 |
 |
 |
| |
Development of a Highly Sensitive Multiplex
Platform Assay to Monitor Low Levels of HBV DNA and pgRNA
|
| |
| |
EASL 2020 Aug 27-29 virtual
Reported by Jules Levin
Qi Huang, Ran Yan, DaweiCai, Ariel Tang, Xiang Xu and Richard Colonno
Assembly Biosciences, Inc., South San Francisco, CA, United States



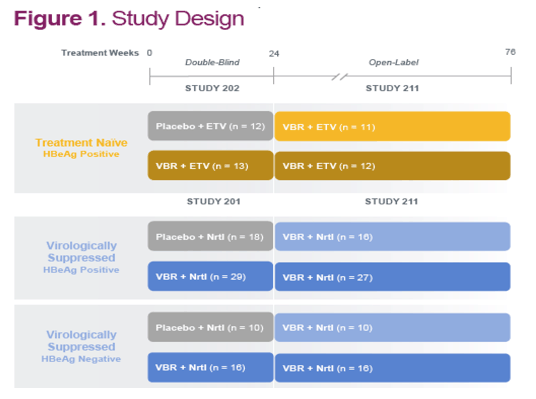

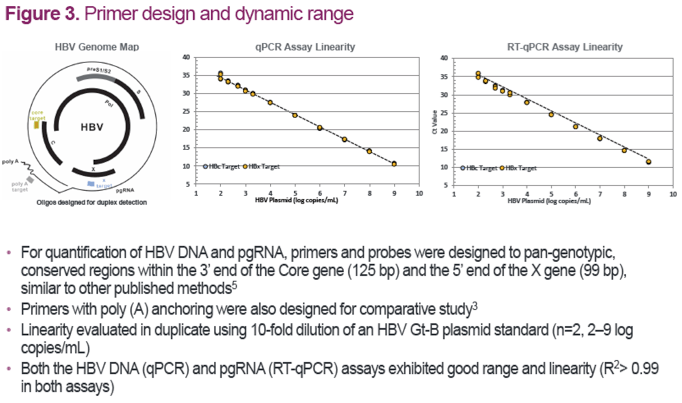
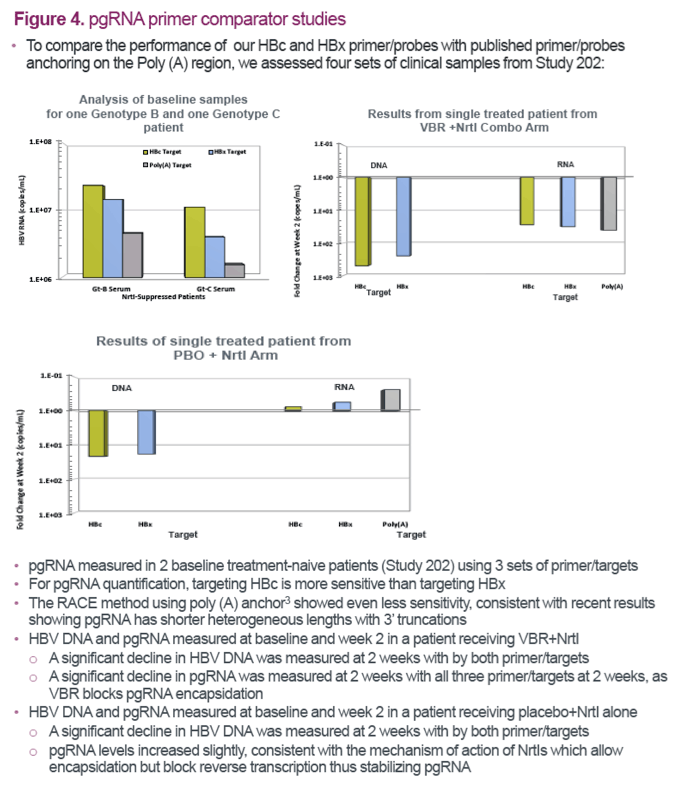
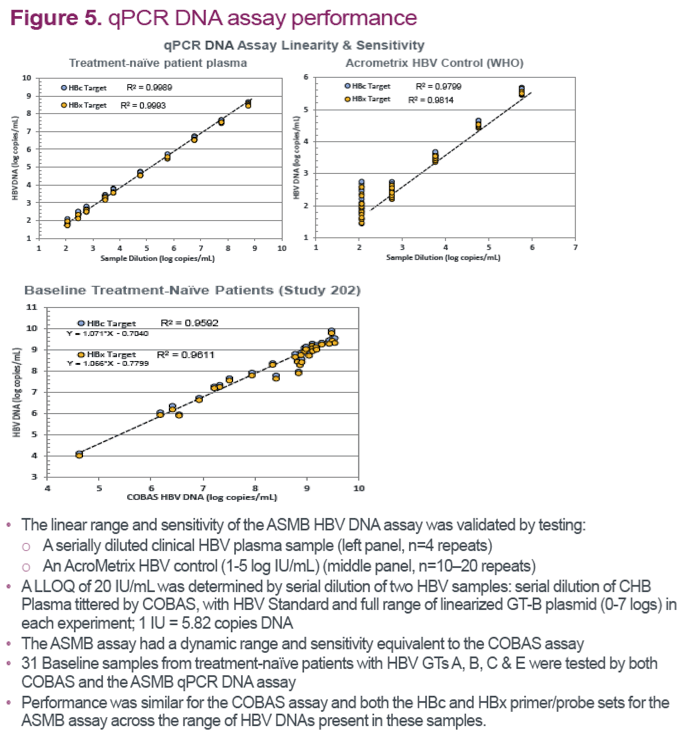
References
1) Fan R, Hou J et al. Association Between Negative Results From Tests for HBV DNA and RNA and Durability of Response After Discontinuation of Nucles(t)ide Analogue Therapy. Clinical Gastroenterology and Hepatology; 2) Carey I, GerschJ, Wang B, et al. Pre-genomic HBV RNA and HBcrAgpredict outcomes in HBeAgnegative chronic hepatitis B patients suppressed on nucleos(t)ide analogue therapy [published online ahead of print, 2019 Nov 7]. Hepatology. 2019;10.1002/hep.31026; 3) van BömmelF, etcSerum hepatitis B virus RNA levels as an early predictor of hepatitis B envelope antigen seroconversion during treatment with polymerase inhibitors. Hepatology. 2015;61:66-76; 4) Marcellin P, GaneEJ, FlisiakR, MannsMP, KaitaK, GaggarA, et al. Evidence for ongoing low-level viremia in patients with chronic hepatitis B receiving long-term nucleos(t)ide analog therapy. HEPATOLOGY 2014;60:1093A; 5) Butler EK, etc. Hepatitis B Virus Serum DNA and RNA Levels in Nucleos(t)ide Analog-Treated or Untreated Patients During Chronic and Acute Infection. Hepatology. 2018; 68: 2106-2117; 6) Saldanha J, GerlichW., LelieN., Dawson P., HeermannK., Heath A. and The WHO Collaborative Study Group. An international collaborative study to establish a World Health Organization international standard for hepatitis B virus DNA nucleic acid amplification techniques. Vox Sanguinis 2001 80, 63-71; 7) Yuen MF et al. Safety, pharmacokinetics, and antiviral effects of ABI-H0731, a hepatitis B virus core inhibitor: a randomised, placebo-controlled phase 1 trial. Lancet Gastroenterol Hepatol. 2019 Nov 8. pii: S2468-1253 (19) 30346-2; 8) Ma, X. et al. Interim Safety and Efficacy Results of the ABI-H0731 Phase 2a Program Exploring the Combination of ABI-H0731 with NucTherapy in Treatment-Naive and Treatment-Suppressed Chronic Hepatitis B Patients. 2019 EASL.
|
| |
|
 |
 |
|
|