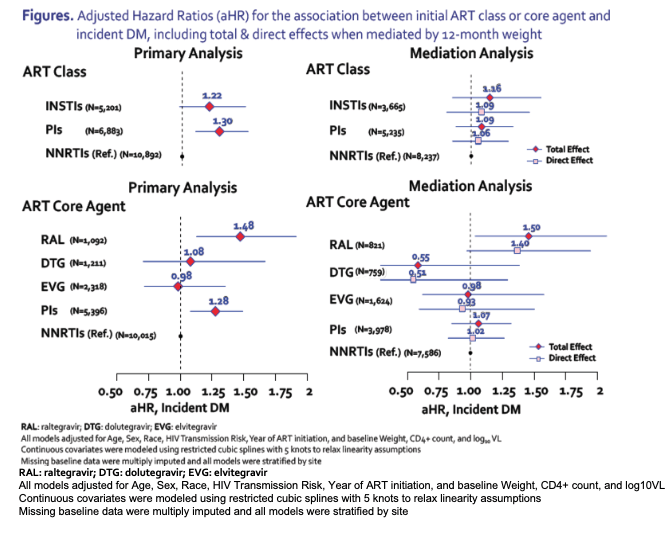
Multivariate Cox analysis determined that, compared with people starting an NNRTI, those starting a PI had a 30% higher risk of incident diabetes (adjusted hazard ratio [aHR] 1.30) and those starting an InSTI had a 22% higher risk (aHR 1.22). The difference between PIs and NNRTIs reached statistical significance, while the difference between InSTIs and NNRTIs fell just short of statistical significance. An analysis also adjusted for 12-month weight gain determined that weight gain only modestly attenuated the impact of PIs on incident diabetes (aHR 1.09 versus 1.06) or InSTIs on incident diabetes (aHR 1.16 versus 1.09).
When the NA-ACCORD researchers ran the same kind of analyses considering individual InSTIs, they found that raltegravir independently predicted greater risk of incident diabetes compared with NNRTIs (aHR 1.48), while dolutegravir (aHR 1.08) and elvitegravir (aHR 0.98) did not. In the same analysis, PIs as a group were independently associated with a 28% greater risk of new diabetes than NNRTIs (aHR 1.28). Considering 12-month weight gain in this analysis only moderately tempered the impact of raltegravir on diabetes risk (aHR from 1.50 to 1.40). Considering weight in the analysis had even smaller impacts on the associations recorded for dolutegravir, elvitegravir, or PIs as a group.
The NA-ACCORD team concluded that starting ART with a PI or an InSTI, particularly raltegravir, "may confer greater risk of incident diabetes mellitus." Weight gain through the first 12 months of therapy could only partly explain those increased risks.
Reference
1. Rebeiro PF, Jenkins CA, Bian A, et al. Diabetes, weight gain, and integrase inhibitor use in North American HIV+ persons. Conference on Retroviruses and Opportunistic Infections (CROI). March 8-11, 2020. Boston. Abstract 680.
---------------------
Diabetes, Weight Gain, & Integrase Inhibitor Use in North American HIV+ Persons
Peter F. Rebeiro1, Cathy A. Jenkins1, Aihua Bian1, Jordan E. Lake2, KassemBourgi3, Keri N. Althoff4, Michelle Floris-Moore5
Michael A. Horberg6, Janet Tate7, Amanda Willig8, Richard D. Moore4, Stephen J. Gange4, Timothy R. Sterling1, and John R. Koethe1 for the NA-ACCORD
1. Vanderbilt University School of Medicine, Nashville, TN; 2. University of Texas at Houston, Houston, TX; 3. Indiana University, Indianapolis, IN; 4. Johns Hopkins Bloomberg School of Public Health, Baltimore, MD;
5. University of North Carolina at Chapel Hill, Chapel Hill, NC; 6. Kaiser Permanente Mid-Atlantic States, Rockville, MD; 7.VA Connecticut Healthcare System, West Haven, CT; 8. University of Alabama at Birmingham, Birmingham, AL


|
| |
|
 |
 |
|
|